No: Measurement Is Unnecessary and a Poor Use of Medical Resources
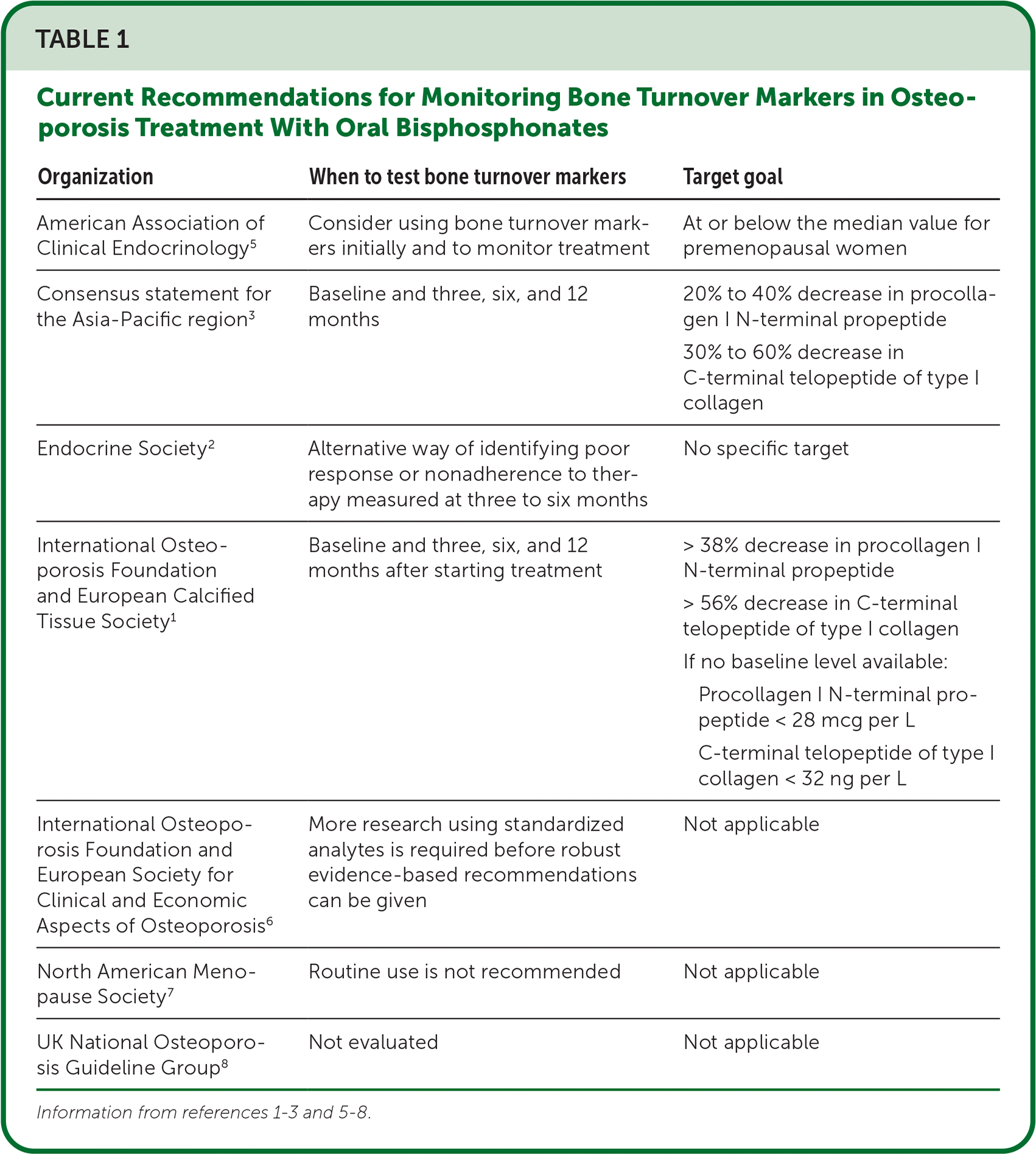
The International Osteoporosis Foundation, the Endocrine Society, and the consensus statement for the Asia-Pacific Region recommend measuring bone turnover markers to assess effectiveness of and adherence to osteoporosis therapy.1–3 The recommended bone turnover markers are procollagen I N-terminal propeptide and C-terminal telopeptide of type I collagen.4 The organizations recommend measuring these markers at baseline, three to six months after starting oral antiresorptive therapy, and, if levels are reduced by a certain amount, annually thereafter (Table 1).1–3,5–8 If bone turnover markers do not change, then physicians should suspect nonadherence to bisphosphonate therapy.
TABLE 1. Current Recommendations for Monitoring Bone Turnover Markers in Osteoporosis Treatment With Oral Bisphosphonates
| Organization | When to test bone turnover markers | Target goal |
|---|---|---|
| American Association of Clinical Endocrinology5 | Consider using bone turnover markers initially and to monitor treatment | At or below the median value for premenopausal women |
| Consensus statement for the Asia-Pacific region3 | Baseline and three, six, and 12 months | 20% to 40% decrease in procollagen I N-terminal propeptide 30% to 60% decrease in C-terminal telopeptide of type I collagen |
| Endocrine Society 2 | Alternative way of identifying poor response or nonadherence to therapy measured at three to six months | No specific target |
| International Osteoporosis Foundation and European Calcified Tissue Society1 | Baseline and three, six, and 12 months after starting treatment | > 38% decrease in procollagen I N-terminal propeptide > 56% decrease in C-terminal telopeptide of type I collagen If no baseline level available: Procollagen I N-terminal propeptide < 28 mcg per L C-terminal telopeptide of type I collagen < 32 ng per L |
| International Osteoporosis Foundation and European Society for Clinical and Economic Aspects of Osteoporosis6 | More research using standardized analytes is required before robust evidence-based recommendations can be given | Not applicable |
| North American Menopause Society 7 | Routine use is not recommended | Not applicable |
| UK National Osteoporosis Guideline Group8 | Not evaluated | Not applicable |
Bone Turnover Markers Are Unlikely to Uncover Treatment Ineffectiveness
It is uncommon for patients who regularly take an oral bisphosphonate to not respond to treatment. Fewer than 10% of patients taking bisphosphonates have no significant improvement in bone density based on repeat dual energy x-ray absorptiometry testing.9 More than 70% of patients respond to oral bisphosphonates, as shown by significant decreases in markers.10 In the TRIO study, the response rate at three months was high for the three oral bisphosphonates, alendronate, ibandronate, and risedronate; by 12 weeks, 70% to 90% of patients responded.10
Another argument for routinely measuring bone turnover markers is that it is better to have some data from the markers than none at all.4 Studies have yet to find improved patient outcomes with their use; therefore, the assessment of bone turnover markers should currently be limited to specific populations (e.g., patients at very high fracture risk) and research studies evaluating their effect.
Can the Lack of Response Help Identify Patients With a Secondary Cause of Osteoporosis?
A secondary cause of osteoporosis is rarely found. Because there is no significant correlation between elevated bone turnover markers and endocrine diseases, the markers are not recommended to help physicians identify endocrine diseases in patients diagnosed with osteoporosis or osteopenia.11
Bone Turnover Markers Do Not Independently Improve Adherence
Measuring bone turnover markers continues to be touted as a way to improve adherence to treatment.4,12,13 Although nonadherence has plausibly been associated with fractures, one of the few studies that assessed the relationship did not find correlation between adherence and fracture outcomes at 52 weeks.14
Only three studies have looked at the effect of measuring bone turnover markers on adherence. In a small study that measured urine bone turnover markers in patients with osteopenia who were treated with a selective estrogen receptor modulator, nurse monitoring improved adherence, but measuring urine bone turnover markers did not result in further improvement.15 The largest study (more than 2,000 patients), which evaluated daily oral bisphosphonate and urine bone turnover markers, found a statistically significant difference in adherence, but this difference was marginal.16 The final study found that providing urine bone turnover marker results did not improve adherence.17 The latest systematic review regarding ways to improve adherence did not mention routine measurement of bone turnover markers; the review found that multicomponent interventions based on patient education and counseling were the most effective interventions to increase adherence to osteoporosis medications.18
Problems With the Measurement of Bone Turnover Markers
The process of bone turnover marker measurement is problematic.19 First, unless a baseline measurement is obtained, measuring bone turnover markers after the onset of oral bisphosphonate therapy is almost useless; second, individual and diurnal variations in levels can occur. Because bone turnover markers are affected by food, patients must be fasting for the test.20 Multivitamins or supplements containing biotin should be stopped 24 hours before measuring the markers.13 Additionally, patients who are obese have lower bone turnover markers at baseline.21
A decision analysis indicated that monitoring at three months after starting antiresorptive osteoporosis treatment could slightly increase the expected benefit of treatment.22 However, the cost to monitor all patients would be significant because commercial laboratories currently charge $75 to $250 per assay.23 Testing 100% of patients to find the 10% who do not respond or are nonadherent is unwise and expensive. Taking an interval history, performing a physical examination, and noting medication refill data could identify nonadherence.
Although our recommendation differs from some expert guidelines, it is consistent with many published opinions.7,23–25 Bone turnover markers are not needed to assess bisphosphonate effectiveness, and they do not have a direct impact on adherence.1 Family physicians can manage osteoporosis and monitor its treatment by talking to their patients and noting refill data. Routine measurement of bone turnover markers in patients treated with an oral bisphosphonate does not benefit patients and is a poor use of medical resources.
This is one in a series of pro/con editorials discussing controversial issues in family medicine.
A collection of Editorials: Controversies in Family Medicine published in AFP is available at https://www.aafp.org/afp/pro-con.

