Sleep disorders are common in the general adult population and are associated with adverse effects such as motor vehicle collisions, decreased quality of life, and increased mortality. Patients with sleep disorders can be categorized into three groups: people with problems falling asleep, people with behavior and movement disturbances during sleep, and people with excessive daytime sleepiness. Insomnia, the most common sleep disorder, is defined by difficulty initiating sleep, maintaining sleep, or both, resulting in daytime consequences. Insomnia is diagnosed by history and is treated with cognitive behavior therapy, with or without medications. Rapid eye movement sleep behavior disorder is characterized by increased muscle tone during rapid eye movement sleep, resulting in patients acting out their dreams with potentially harmful effects. Rapid eye movement sleep behavior disorder is diagnosed by polysomnography and treated with melatonin or clonazepam. Restless legs syndrome is defined by an urge to move the legs that worsens when at rest. Restless legs syndrome is treated with gabapentin or dopamine agonists, depending on the severity. Narcolepsy is characterized by excessive daytime sleepiness, cataplexy, sleep paralysis, and sleep hallucinations. Diagnosis is suggested by the history and can be confirmed with polysomnography and a multiple sleep latency test the following day. Narcolepsy is treated with behavior modifications and medications such as stimulants, selective serotonin reuptake inhibitors, sodium oxybate, and pitolisant. Obstructive sleep apnea may be diagnosed in patients with excessive snoring and witnessed apneas and can be diagnosed using overnight polysomnography. Treatment consists of positive airway pressure therapy while sleeping in conjunction with weight loss.
Sleep disorders are common in the United States, with the Institute of Medicine estimating that 50 million to 70 million adults report chronically disturbed sleep.1 Current recommendations for adult sleep duration range from seven to nine hours; however, nearly 40% of Americans sleep six hours or less per night.2 Sleep disturbances have been associated with motor vehicle collisions, hypertension, decreased quality of life, and increased all-cause mortality, which may be linked to increased arousal burden (unconscious wakefulness) with daytime sequelae.3,4 Sleep disorders have been connected to increased health care use and cost more than $94 billion per year.5 Family medicine physicians must regularly inquire about healthy sleep and should be able to diagnose and treat sleep disorders in the primary care setting.6
SORT: KEY RECOMMENDATIONS FOR PRACTICE
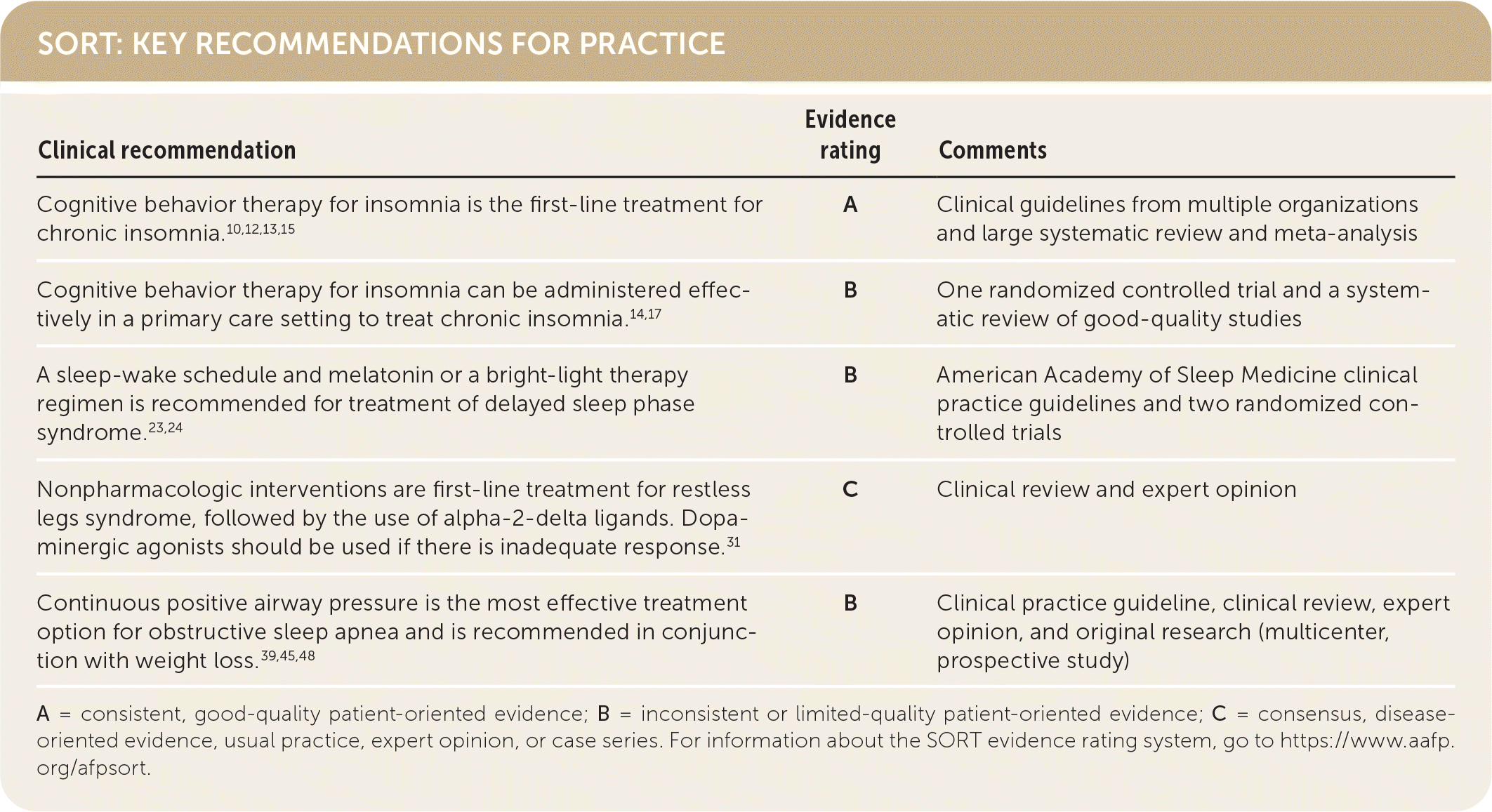
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
BEST PRACTICES IN SLEEP MEDICINE
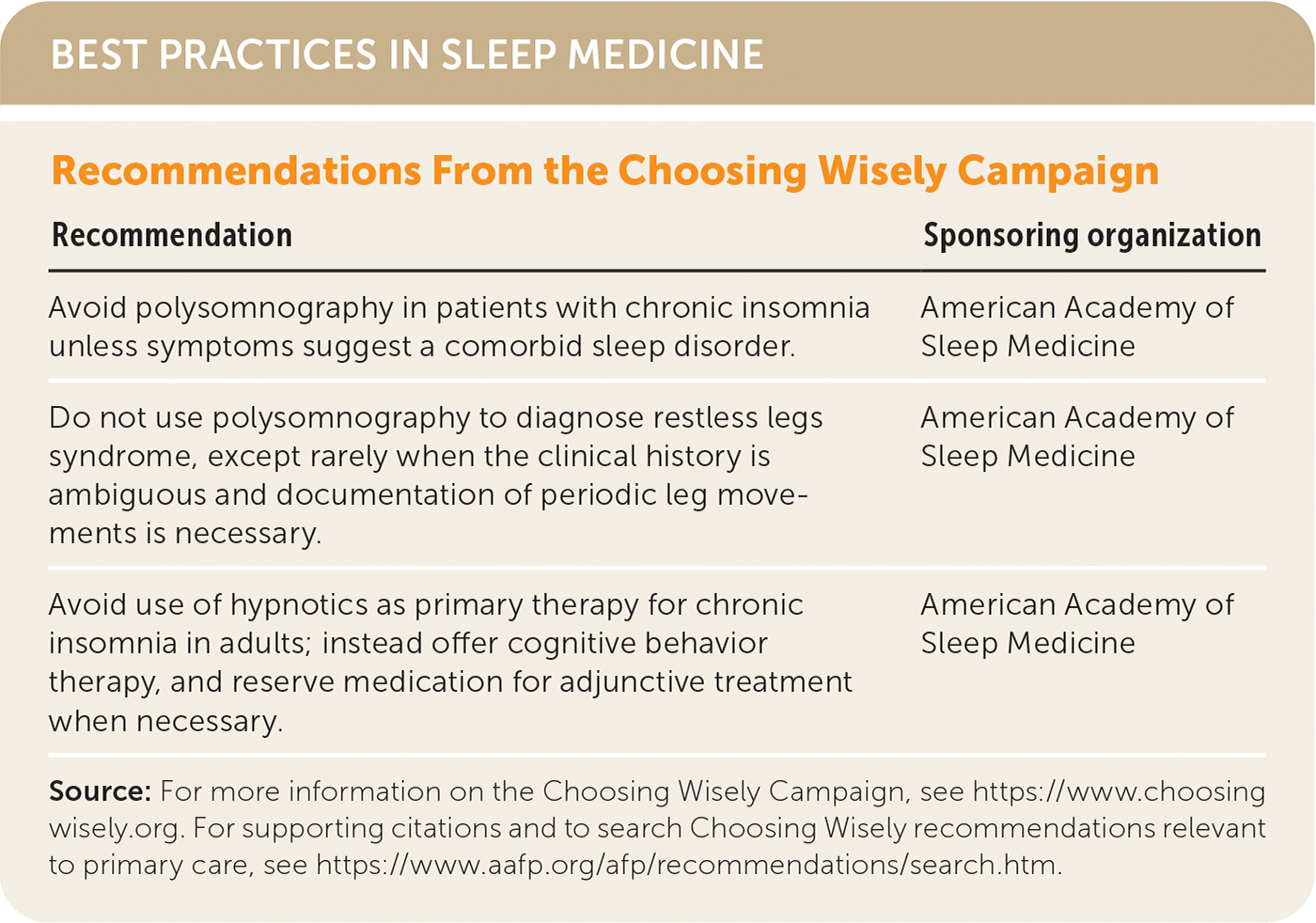
| Recommendation | Sponsoring organization |
|---|---|
| Avoid polysomnography in patients with chronic insomnia unless symptoms suggest a comorbid sleep disorder. | American Academy of Sleep Medicine |
| Do not use polysomnography to diagnose restless legs syndrome, except rarely when the clinical history is ambiguous and documentation of periodic leg movements is necessary. | American Academy of Sleep Medicine |
| Avoid use of hypnotics as primary therapy for chronic insomnia in adults; instead offer cognitive behavior therapy, and reserve medication for adjunctive treatment when necessary. | American Academy of Sleep Medicine |
Source: For more information on the Choosing Wisely Campaign, see https://www.choosingwisely.org. For supporting citations and to search Choosing Wisely recommendations relevant to primary care, see https://www.aafp.org/afp/recommendations/search.htm.
Sleep disorders are classified formally into seven categories by the International Classification of Sleep Disorders, 3rd ed.7 Sleep disorders manifest as problems associated with falling asleep, disturbances occurring during sleep, and excessive daytime sleepiness.7 The most common sleep conditions encountered by family physicians are discussed in this article. Insomnia, delayed sleep-wake phase disorder, and restless legs syndrome (RLS) are common examples of problems that occur while falling asleep. RLS also can interfere with sleep late into the night. Other problems during sleep include parasomnias, such as rapid eye movement (REM) sleep behavior disorder. Parasomnias are disruptive sleep-related disorders that include sleepwalking, nightmares, sleep-related eating disorder, and sleep paralysis. Sleep disorders causing excessive daytime sleepiness include obstructive sleep apnea (OSA) and narcolepsy. Table 1 summarizes common sleep disorders.
TABLE 1. Summary of Common Sleep Disorders

| Disorder | Symptoms and signs | Most effective treatment |
|---|---|---|
| Problems falling asleep | ||
| Delayed sleep-wake phase disorder | Delayed sleep onset and late wake-up time | Melatonin before bed and bright light therapy at awakening for adolescents |
| Insomnia | Difficulty initiating or maintaining sleep, daytime sleepiness with inability to nap, daytime impairment | Cognitive behavior therapy for insomnia, benzodiazepine receptor agonists |
| Behavior and movement disturbances during sleep | ||
| Rapid eye movement sleep behavior disorder | Motor activity during sleep, acting out of dreams, polysomnography showing increased muscle tone | Behavior modification, clonazepam (Klonopin), melatonin |
| Restless legs syndrome | Uncomfortable sensation in both legs, symptoms are worse in the evening, improve with movement such as walking or stretching | Nonpharmacologic treatments, alpha-2-delta ligands, dopamine agonists |
| Excessive daytime sleepiness | ||
| Narcolepsy | Excessive daytime sleepiness, cataplexy, hallucinations when falling asleep or awakening | Modafinil (Provigil), armodafinil (Nuvigil), stimulants, gamma hydroxybutyric acid (sodium oxybate [Xyrem]), pitolisant (Wakix), selective serotonin reuptake inhibitors (venlafaxine, fluoxetine [Prozac], clomipramine [Anafranil]) |
| Obstructive sleep apnea | Snoring, witnessed apneas, gasping or choking, excessive daytime sleepiness | Positive airway pressure therapy |
Diagnosis
Many sleep disorders can be diagnosed by history alone; however, overnight polysomnography (PSG) may help diagnose certain sleep disorders such as OSA.6 PSG monitors brain wave activity, eye movements, muscle activity, heart rate and rhythm, and respiration. Brain wave activity is used to assess sleep stages, sleep latency, and arousals. Eye movements are monitored to assess REM sleep, and muscle activity is monitored to assess for parasomnias and RLS. Vital signs are monitored to assess for apneas that would indicate a diagnosis of OSA. PSG is commonly performed in a sleep laboratory, but home sleep apnea tests are sometimes used for the diagnosis of sleep apnea in patients with less severe sleep problems.8 Table 2 lists the most common indications for PSG.6
TABLE 2. Common Indications for Polysomnography

| Diagnosis of a sleep disorder |
| Narcolepsy |
| Parasomnias |
| Periodic limb movement disorder |
| Rapid eye movement sleep behavior disorder |
| Sleep-related breathing disorders (e.g., obstructive, central sleep apnea) |
| Sleep-related seizure disorders |
| Evaluation of sleep-related symptoms |
| Sleep maintenance insomnia |
| Snoring |
| Unexplained daytime fatigue or sleepiness |
| Treatment of sleep-related breathing disorders (i.e., positive airway pressure titration) |
Adapted with permission from Ramar K, Olson EJ. Management of common sleep disorders. Am Fam Physician. 2013;88(4):232.
Problems Falling Asleep
INSOMNIA
Insomnia is the most common sleep disorder, with approximately 33% of the adult population experiencing symptoms and 6% to 10% meeting diagnostic criteria for insomnia disorder.9 Chronic insomnia is classified as the report of difficulty initiating sleep (less than 30 minutes for people without insomnia [i.e., sleep latency]), maintaining sleep, or waking up too early with daytime consequences that occur at least three times per week for at least three months.7 Daytime consequences can involve symptoms of fatigue; difficulty with memory and concentration; and disturbances in mood or irritability.9
Diagnosis is often determined by the patient’s history and should include an evaluation for medical or psychiatric conditions that could be contributing. Approximately 40% to 50% of individuals presenting with insomnia have a comorbid mental illness.10 If the diagnosis is not clear from the history, other tools such as a sleep diary or actigraphy may be helpful.11 Actigraphy is a procedure where the patient continuously wears a relatively unobtrusive device on the wrist or ankle for days to weeks. The device measures the occurrence and degree of limb movements over time to provide information on sleep phases, sleep duration, daily patterns, and efficiency of sleep.11 PSG is not recommended for the diagnosis of chronic insomnia except to evaluate concurrent sleep disorders or look for other causes of the sleep disturbance when treatment has been ineffective.10
Treatments for insomnia include cognitive behavior therapy (CBT) for insomnia and hypnotic medications. Table 3 provides a summary of the treatment.1,10,12–14 CBT for insomnia is recommended as first-line therapy in adults with chronic insomnia.10,12,13,15 CBT for insomnia is preferred to hypnotic medications because it has better long-term effectiveness, more improvement in daytime symptoms, and fewer adverse effects.10,13 CBT for insomnia can be delivered in weekly sessions for six to eight weeks.16 A primary care physician can effectively provide CBT for insomnia or focus on individual components (although less effective) to treat chronic insomnia.10,14,17 Web-based CBT for insomnia is equally effective as in-person or group therapy and requires fewer resources.10,15,18 Sleep hygiene education alone provides minimal improvements in patients and is significantly less effective as monotherapy than as part of another therapy (e.g., CBT for insomnia).19
TABLE 3. Summary of Cognitive Behavior Therapy for Insomnia

| Cognitive behavior therapy |
| Focuses on changing false beliefs and attitudes about sleep (e.g., everyone needs at least 8 hours of sleep for good health) |
| Sleep hygiene education |
| Avoid alcohol for 4 to 6 hours before bed |
| Avoid caffeine or nicotine after 4 p.m. |
| Avoid exercise close to bedtime |
| Go to bed and wake up at the same time every day |
| Keep bedroom cool and conducive to sleep |
| No clock watching |
| No napping during the day |
| No watching television or other electronic devices in bed |
| Sleep restriction |
| After sleep efficiency (ratio of time sleeping to time in bed) reaches 90%, the time in bed can be increased by 15 minutes every week |
| Time in bed can be decreased by estimating the actual total time that the patient is sleeping (e.g., if the patient is in bed for 8 hours but sleeps for 5.5 hours, time in bed could be decreased to 5.5 hours); time in bed usually should not be decreased to less than 5 hours |
| Stimulus control |
| Go to another room if unable to fall asleep within 15 to 20 minutes |
| Go to bed only when sleepy |
| Read or engage in other quiet activities and return to bed only when sleepy |
| Use the bedroom only for sleep and sex |
A shared decision-making approach should be used to decide if pharmacologic therapy should be initiated when CBT for insomnia is ineffective or when there is incomplete symptom resolution.12,15 Medications approved by the U.S. Food and Drug Administration for the treatment of insomnia are summarized in Table 4.6,20 Medication choice should include the patient’s predominant sleep problem and the risk of potential adverse effects.12,15,21 Although antihistamines such as diphenhydramine (Benadryl) are used, the American Academy of Sleep Medicine does not recommend them for the treatment of insomnia because the benefits of use are approximately equal to the potential harms (e.g., drowsiness, dizziness).21 Melatonin is not recommended for treating insomnia because there are no consistent benefits, and multiple formulations are found in over-the-counter preparations. The American Academy of Sleep Medicine recommends against using trazodone because the harms outweigh the benefits.21 The strength of evidence supporting approved pharmacologic interventions is weak.21 Benzodiazepine receptor agonists (e.g., zolpidem [Ambien]) can be useful for treating sleep onset and sleep maintenance12,21; however, they should be used for short periods or only intermittently to avoid potential dependence. Orexin-receptor agonists (e.g., suvorexant [Belsomra], lemborexant [DayVigo]) may have some benefit in the treatment of chronic insomnia in adults.12,15,21
TABLE 4. Medications for the Treatment of Insomnia
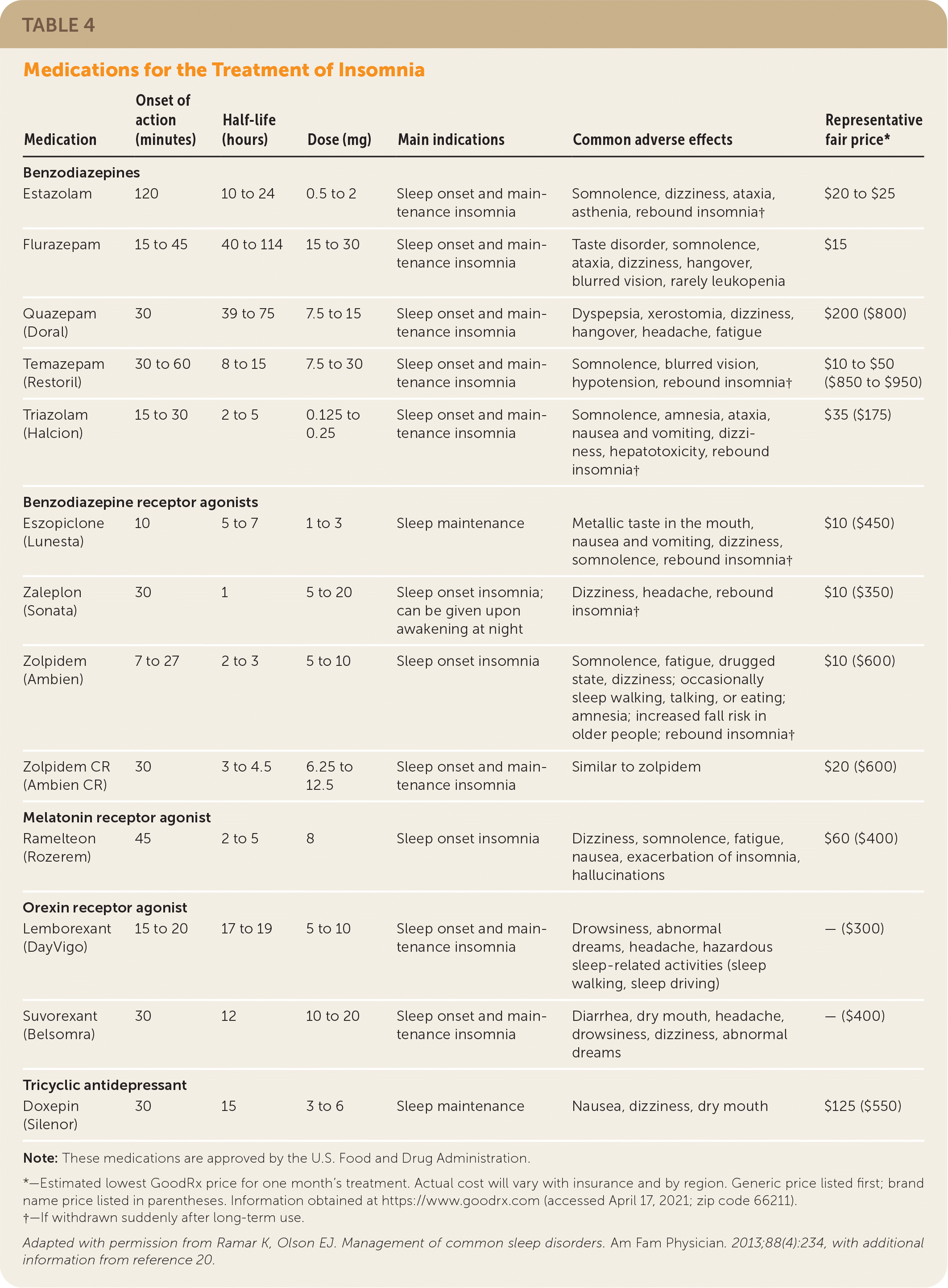
| Medication | Onset of action (minutes) | Half-life (hours) | Dose (mg) | Main indications | Common adverse effects | Representative fair price* |
|---|---|---|---|---|---|---|
| Benzodiazepines | ||||||
| Estazolam | 120 | 10 to 24 | 0.5 to 2 | Sleep onset and maintenance insomnia | Somnolence, dizziness, ataxia, asthenia, rebound insomnia† | $20 to $25 |
| Flurazepam | 15 to 45 | 40 to 114 | 15 to 30 | Sleep onset and maintenance insomnia | Taste disorder, somnolence, ataxia, dizziness, hangover, blurred vision, rarely leukopenia | $15 |
| Quazepam (Doral) | 30 | 39 to 75 | 7.5 to 15 | Sleep onset and maintenance insomnia | Dyspepsia, xerostomia, dizziness, hangover, headache, fatigue | $200 ($800) |
| Temazepam (Restoril) | 30 to 60 | 8 to 15 | 7.5 to 30 | Sleep onset and maintenance insomnia | Somnolence, blurred vision, hypotension, rebound insomnia† | $10 to $50 ($850 to $950) |
| Triazolam (Halcion) | 15 to 30 | 2 to 5 | 0.125 to 0.25 | Sleep onset and maintenance insomnia | Somnolence, amnesia, ataxia, nausea and vomiting, dizziness, hepatotoxicity, rebound insomnia† | $35 ($175) |
| Benzodiazepine receptor agonists | ||||||
| Eszopiclone (Lunesta) | 10 | 5 to 7 | 1 to 3 | Sleep maintenance | Metallic taste in the mouth, nausea and vomiting, dizziness, somnolence, rebound insomnia† | $10 ($450) |
| Zaleplon (Sonata) | 30 | 1 | 5 to 20 | Sleep onset insomnia; can be given upon awakening at night | Dizziness, headache, rebound insomnia† | $10 ($350) |
| Zolpidem (Ambien) | 7 to 27 | 2 to 3 | 5 to 10 | Sleep onset insomnia | Somnolence, fatigue, drugged state, dizziness; occasionally sleep walking, talking, or eating; amnesia; increased fall risk in older people; rebound insomnia† | $10 ($600) |
| Zolpidem CR (Ambien CR) | 30 | 3 to 4.5 | 6.25 to 12.5 | Sleep onset and maintenance insomnia | Similar to zolpidem | $20 ($600) |
| Melatonin receptor agonist | ||||||
| Ramelteon (Rozerem) | 45 | 2 to 5 | 8 | Sleep onset insomnia | Dizziness, somnolence, fatigue, nausea, exacerbation of insomnia, hallucinations | $60 ($400) |
| Orexin receptor agonist | ||||||
| Lemborexant (DayVigo) | 15 to 20 | 17 to 19 | 5 to 10 | Sleep onset and maintenance insomnia | Drowsiness, abnormal dreams, headache, hazardous sleep-related activities (sleep walking, sleep driving) | — ($300) |
| Suvorexant (Belsomra) | 30 | 12 | 10 to 20 | Sleep onset and maintenance insomnia | Diarrhea, dry mouth, headache, drowsiness, dizziness, abnormal dreams | — ($400) |
| Tricyclic antidepressant | ||||||
| Doxepin (Silenor) | 30 | 15 | 3 to 6 | Sleep maintenance | Nausea, dizziness, dry mouth | $125 ($550) |
Note: These medications are approved by the U.S. Food and Drug Administration.
*—Estimated lowest GoodRx price for one month’s treatment. Actual cost will vary with insurance and by region. Generic price listed first; brand name price listed in parentheses. Information obtained at https://www.goodrx.com (accessed April 17, 2021; zip code 66211).
†—If withdrawn suddenly after long-term use.
Adapted with permission from Ramar K, Olson EJ. Management of common sleep disorders. Am Fam Physician. 2013;88(4):234, with additional information from reference 20.
DELAYED SLEEP-WAKE PHASE DISORDER
Delayed sleep-wake phase disorder is a circadian rhythm disorder characterized by delayed sleep onset and a late wake-up time that results in excessive daytime sleepiness, difficulty falling asleep, and functional impairments that occur for at least three months.7 Delayed sleep-wake phase disorder may be genetic or socially reinforced. It most commonly occurs in adolescents and young adults.7,9 If a diagnosis is not evident from the patient’s history, a sleep diary and actigraphy can confirm.
Treatment of delayed sleep-wake phase disorder that includes melatonin (0.3 mg to 5 mg) administered before the desired bedtime may be effective in the setting of circadian misalignment; however, there are concerns about differences in product formulation.22 Melatonin should be combined with behavior changes in sleep-wake scheduling.23 Bright-light therapy at awakening is only recommended for children and adolescents, but it has shown improvement in shifting the circadian rhythm in adults.21,24
Behavior and Movement Disturbances During Sleep
REM SLEEP BEHAVIOR DISORDER
REM sleep behavior disorder is characterized by complex motor behaviors during REM sleep due to a loss of REM sleep atonia. This loss of atonia may result in the acting out of dreams and can involve harmful activities for the patient and bed partner, although this does not occur every night. Loss of atonia is additionally associated with neurologic and psychiatric conditions and most frequently presents between 40 and 60 years of age, with males being more commonly affected. REM sleep behavior disorder is linked to antidepressant use and may be an early indicator of Parkinson disease, Lewy body dementia, and multiple system atrophy because more than 90% of patients diagnosed with idiopathic REM sleep behavior disorder are eventually diagnosed with one of these disorders.25 If REM sleep behavior disorder is suspected based on the initial assessment and a baseline neurologic evaluation, a referral should be made to a sleep medicine or neurology specialist. PSG and further testing, when indicated, should be obtained to establish the diagnosis.25
Treatment of REM sleep behavior disorder focuses on reducing the injury potential for the patient and bed partner using behavior modification as a first-line intervention. Methods to decrease the potential for injury include creating a safe sleep environment by padding or removing furniture, lowering the mattress to the floor, and using a bed alarm.25 If these methods are not successful, the use of pharmacologic therapy with melatonin or clonazepam (Klonopin) may be necessary. Melatonin should be initiated at a dose of 3 mg at bedtime and titrated up by 3 mg every two weeks to the desired effect with a ceiling of 12 mg nightly.26 There are more adverse events associated with benzodiazepine use; however, if used, clonazepam can be administered at doses between 0.25 mg and 2.0 mg at bedtime.26
RESTLESS LEGS SYNDROME
RLS and periodic limb movement disorder are movement disorders, with RLS typically occurring in a wakeful state at rest and periodic limb movement disorder occurring less commonly and during sleep.27,28 RLS is characterized by an uneasy feeling and the urge to move the legs, which are described by the diagnostic mnemonic URGED (Table 5).29 Symptoms often occur at bedtime and may be associated with repetitive leg movements in sleep.27 Approximately 5% to 10% of adults are affected by RLS, with 2% to 3% seeking treatment.30,31 RLS is poorly understood at a biochemical level; however, there appears to be an interplay between iron, dopamine, and certain genetic markers.32
A diagnosis of RLS is based on meeting the five self-reported diagnostic criteria listed in Table 5.29 Once RLS is diagnosed, an evaluation of a patient’s iron status (specifically the ferritin level) should be conducted. Correcting an iron deficiency may yield symptomatic relief.27 The patient’s medications should be reviewed for any that may exacerbate symptoms. These include dopamine antagonists, antipsychotics, serotonergic antidepressants, and centrally acting antihistamines.33
TABLE 5. Diagnostic Criteria for Restless Legs Syndrome (URGED Mnemonic)

| Urge to move the legs that is usually accompanied by, or felt to be caused by, an uncomfortable sensation in the legs |
| Resting, sitting, or lying down worsens or initiates an uncomfortable sensation in the legs, resulting in an urge to move the legs |
| Getting up or moving the legs relieves all or part of the urge to move the legs and the accompanying uncomfortable sensation |
| Evenings and nighttime are associated with worsening of the urge to move the legs and accompanying uncomfortable sensation |
| Differential diagnosis does not adequately explain symptoms |
Note: All criteria must be met.
Adapted with permission from Winkelman JW, Bottary RM. Restless legs syndrome: clinical implications for psychiatrists. Psychiatr Times. 2017;34(7). July 20, 2017. Accessed February 12, 2021. https://www.psychiatrictimes.com/view/restless-legs-syndrome-clinical-implications-psychiatrists
A greater emphasis is placed on nonpharmacologic interventions to decrease symptom frequency, such as adjusting medications, counseling on daily exercise, and addressing caffeine and alcohol intake that may worsen symptoms.33 If these measures are not effective and sleep is disturbed, alpha-2-delta ligands (i.e., gabapentin [Neurontin], pregabalin [Lyrica]) should be used as the first-line treatment.31 Typical effective dosages of gabapentin can be as high as 1,200 mg to 1,800 mg per day, and pregabalin dosages range from 150 mg to 450 mg per day. If there is insufficient effect from alpha-2-delta ligands, dopamine agonists (e.g., ropinirole [Requip], pramipexole [Mirapex], rotigotine [Neupro]) can be considered with the caveat that these drugs may lose effectiveness or worsen the symptoms of RLS over time.31 In refractory RLS, benzodiazepines and opioids have been used with good effect.34
The diagnosis of periodic limb movement disorder must be confirmed by PSG. If movements appear to cause sleep disturbances resulting in daytime symptoms, the disorder is often managed with anticonvulsants, opioids, benzodiazepines, and dopamine agonists; however, there is no evidence to support the use of these therapies.27
Excessive Daytime Sleepiness
NARCOLEPSY
Narcolepsy affects less than 0.04% of the general population.9 Symptoms often begin to manifest between 10 and 20 years of age.35 The classic features of narcolepsy include excessive daytime sleepiness, cataplexy, sleep paralysis, and sleep hallucinations.9,36 Narcolepsy is divided into types 1 and 2. The major clinical difference between the types is that patients with type 2 do not experience cataplexy.35 Cataplexy is a sudden episode of complete or partial paralysis of voluntary muscles that is often triggered by strong positive and sometimes negative emotions. These episodes normally last from several seconds to one to two minutes.35,36 Sleep hallucinations associated with narcolepsy are characterized as hypnagogic (hallucinations upon falling asleep) and hypnopompic (hallucinations upon awakening).
A diagnosis can be apparent from the clinical history, but confirmation should be obtained through PSG followed by a multiple sleep latency test the next day. The multiple sleep latency test consists of having the patient nap every two hours for 20 minutes over the course of a day while being monitored. People with narcolepsy generally fall asleep within eight minutes (compared with 15 minutes for the general population) and have REM sleep during at least two of the daytime naps (unaffected people usually experience REM sleep only at night).7
Treatment consists of behavior approaches and medications for the symptoms of daytime sleepiness and cataplexy.35,36 Scheduled naps, consistent sleep schedules with good sleep hygiene, and strategic use of caffeine are behavior approaches that have been shown to improve daytime sleepiness.36 Modafinil (Provigil), armodafinil (Nuvigil), or stimulants such as methylphenidate (Ritalin), dextroamphetamine, or amphetamine-dextroamphetamine can treat excessive daytime sleepiness when behavior modifications alone are not adequate. Cataplexy is treated with medications including antidepressants, such as venlafaxine, fluoxetine (Prozac), and clomipramine (Anafranil). There are two medications to treat cataplexy and daytime sleepiness: gamma hydroxybutyrate acid (sodium oxybate [Xyrem]) and pitolisant (Wakix).37 Pitolisant was found to be as effective as sodium oxybate and had fewer adverse effects.37,38
OBSTRUCTIVE SLEEP APNEA
OSA is characterized by multiple episodes of hypopnea (slow or shallow breathing) or apnea (pauses in breathing) throughout the night due to upper airway obstruction, often accompanied by snoring. These episodes lead to excessive daytime sleepiness, hypertension, dry mouth, and morning headaches.39 The incidence of OSA has continued to increase in the United States with nearly 18 million estimated patients affected.40 It affects males more than females and is strongly associated with such downstream effects as hypertension, atrial fibrillation, heart failure, stroke, type 2 diabetes mellitus, and Alzheimer disease.39 A recent meta-analysis found that patients with severe OSA have a twofold increase of sudden death and cardiovascular mortality.41
The U.S. Preventive Services Task Force does not recommend for or against screening asymptomatic adults for OSA due to a lack of evidence; however, this recommendation is being updated because of newer evidence for treatment benefits.42 Screening for OSA can be done during preventive care visits by including sleep as a part of a comprehensive review of systems and when assessing for predisposing risk factors. There are several commonly used OSA screening tools, with the STOP-Bang Questionnaire (https://www.mdcalc.com/stop-bang-score-obstructive-sleep-apnea) and Sleep Apnea Clinical Score demonstrating the best utility in predicting diagnosis. The STOP-Bang Questionnaire yields a numerical result indicating a level of risk.43 Based on this level of risk, patients should be evaluated with home sleep apnea testing or PSG.44 Home sleep apnea testing is appropriate for patients at high risk of OSA with no other known comorbidities or for patients who cannot receive PSG.8
Long-term positive airway pressure therapy should be considered for all patients with OSA. Types of positive airway pressure include continuous, bilevel, autotitrating, and adaptive servo ventilation, with continuous being the most studied and used. Continuous positive airway pressure decreases the development of diabetes and cardiovascular disease and overall mortality with good aherence.45 Continuous positive airway pressure has also been shown to provide symptom relief, with benefits on quality of life and mood.46,47 A recent meta-analysis demonstrated that long-term positive airway pressure therapy use in moderate to severe OSA decreases all-cause mortality.45
For mild to moderate cases of OSA, therapy can be initiated with nighttime use of positive airway pressure or mandibular advancement devices.48 Patients with severe OSA, should receive positive airway pressure as a first-line therapy.48 Follow-up care after initiation of positive airway pressure should address adverse effects with the goal of ensuring long-term adherence. Symptom resolution should be evaluated because persistent symptoms despite positive airway pressure warrant an in-laboratory sleep evaluation. In mild OSA or if the patient is intolerant to positive airway pressure, patients can be offered a mandibular advancement device, custom fitted by a dentist, or positional changes as an alternative therapy option.48
Nearly 70% of patients with OSA are obese, and weight loss continues to demonstrate significant improvement in the severity and symptoms of OSA49; therefore, weight loss is a mainstay of therapy in conjunction with positive airway pressure.50
This article updates a previous article on this topic by Ramar and Olson.6
Data Sources: A PubMed search was completed in Clinical Queries using the key terms sleep disorders, approach, and management. The search included meta-analyses, randomized controlled trials, clinical trials, guidelines, and reviews. Also searched were the Agency for Healthcare Research and Quality evidence reports, Clinical Evidence, the Cochrane database, Essential Evidence Plus, the Institute for Clinical Systems Improvement, and the National Guideline Clearinghouse database. Search dates: February to April 2021, and December 2021.

