Acute pancreatitis is the most common gastrointestinal-related reason for hospitalization in the United States. It is diagnosed based on the revised Atlanta classification, with the presence of at least two of three criteria (upper abdominal pain, serum amylase or lipase level greater than three times the upper limit of normal, or characteristic findings on imaging studies). Although computed tomography and other imaging studies can be useful to assess severity or if the diagnosis is uncertain, imaging is not required to diagnose acute pancreatitis. Based on limited studies, several scoring systems have comparable effectiveness for predicting disease severity. The presence of systemic inflammatory response syndrome on day 1 of hospital admission is highly sensitive in predicting severe disease. Treatment of acute pancreatitis involves goal-directed fluid resuscitation, analgesics, and oral feedings as tolerated on admission. If oral feedings are not tolerated, nasogastric or nasojejunal feedings are preferred over parenteral nutrition. Cholecystectomy is recommended during the initial admission for patients with mild acute biliary pancreatitis. Medical management is usually sufficient for necrotizing pancreatitis; however, if surgical intervention is needed, a minimally invasive approach is advised over direct endoscopic or open surgical debridement (necrosectomy) because of lower complication rates.
Acute pancreatitis is the most common gastrointestinal condition requiring hospital admission in the United States. This article summarizes the best available patient-oriented evidence for the diagnosis and treatment of acute pancreatitis.
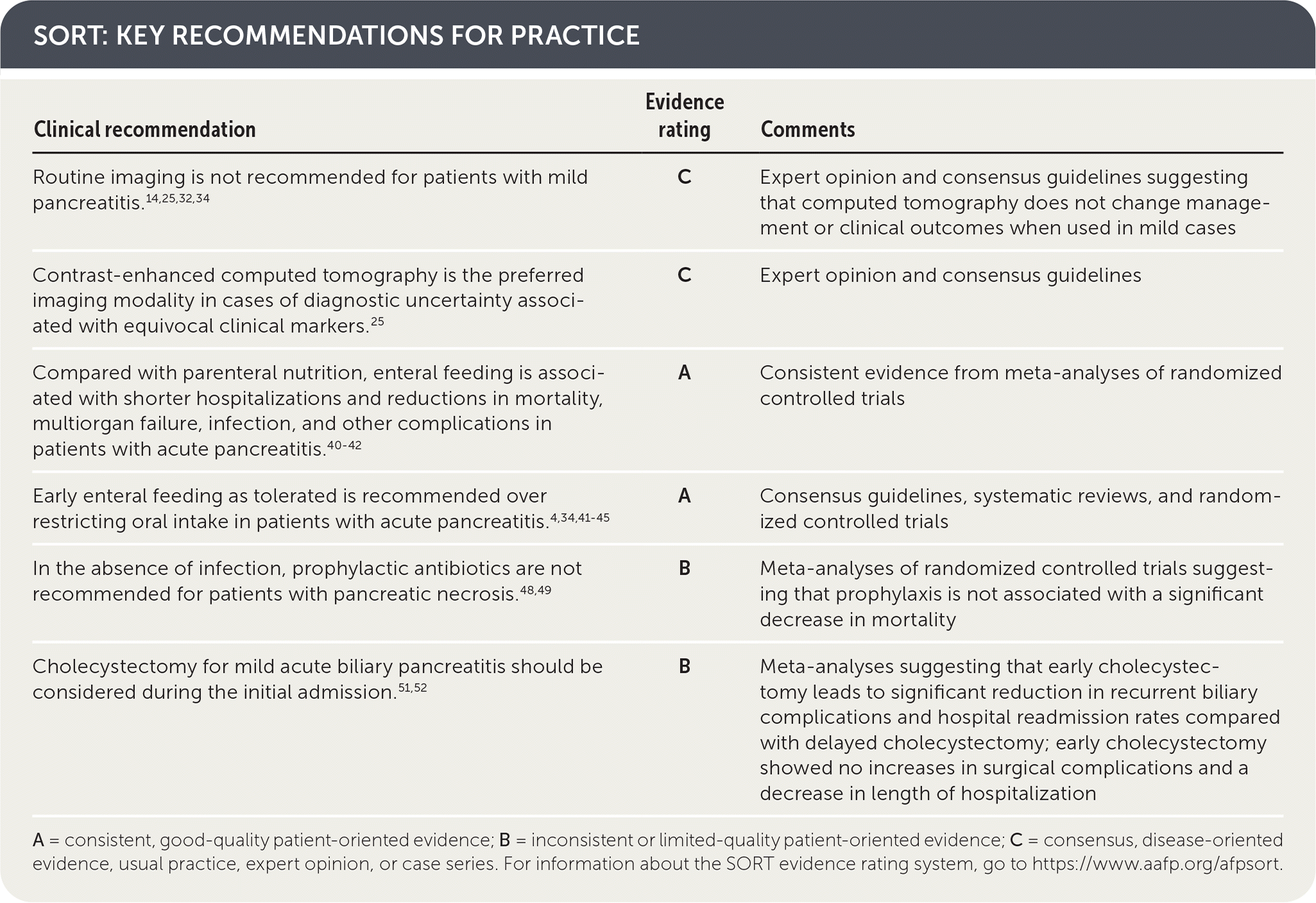
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | Comments |
|---|---|---|
| Routine imaging is not recommended for patients with mild pancreatitis.14,25,32,34 | C | Expert opinion and consensus guidelines suggesting that computed tomography does not change management or clinical outcomes when used in mild cases |
| Contrast-enhanced computed tomography is the preferred imaging modality in cases of diagnostic uncertainty associated with equivocal clinical markers.25 | C | Expert opinion and consensus guidelines |
| Compared with parenteral nutrition, enteral feeding is associated with shorter hospitalizations and reductions in mortality, multiorgan failure, infection, and other complications in patients with acute pancreatitis.40–42 | A | Consistent evidence from meta-analyses of randomized controlled trials |
| Early enteral feeding as tolerated is recommended over restricting oral intake in patients with acute pancreatitis.4,34,41–45 | A | Consensus guidelines, systematic reviews, and randomized controlled trials |
| In the absence of infection, prophylactic antibiotics are not recommended for patients with pancreatic necrosis.48,49 | B | Meta-analyses of randomized controlled trials suggesting that prophylaxis is not associated with a significant decrease in mortality |
| Cholecystectomy for mild acute biliary pancreatitis should be considered during the initial admission.51,52 | B | Meta-analyses suggesting that early cholecystectomy leads to significant reduction in recurrent biliary complications and hospital readmission rates compared with delayed cholecystectomy; early cholecystectomy showed no increases in surgical complications and a decrease in length of hospitalization |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
BEST PRACTICES IN GASTROENTEROLOGY

| Recommendation | Sponsoring organization |
|---|---|
| Do not test for amylase in cases of suspected acute pancreatitis. Instead, test for lipase. | American Society for Clinical Pathology |
Source: For more information on the Choosing Wisely Campaign, see https://www.choosingwisely.org. For supporting citations and to search Choosing Wisely recommendations relevant to primary care, see https://www.aafp.org/afp/recommendations/search.htm.
Epidemiology
- The incidence of acute pancreatitis is approximately 5 to 80 cases per 100,000 people per year.1–4
- Between the years 2001 and 2014, hospitalizations and costs related to acute pancreatitis steadily increased, likely because of the increased prevalence of obesity, an aging population, and gallstone-related disorders.1–5
- Table 1 lists causes of acute pancreatitis.6–13
TABLE 1. Causes of Acute Pancreatitis

| More common | Uncommon |
|---|---|
| Choledocholithiasis (38% to 70%) Chronic alcohol use (25% to 41%) Hypertriglyceridemia (10%) Endoscopic retrograde cholangiopan-creatography (4%) Pancreatic ductal carcinoma (1% to 4%) Medication use (< 2%): aminosalicylates, anticonvulsants, antimicrobials, hormone therapy, oral contraceptives, loop diuretics, nonsteroidal anti-inflammatory drugs, opiates, reverse transcriptase inhibitors, steroids, glucagon-like peptide 1 antagonists | Abnormalities of the pancreas: annular pancreas, pancreas divisum, sphincter of Oddi dysfunction Autoimmune disorders Genetic factors:: variation in CFTR, CTRC, PRSS1, SPINK1, or CASR gene Hypercalcemia: excessive vitamin D supplementation, hyperparathyroidism, total parenteral nutrition Infections: viral, bacterial, fungal, parasitic Surgical procedures and trauma Toxins: scorpion and snake bites Vascular abnormalities: ischemia, vasculitis |
Adapted with permission from Quinlan JD. Acute pancreatitis. Am Fam Physician. 2014;90(9): 633, with additional information from references 7–13.
Diagnosis
- Acute pancreatitis is diagnosed based on the revised Atlanta classification, which includes the presence of at least two of the following criteria: (1) upper abdominal pain, (2) serum amylase or lipase level more than three times the upper limit of normal, or (3) characteristic imaging findings.14
- The differential diagnosis of acute pancreatitis is shown in Table 2.
TABLE 2. Differential Diagnosis of Acute Pancreatitis
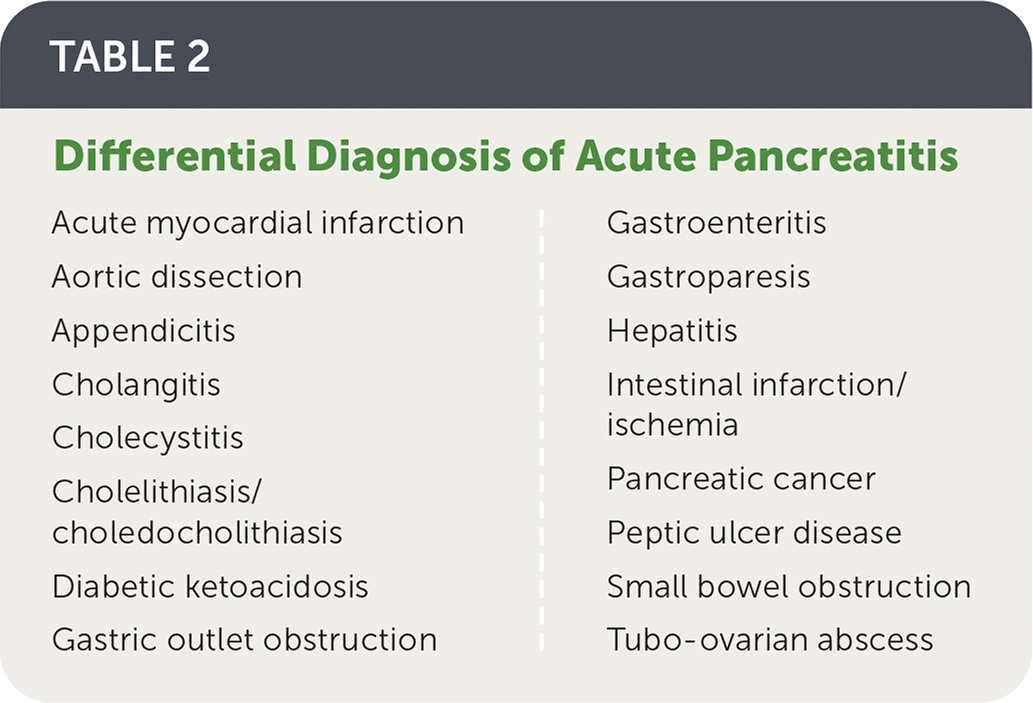
| Acute myocardial infarction Aortic dissection Appendicitis Cholangitis Cholecystitis Cholelithiasis/choledocholithiasis Diabetic ketoacidosis Gastric outlet obstruction | Gastroenteritis Gastroparesis Hepatitis Intestinal infarction/ischemia Pancreatic cancer Peptic ulcer disease Small bowel obstruction Tubo-ovarian abscess |
SIGNS AND SYMPTOMS
- Acute pancreatitis should be suspected in patients with sudden onset of left upper quadrant or epigastric abdominal pain, although it is sometimes painless. Pain often radiates to the back, worsens with eating or drinking, and is associated with nausea and vomiting.10
- Physical examination findings may include tenderness on palpation of the upper abdomen, decreased bowel sounds, fever, tachycardia, hypotension, or jaundice.10
- Overall, the history and physical examination have moderate accuracy when findings are abnormal (positive likelihood ratio = 3.2; negative likelihood ratio = 0.8).10
- Ecchymosis around the umbilicus (Cullen sign) or on the flank (Grey Turner sign) is present in only 3% of patients with acute pancreatitis and may occur with other diseases. Therefore, the diagnostic significance of these findings is low.15,16
DIAGNOSTIC TESTING
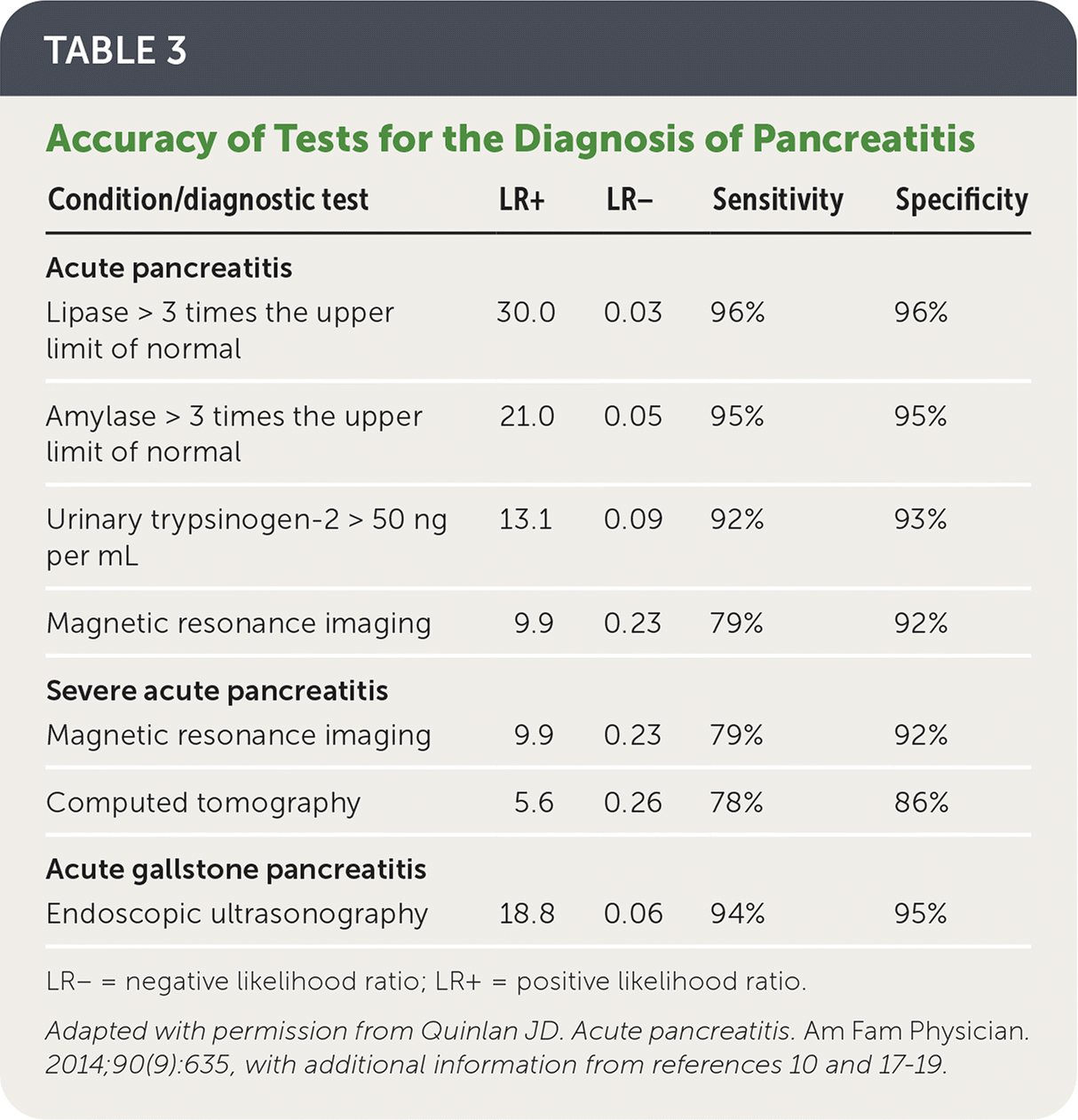
- The accuracy of laboratory and imaging tests in the diagnosis of acute pancreatitis is summarized in Table 3.6,10,17–19
- Serum lipase measurement should be the initial test in suspected acute pancreatitis, although serial measurements are not indicated. Amylase and lipase levels increase rapidly in pancreatitis, but serum amylase normalizes more quickly. Elevation of amylase or lipase that is more than three times the upper limit of normal strongly suggests acute pancreatitis.18,20–22
- A urinary trypsinogen-2 level of more than 50 ng per mL on dipstick testing also suggests pancreatitis, but this test is not as widely available as serum amylase testing.18
- Laboratory evaluation should include a comprehensive metabolic panel, complete blood count, and triglyceride levels to aid in diagnosis and severity assessment. An aspartate transaminase level of more than 60 U per L (1.00 μkat per L) and an alkaline phosphatase level of more than 12 U per L (0.20 μkat per L) are helpful in the diagnosis of acute biliary pancreatitis, with positive predictive values of 90.2 and 81.9, respectively.10,23
- Imaging is not required for the diagnosis of acute pancreatitis, although computed tomography can be useful to assess severity or in cases of diagnostic uncertainty, failure of conservative treatment, or clinical deterioration.14,19,24
- Ultrasonography is useful in patients with suspected acute biliary pancreatitis; however, sensitivity for detection of gallstones is only 67% to 78%. Sensitivity of ultrasonography for choledocholithiasis is between 50% and 80%. Endoscopic ultrasonography is more accurate for choledocholithiasis, with a sensitivity of more than 90%, but it is more invasive.25–27
- Magnetic resonance cholangiopancreatography is recommended when there is concern for acute biliary pancreatitis and the common bile duct is not adequately visualized or appears normal on ultrasonography. Magnetic resonance cholangiopancreatography has a similar sensitivity to endoscopic ultrasonography for detection of common bile duct stones that are 6 mm or larger.25,28,29
TABLE 3. Accuracy of Tests for the Diagnosis of Pancreatitis
| Condition/diagnostic test | LR+ | LR− | Sensitivity | Specificity |
|---|---|---|---|---|
| Acute pancreatitis | ||||
| Lipase > 3 times the upper limit of normal | 30.0 | 0.03 | 96% | 96% |
| Amylase > 3 times the upper limit of normal | 21.0 | 0.05 | 95% | 95% |
| Urinary trypsinogen-2 > 50 ng per mL | 13.1 | 0.09 | 92% | 93% |
| Magnetic resonance imaging | 9.9 | 0.23 | 79% | 92% |
| Severe acute pancreatitis | ||||
| Magnetic resonance imaging | 9.9 | 0.23 | 79% | 92% |
| Computed tomography | 5.6 | 0.26 | 78% | 86% |
| Acute gallstone pancreatitis | ||||
| Endoscopic ultrasonography | 18.8 | 0.06 | 94% | 95% |
LR− = negative likelihood ratio; LR+ = positive likelihood ratio.
Adapted with permission from Quinlan JD. Acute pancreatitis. Am Fam Physician. 2014;90(9):635, with additional information from references 10 and 17–19.
ASSESSMENT OF SEVERITY
- The goal of risk stratification is to identify patients with acute pancreatitis who are at risk of severe disease.30–32
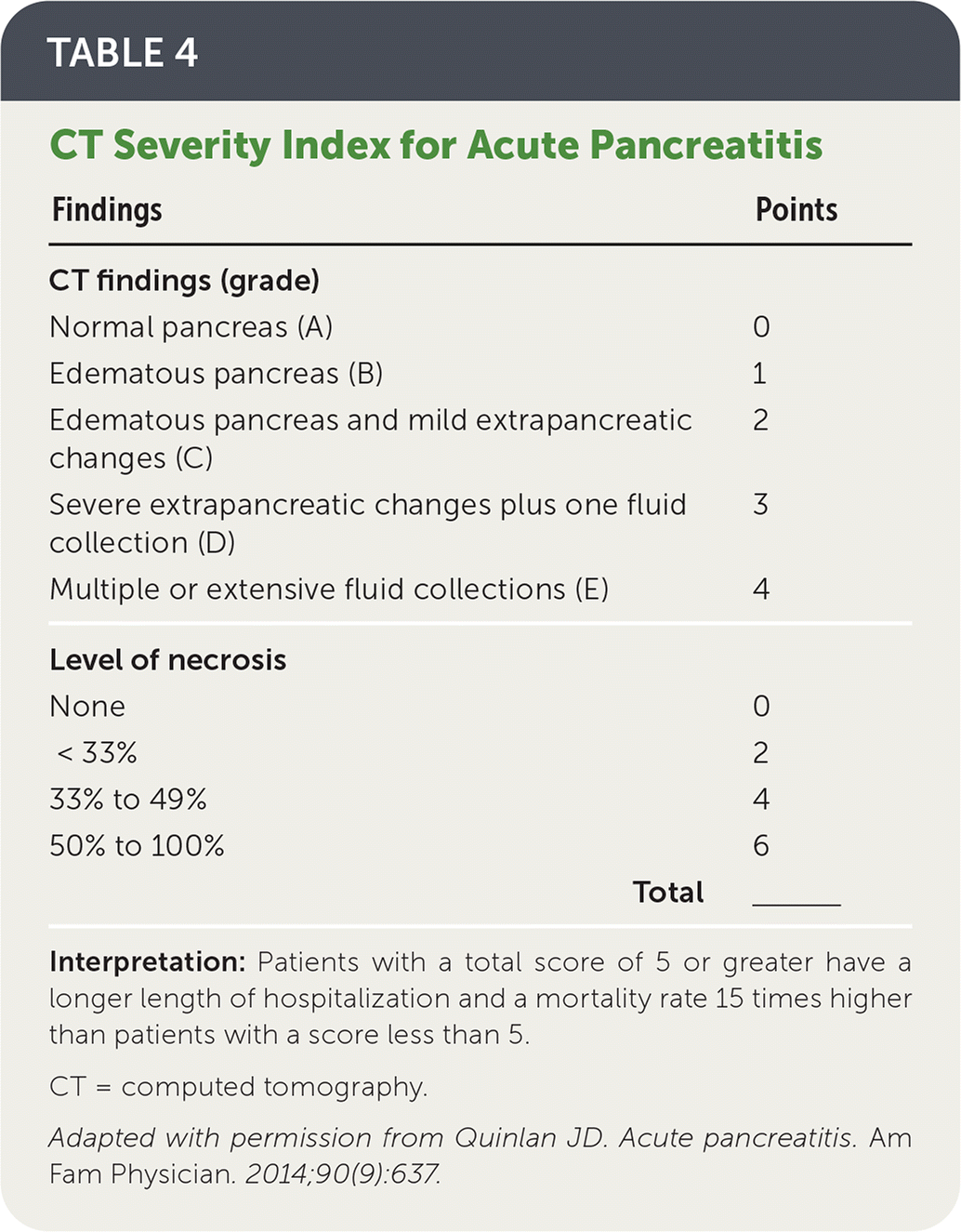
- Limited studies suggest that Ranson criteria for pancreatitis mortality (https://www.mdcalc.com/ransons-criteria-pancreatitis-mortality), the Acute Physiology and Chronic Health Evaluation II (https://www.mdcalc.com/apache-ii-score), the Bedside Index for Severity in Acute Pancreatitis (https://www.mdcalc.com/bisap-score-pancreatitis-mortality), and the Computed Tomography Severity Index (Table 46 ) have comparable accuracy in predicting disease severity.30–32
- A 2009 study found that the presence of systemic inflammatory response syndrome (SIRS) on day 1 of admission had high sensitivity for predicting severe disease (85% to 100%).33 Conversely, the absence of SIRS had a high negative predictive value of around 98% to 100% for severe disease.33
- Based on moderate evidence, the 2013 International Association of Pancreatology/American Pancreatic Association guideline supports using SIRS criteria (Table 533 ) to predict severity of acute pancreatitis within the first 48 hours of admission and to monitor response to therapy.34
TABLE 4. CT Severity Index for Acute Pancreatitis
| Findings | Points |
|---|---|
| CT findings (grade) | |
| Normal pancreas (A) | 0 |
| Edematous pancreas (B) | 1 |
| Edematous pancreas and mild extrapancreatic changes (C) | 2 |
| Severe extrapancreatic changes plus one fluid collection (D) | 3 |
| Multiple or extensive fluid collections (E) | 4 |
| Level of necrosis | |
| None | 0 |
| < 33% | 2 |
| 33% to 49% | 4 |
| 50% to 100% | 6 |
| Total | ________ |
Interpretation: Patients with a total score of 5 or greater have a longer length of hospitalization and a mortality rate 15 times higher than patients with a score less than 5.
CT = computed tomography.
Adapted with permission from Quinlan JD. Acute pancreatitis. Am Fam Physician. 2014;90(9):637.
TABLE 5. Criteria for Diagnosing Systemic Inflammatory Response Syndrome in Patients With Acute Pancreatitis

| Criteria are met if at least two of the following are present: |
| Pulse > 90 beats per minute |
| Respiratory rate > 20 breaths per minute or PaCO2 < 32 mm Hg |
| Temperature > 38°C (100.4°F) or < 36°C (96.8°F) |
| White blood cell count < 4,000 per μL (4 × 109 per L) or > 12,000 per μL (12 × 109 per L), or > 10% immature bands |
PaCO2 = partial pressure of carbon dioxide, arterial.
Adapted with permission from Singh VK, Wu BU, Bollen TL, et al. Early systemic inflammatory response syndrome is associated with severe acute pancreatitis. Clin Gastroenterol Hepatol. 2009;7(11): 1248.
Treatment
MEDICAL MANAGEMENT
- Medical management of acute pancreatitis includes fluid resuscitation, pain control, and early enteral feeding.
- Lactated Ringer solution should be considered over normal saline for initial fluid resuscitation in patients without hypercalcemia. A 2018 systematic review and meta-analysis of five studies with 428 total patients showed that those receiving lactated Ringer solution had significantly lower odds of SIRS at 24 hours compared with those receiving normal saline (pooled odds ratio [OR] = 0.38; 95% CI, 0.15 to 0.98; P = .05). A numerically lower odds of mortality was also observed with lactated Ringer solution compared with normal saline, but this was not statistically significant (OR = 0.61; 95% CI, 0.28 to 1.29; P = .20).24,34,35
- A 2020 meta-analysis of 11 studies with 2,626 total patients found no difference in mortality between more and less rapid fluid resuscitation strategies, but higher rates of kidney injury and respiratory failure were noted with more rapid strategies.36 Current guidelines suggest goal-directed fluid resuscitation with an initial rate of 5 to 10 mL per kg per hour. Goals include heart rate lower than 120 beats per minute, mean arterial pressure of 65 to 85 mm Hg, urinary output greater than 0.5 to 1.0 mL per kg per hour, and hematocrit of 35% to 44%.4,34
- Nonopioid analgesics (nonsteroidal anti-inflammatory drugs or acetaminophen) are reasonable to use initially in appropriate patients, with no increased risk of complications compared with opioid analgesics. If pain control is inadequate, enteral or parenteral opioids are recommended.37–39
- A 2010 Cochrane review and 2018 meta-analysis showed that enteral feeding was associated with reductions in mortality, multiorgan failure, infection, and other complications compared with parenteral nutrition. Enteral feeding was also associated with shorter hospitalization.40–42
- Early enteral feeding should be initiated as tolerated on admission and is recommended over restricting oral intake. If oral feeding is not tolerated, nasogastric or nasojejunal feeding should be attempted before parenteral nutrition. Outcomes are similar between nasogastric and nasojejunal feedings.4,34,41–46
- Prophylactic antibiotics are not recommended for patients with predicted severe pancreatitis or pancreatic necrosis.4,34,47–49
- Antibiotics are indicated for infected pancreatic necrosis diagnosed via imaging (emphysematous findings) or clinically (fever, bacteremia, clinical decline). Broad-spectrum antibiotics are recommended and may include carbapenems, quinolones, metronidazole (Flagyl), and third-generation or higher cephalosporins.34,47
- Probiotic therapy has been associated with increased mortality in patients with acute pancreatitis and should not be used.50
SURGICAL MANAGEMENT
- Cholecystectomy for mild acute biliary pancreatitis should be considered during the initial admission.51,52
- Endoscopic retrograde cholangiopancreatography with sphincterotomy is indicated within 24 hours in patients with acute pancreatitis associated with cholangitis or persistent biliary obstruction. In the absence of these conditions, prompt endoscopic retrograde cholangiopancreatography does not improve complication or mortality rates compared with conservative treatments.53,54
- Medical management is usually sufficient for necrotizing pancreatitis. Indications for procedural intervention may include infected necrotizing pancreatitis, persistent organ failure, ongoing intestinal obstruction, and persistent pain. If possible, intervention should be delayed until fluid collections become walled off, usually around four weeks.55
- Imaging-guided percutaneous drainage or endoscopic transluminal drainage should be considered before direct endoscopic or open surgical debridement (necrosectomy) because of decreased short- and long-term complications.45,55
Prognosis
- Possible complications of acute pancreatitis are summarized in Table 6.14,34,43,56–66
- Most episodes of acute pancreatitis resolve without complications. The mortality rate is approximately 5%.3
- Up to 20% of patients with acute pancreatitis develop pancreatic necrosis, which is associated with a 20% to 30% mortality rate if infected.47
- Complicated acute pancreatitis is best managed via a multidisciplinary approach, including gastroenterology, surgical, interventional radiology, critical care, and nutrition specialists.10
TABLE 6. Possible Complications of Acute Pancreatitis
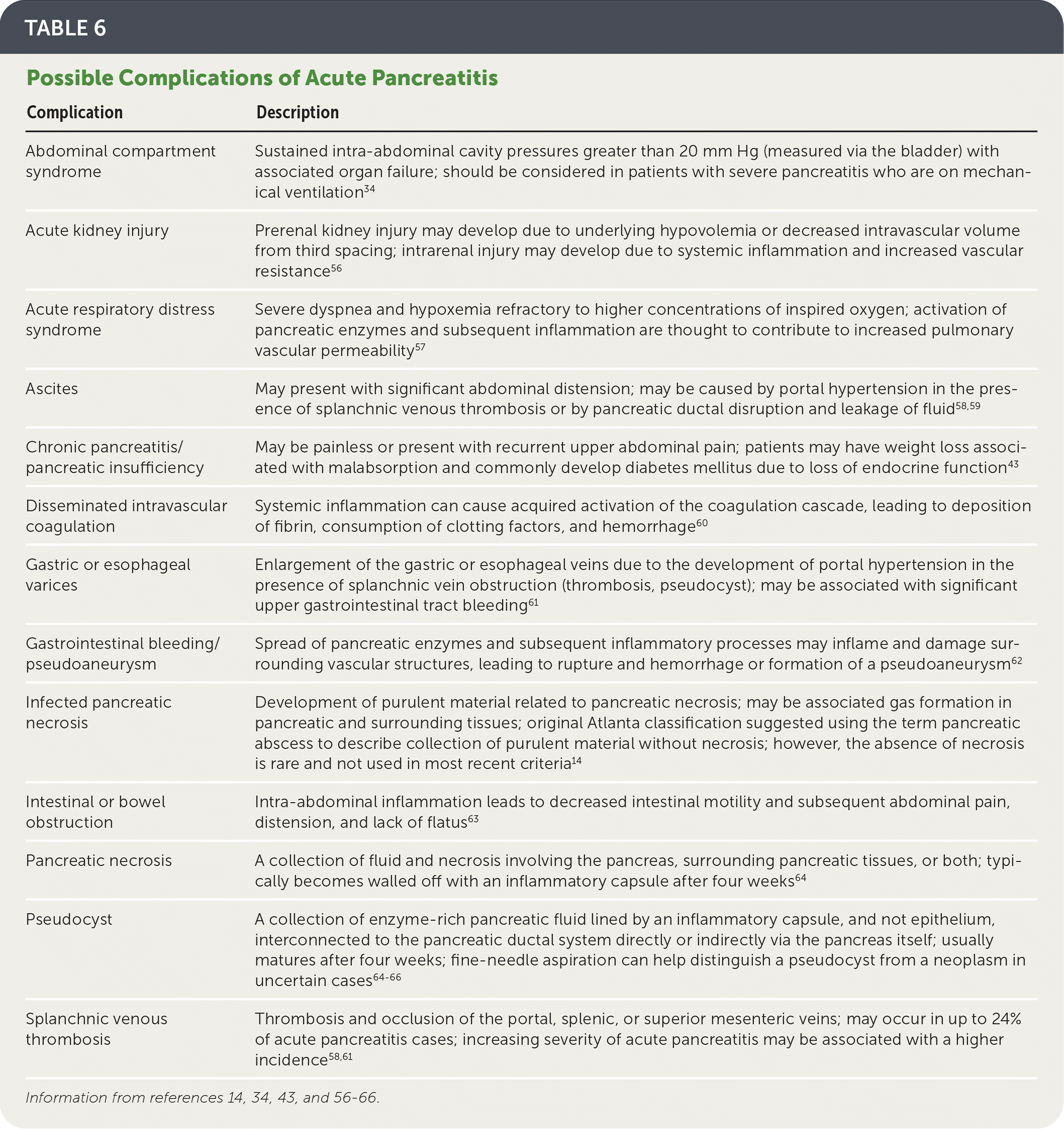
| Complication | Description |
|---|---|
| Abdominal compartment syndrome | Sustained intra-abdominal cavity pressures greater than 20 mm Hg (measured via the bladder) with associated organ failure; should be considered in patients with severe pancreatitis who are on mechanical ventilation34 |
| Acute kidney injury | Prerenal kidney injury may develop due to underlying hypovolemia or decreased intravascular volume from third spacing; intrarenal injury may develop due to systemic inflammation and increased vascular resistance56 |
| Acute respiratory distress syndrome | Severe dyspnea and hypoxemia refractory to higher concentrations of inspired oxygen; activation of pancreatic enzymes and subsequent inflammation are thought to contribute to increased pulmonary vascular permeability57 |
| Ascites | May present with significant abdominal distension; may be caused by portal hypertension in the presence of splanchnic venous thrombosis or by pancreatic ductal disruption and leakage of fluid58,59 |
| Chronic pancreatitis/pancreatic insufficiency | May be painless or present with recurrent upper abdominal pain; patients may have weight loss associated with malabsorption and commonly develop diabetes mellitus due to loss of endocrine function43 |
| Disseminated intravascular coagulation | Systemic inflammation can cause acquired activation of the coagulation cascade, leading to deposition of fibrin, consumption of clotting factors, and hemorrhage60 |
| Gastric or esophageal varices | Enlargement of the gastric or esophageal veins due to the development of portal hypertension in the presence of splanchnic vein obstruction (thrombosis, pseudocyst); may be associated with significant upper gastrointestinal tract bleeding61 |
| Gastrointestinal bleeding/pseudoaneurysm | Spread of pancreatic enzymes and subsequent inflammatory processes may inflame and damage surrounding vascular structures, leading to rupture and hemorrhage or formation of a pseudoaneurysm62 |
| Infected pancreatic necrosis | Development of purulent material related to pancreatic necrosis; may be associated gas formation in pancreatic and surrounding tissues; original Atlanta classification suggested using the term pancreatic abscess to describe collection of purulent material without necrosis; however, the absence of necrosis is rare and not used in most recent criteria14 |
| Intestinal or bowel obstruction | Intra-abdominal inflammation leads to decreased intestinal motility and subsequent abdominal pain, distension, and lack of flatus63 |
| Pancreatic necrosis | A collection of fluid and necrosis involving the pancreas, surrounding pancreatic tissues, or both; typically becomes walled off with an inflammatory capsule after four weeks64 |
| Pseudocyst | A collection of enzyme-rich pancreatic fluid lined by an inflammatory capsule, and not epithelium, interconnected to the pancreatic ductal system directly or indirectly via the pancreas itself; usually matures after four weeks; fine-needle aspiration can help distinguish a pseudocyst from a neoplasm in uncertain cases64–66 |
| Splanchnic venous thrombosis | Thrombosis and occlusion of the portal, splenic, or superior mesenteric veins; may occur in up to 24% of acute pancreatitis cases; increasing severity of acute pancreatitis may be associated with a higher incidence58,61 |
This article updates previous articles on this topic by Quinlan6; Carroll, et al.67; and Munoz and Katerndahl.68
Data Sources: The primary data sources used to identify literature were PubMed and the Cochrane database. Essential Evidence Plus was searched to identify further sources. Primary search terms included acute pancreatitis free text, and acute pancreatitis (MeSH) with one of the following: risk factors, causes, diagnosis, treatment, complications. Search dates: June 2021 to August 2021, and April 2022.
The opinions and assertions contained herein are the private views of the authors and are not to be construed as the official policy or position of the Army, Department of Defense, or U.S. government.

