Primary aldosteronism is the underlying cause of hypertension in primary care settings in approximately 6% of cases, and it is even more common in patients with resistant hypertension. However, it is estimated that only about 2% of patients who have risk factors for primary aldosteronism have been formally tested or diagnosed. The first step in the diagnosis of primary aldosteronism is case detection and involves testing patients who are at risk, including individuals with resistant hypertension, as well as those with well-controlled hypertension and a first-degree relative with primary aldosteronism, hypokalemia, an adrenal nodule, atrial fibrillation, obstructive sleep apnea, or a family history of an early stroke (i.e., younger than 40 years). Initial case detection is performed by simultaneously measuring plasma aldosterone concentration and plasma renin activity; an elevated aldosterone-renin ratio (greater than 30) indicates independent aldosterone secretion (i.e., aldosteronism). After a positive case detection, confirmatory testing should be performed. Confirmatory tests include the captopril challenge, oral or intravenous salt loading, or fludrocortisone suppression. Results are positive if aldosterone levels remain high after interventions that suppress or interrupt physiologic production of aldosterone. If the confirmatory test is positive, adrenal computed tomography and adrenal vein sampling should be performed to differentiate unilateral from bilateral adrenal production of aldosterone. Patients with unilateral primary aldosteronism should undergo adrenalectomy, whereas those with bilateral production should be treated with mineralocorticoid receptor antagonists, such as spironolactone or eplerenone.
Primary aldosteronism, first described by Dr. Jerome Conn in 1955, has long been considered a rare secondary cause of hypertension.1 Newer evidence, however, suggests that primary aldosteronism is common and largely under-recognized. It has been shown to be the underlying cause of hypertension in about 6% of patients in primary care settings2 and in more than 20% of patients with stage 2 hypertension.3 Even for patients with an indication for case detection, appropriate testing is performed in only 1.6% to 2.1% of cases.4,5 This under-recognition of primary aldosteronism can lead to delayed diagnosis and treatment, which can cause irreversible end-organ damage.6
SORT: KEY RECOMMENDATIONS FOR PRACTICE
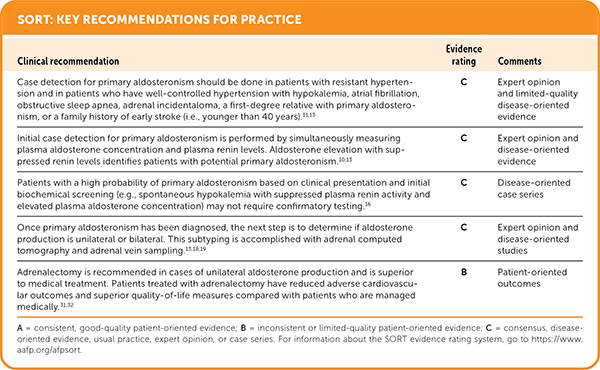
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Background
Primary aldosteronism is the overproduction and over-secretion of aldosterone, occurring independently of the renin-angiotensin-aldosterone system (RAAS). Aldosterone can cause sodium retention and potassium excretion in the renal tubules.
Aldosterone (mineralocorticoid) receptors are also present in vascular endothelial smooth muscle cells and the myocardium. Chronic activation of these receptors is associated with proinflammatory and profibrotic effects,7 likely explaining the increased risk of cerebrovascular, cardiovascular, and renal disease in primary aldosteronism compared with essential hypertension8–12 (Table 110–12 ).
TABLE 1. Adverse Effects of Primary Aldosteronism Compared With Essential Hypertension
Case Detection
The first step in diagnosing primary aldosteronism is case detection. Current guidelines recommend performing case detection in patients with resistant hypertension, as well as those who have well-controlled hypertension with certain clinical characteristics (Table 2).13 [corrected] Normokalemia should not dissuade clinicians from considering the diagnosis of primary aldosteronism. Hypokalemia is the exception, not the rule, and is seen in only about 30% of patients with primary aldosteronism.14
TABLE 2. Clinical Criteria for Patients Who Require Case Detection Testing for Primary Aldosteronism
| Controlled hypertension (any one of the following) |
| Adrenal nodule |
| Atrial fibrillation* |
| Family history of early stroke (i.e., younger than 40 years) |
| First-degree relative with primary aldosteronism |
| Hypokalemia |
| Obstructive sleep apnea |
| Resistant hypertension† |
| All patients |
Note: Criteria are based on the 2016 Endocrine Society Guidelines.
*—The Endocrine Society does not distinctly list atrial fibrillation as criteria, but it acknowledges that some centers recommend testing given its association with primary aldosteronism.
†—Resistant hypertension is hypertension that persists despite the concurrent use of three different classes of antihypertensive medications or the use of four antihypertensive medications to achieve adequate blood pressure control.
Initial case detection for primary aldosteronism is performed by simultaneously measuring plasma aldosterone concentration and plasma renin activity.10 Aldosterone elevation with suppressed plasma renin activity identifies patients with potential primary aldosteronism10,13 (Figure 110,13,15 ). An aldosterone-renin ratio of greater than 30 is considered positive.
FIGURE 1.
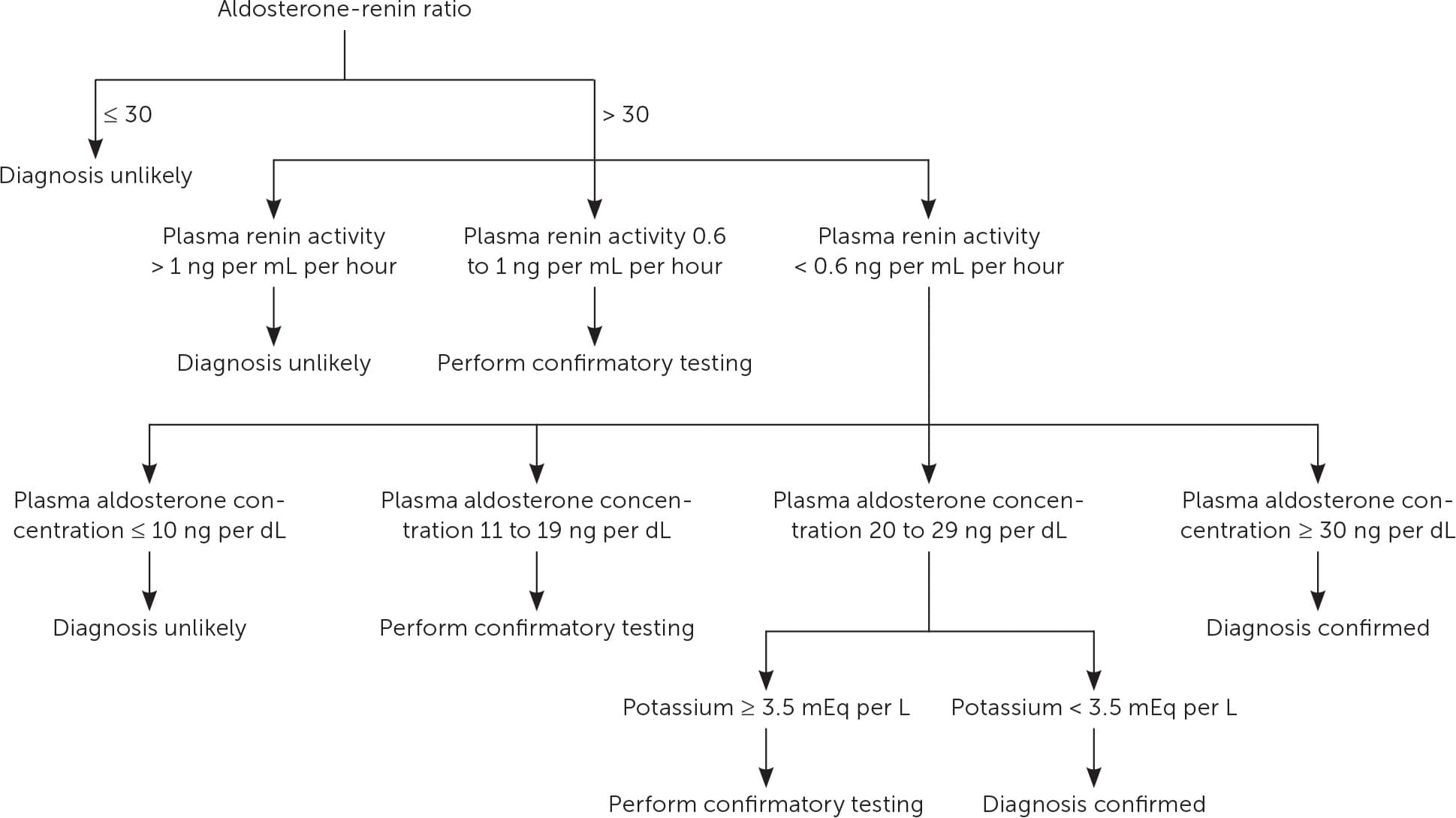
Suggested interpretation of initial case detection testing for primary aldosteronism.
Note: An aldosterone-renin ratio > 30 is the most common cutoff during initial case detection when plasma aldosterone concentration and plasma renin activity are in conventional units (ng per dL and ng per mL per hour, respectively). Values above the threshold should not be viewed in isolation because the aldosterone-renin ratio may be exaggerated in cases of very low renin levels without significant elevation of aldosterone.
Medications that significantly alter the RAAS, such as spironolactone, eplerenone, and amiloride, are ideally held before testing and, if needed, other antihypertensives should be substituted (Table 3).13
TABLE 3. Medications to Hold Before Primary Aldosteronism Testing*
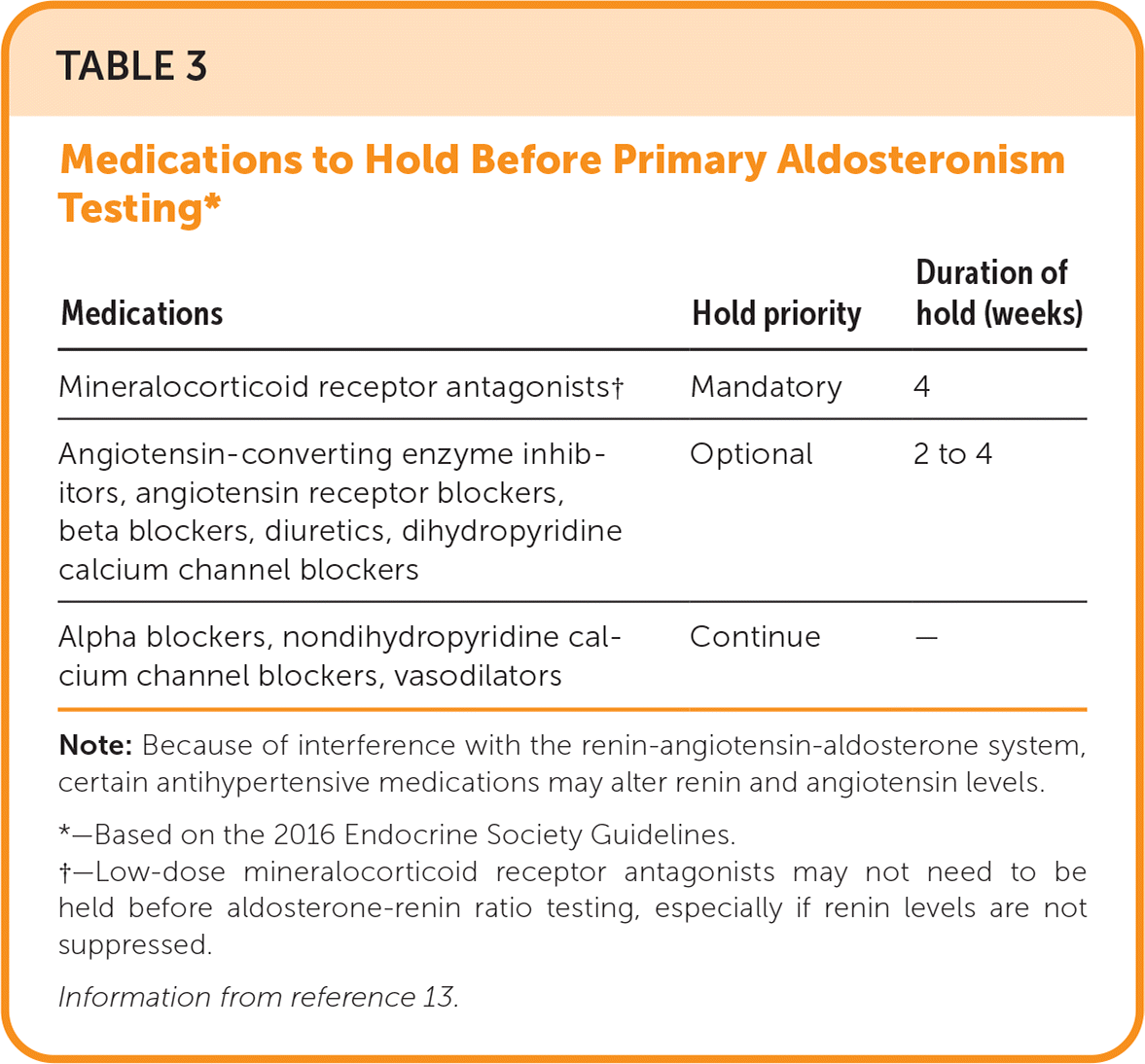
| Medications | Hold priority | Duration of hold (weeks) |
|---|---|---|
| Mineralocorticoid receptor antagonists† | Mandatory | 4 |
| Angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, beta blockers, diuretics, dihydropyridine calcium channel blockers | Optional | 2 to 4 |
| Alpha blockers, nondihydropyridine calcium channel blockers, vasodilators | Continue | — |
Note: Because of interference with the renin-angiotensin-aldosterone system, certain antihypertensive medications may alter renin and angiotensin levels.
*—Based on the 2016 Endocrine Society Guidelines.
†—Low-dose mineralocorticoid receptor antagonists may not need to be held before aldosterone-renin ratio testing, especially if renin levels are not suppressed.
Information from reference 13.
Confirmatory Testing
Confirmatory testing is often needed to make a formal diagnosis of primary aldosteronism. However, in some patients with a high probability of primary aldosteronism based on clinical presentation and initial biochemical screening (e.g., spontaneous hypokalemia [potassium level of less than 3.5 mEq per L] with suppressed plasma renin activity and elevated plasma aldosterone concentration), confirmatory testing can be omitted.16 Confirmatory testing had a near 100% specificity for a final diagnosis of primary aldosteronism in cases where plasma aldosterone concentration was greater than 30 ng per dL and plasma renin activity was less than 0.6 ng per mL per hour.15
For patients who require confirmatory testing, several tests are available, but there is no consensus on which one is preferred.17 Test results are considered positive when aldosterone levels remain elevated despite an intervention that would suppress physiologic aldosterone production. Salt loading and synthetic mineralocorticoid administration can worsen hypertension and hypokalemia; therefore, monitoring of blood pressure and serum potassium levels is recommended. These tests are time-consuming and require meticulous attention to detail; therefore, physicians may consider referral to an endocrinologist for confirmatory testing. These confirmatory tests are described below.
CAPTOPRIL CHALLENGE TEST
In the captopril challenge test, aldosterone levels are measured at baseline and then two hours after oral administration of 25 to 50 mg of the angiotensin-converting enzyme inhibitor captopril. In patients without primary aldosteronism, interruption of the RAAS by an angiotensin-converting enzyme inhibitor will cause a significant decrease in plasma aldosterone levels. A decrease of less than 30% is consistent with autonomous aldosterone secretion (i.e., primary aldosteronism).
ORAL SALT LOADING TEST
For the oral salt loading test, a high-salt diet supplemented with sodium chloride tablets is consumed for three days with a goal sodium intake of 6 g per day. High salt intake should cause physiologic suppression of the RAAS and a marked decrease in aldosterone levels. A 24-hour urine collection is performed on the third day. Persistently elevated 24-hour urine aldosterone levels (more than 12 mcg in 24 hours) are consistent with nonphysiologic production of aldosterone and confirm the diagnosis of primary aldosteronism. The saline infusion test can also confirm pathologic aldosterone production if plasma aldosterone concentration is greater than 10 ng per dL after an infusion of 2 L of normal saline.
FLUDROCORTISONE TEST
The fludrocortisone test involves administration of the synthetic mineralocorticoid fludrocortisone at a dosage of 0.1 mg every six hours for four days. Exogenous mineralocorticoid administration should suppress serum aldosterone levels. A plasma aldosterone concentration of greater than 6 ng per dL on day 4 confirms the diagnosis of primary aldosteronism.
Subtyping
Once primary aldosteronism has been diagnosed, the next step is to determine if aldosterone production is unilateral or bilateral.13,18,19 Unilateral production is typically caused by an aldosterone-producing adenoma and should be treated surgically, whereas bilateral production is typically from idiopathic hyperplasia and is treated medically.
This differentiation, termed subtyping, is accomplished with adrenal computed tomography (CT) and adrenal vein sampling.13,18,19 Adrenal CT has three phases: an initial scan without contrast media, a scan at 60 to 75 seconds after contrast media administration, and again at 15 minutes. By assessing baseline nodule density, as well as contrast media uptake and subsequent washout, benign adenomas can be reliably distinguished from malignant masses.20 CT has limited sensitivity for the detection of sub-centimeter nodules. Additionally, CT is unable to distinguish nonfunctioning adenomas from functioning adenomas. A systematic review showed an almost 40% rate of discordance between CT and adrenal vein sampling in subtyping patients with primary aldosteronism.21 Therefore, adrenal vein sampling is considered the preferred method of subtyping. A systematic review and meta-analysis performed in 2022 found a statistically significant higher rate of complete biochemical success when adrenalectomy was guided by adrenal vein sampling compared with adrenalectomy guided by CT alone (odds ratio = 2.78; 95% CI, 1.88 to 4.12).18
Adrenal vein sampling is a nuanced procedure. Blood samples are taken from a peripheral vein and the right and left adrenal veins and tested for aldosterone and cortisol levels.22 Success rates for adequate sampling range from 30.5%23 to 99.2%.24 Operator experience at a center that performs at least 12 procedures per year has been shown to be associated with higher sampling adequacy.25,26 An experienced and dedicated laboratory is necessary for a successful adrenal vein sampling program,27 and the results should be interpreted based on expert consensus guidelines.28,29
Treatment
UNILATERAL ALDOSTERONE PRODUCTION
Adrenalectomy is recommended in cases of unilateral aldosterone production. Although hypertension is cured in only approximately one-third of cases, biochemical cure is achieved in 94% of cases.30 Compared with medical management, adrenalectomy reduces the rate of composite adverse cardiovascular outcomes by one-half 31 and is associated with superior quality of life.32
BILATERAL ALDOSTERONE PRODUCTION
When aldosterone production is bilateral, medical therapy is necessary. Mineralocorticoid receptor antagonists are the cornerstone of therapy for patients with primary aldosteronism. They are often used concurrently with other antihypertensives. Dietary sodium restriction of less than 1,500 mg per day is recommended.33
Spironolactone is a nonselective mineralocorticoid receptor antagonist and is the initial medication of choice. Typical starting dosages are 12.5 to 25 mg per day and are increased, as needed, to a maximum dosage of 400 mg per day.10 Its dose-dependent antiandrogenic properties can lead to adverse effects, such as gynecomastia (more than 10%), erectile dysfunction, decreased libido, and irregular menses (1% to 10%).34 If these adverse effects occur, eplerenone, a more selective but less potent and more expensive mineralocorticoid receptor blocker, may be used.13,34
Recent observational studies have shown that titrating mineralocorticoid receptor antagonists based on plasma renin concentrations may lead to better outcomes.35,36 Therefore, future guidelines may include interval measurements of renin as part of the mineralocorticoid receptor antagonist dosing strategy.36
Data Sources: A PubMed search of clinical trials, meta-analyses, randomized controlled trials, and systematic reviews from 2000 to 2022 was completed using the key terms primary hyperaldosteronism, primary aldosteronism, and hyperaldosteronism. We also searched the Cochrane database, Agency for Healthcare Research and Quality (AHRQ), and Essential Evidence Plus using the same terms, but with limited results. If studies used race and/or gender as a patient category but did not define how these categories were assigned, they were excluded. Studies may not represent all populations. Physicians may need to exercise caution in applying such guidelines to populations not included (e.g., patients of color, younger individuals). Search dates: October 2, 2022, and May 19, 2023.


