Birthmarks in newborns can be classified as vascular, melanocytic or pigmented, or markers of underlying developmental abnormalities of the nervous system. A nevus simplex is a benign capillary malformation. Newborns with a nevus flammeus can be safely treated before one year of age with a pulsed dye laser to reduce the visibility of lesions. Infantile hemangiomas should be treated with systemic beta blockers if there is a risk of life-threatening complications, functional impairment, ulceration, underlying abnormalities, permanent scarring, or alteration of anatomic landmarks. Dermal melanocytosis is a benign finding that is easily recognized and does not warrant further evaluation. A solitary congenital melanocytic nevus that is less than 20 cm in diameter may be observed in primary care; children with larger or multiple nevi should be referred to pediatric dermatology due to the risk of melanoma. Newborns with skin markers of occult spinal dysraphism (other than a simple, solitary dimple) should have lumbar spine imaging using ultrasonography or magnetic resonance imaging.
Birthmarks are common sources of parental concern, and some require evaluation and treatment. Birthmarks can be classified as vascular, as melanocytic or pigmented, or as markers of underlying developmental abnormalities of the nervous system. Part II of this article reviews the identification and management of birthmarks that appear in the neonatal period, with an emphasis on prognosis and appropriate counseling for parents. Part I of this article, which also appears in this issue of American Family Physician, discusses the presentation, prognosis, and treatment of common rashes and skin changes that may present during an infant's first four weeks of life.1
SORT: KEY RECOMMENDATIONS FOR PRACTICE
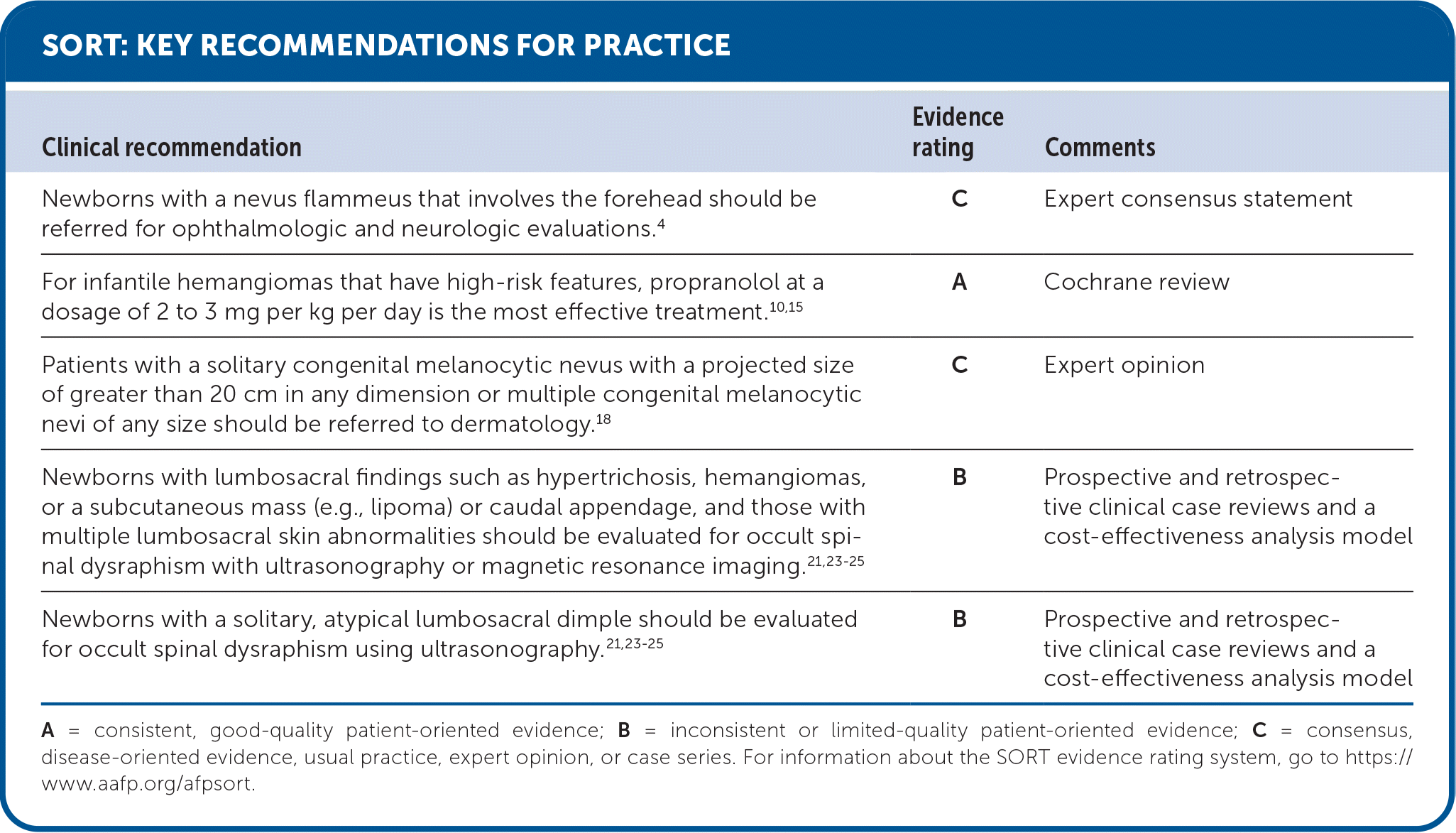
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Vascular Lesions
Vascular lesions result from abnormally formed blood vessels. Capillary malformations, such as a nevus simplex and nevus flammeus, are the most common presentations. Cutaneous vascular lesions can be associated with genetic syndromes or extracutaneous malformations. Early recognition, diagnosis, and treatment of syndromes associated with vascular lesions in infants can significantly impact quality of life.
NEVUS SIMPLEX
A nevus simplex is colloquially known as a stork bite, angel kiss, or salmon patch, and is present in 82% of newborns. These lesions present as pale pink to bright red or violet macules with indistinct borders that can be partially blanched with pressure on the skin (Figure 1). Most are benign and spontaneously regress by one year of age. Rarely, a nevus simplex can be associated with genetic syndromes including Beckwith-Wiedemann syndrome, port-wine nevi-mega cisterna magna-hydrocephalus syndrome (Nova syndrome), odontodysplasia, macrocephaly-capillary malformation syndrome, and Roberts-SC phocomelia syndrome.2 Neurologic symptoms, seizures, large head size, and limb malformations should prompt consideration of referral for genetic evaluation.
FIGURE 1.
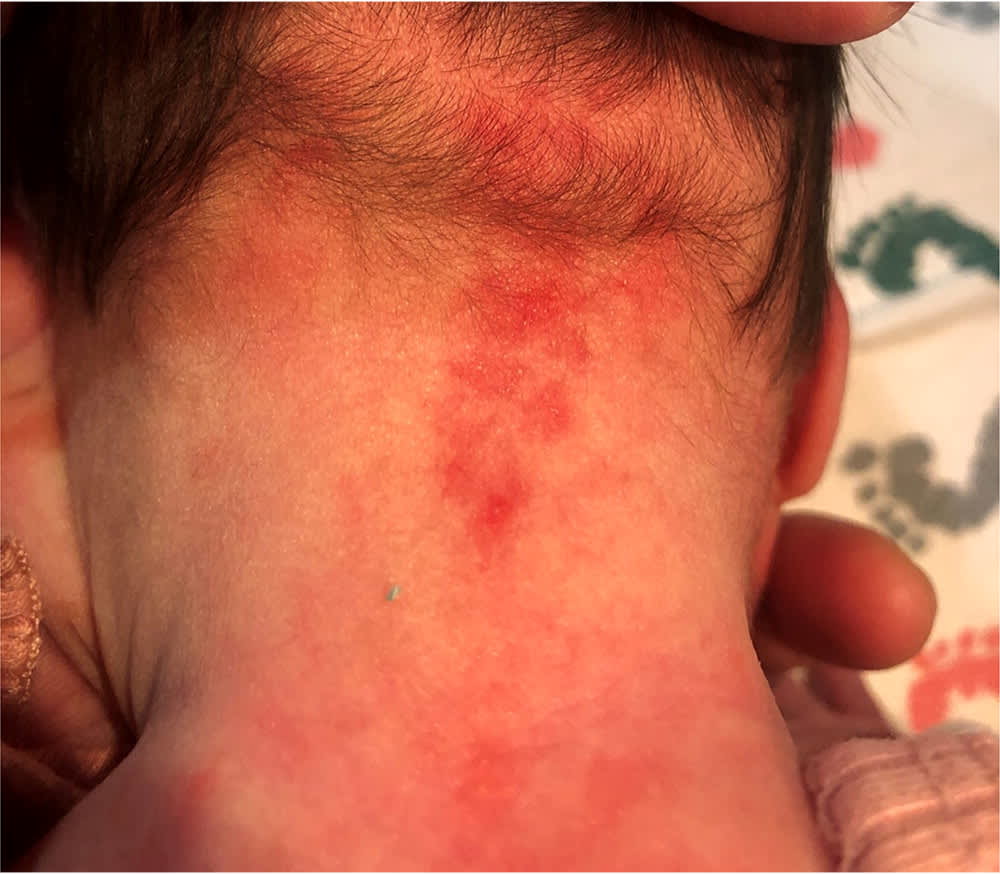
A nevus simplex is a transient capillary malformation.
NEVUS FLAMMEUS
A nevus flammeus, colloquially known as a port-wine stain, occurs in 3 to 5 per 1,000 live births and is most commonly present on the limbs or face. These lesions are well-demarcated, pink or red lesions composed of abnormally dilated capillaries and venules.3 Lesions on the limbs or trunk remain stable or lighten over time, whereas facial lesions tend to become darker, taking on the port-wine color.2 A nevus flammeus that involves the forehead may indicate Sturge-Weber syndrome, a congenital neurocutaneous syndrome characterized by vascular malformations in the brain or eyes; therefore, ophthalmologic and neurologic evaluations are recommended.4 Nevi flammeus can progress to include nodules, pyogenic granulomas, and soft tissue hypertrophy that involves underlying structures, such as gingiva. Treatment can reduce the visibility of lesions and associated psychological morbidity. A pulsed dye laser selectively destructs the involved capillaries. Treatment with pulsed dye laser is typically more effective in children than adults due to differences in skin thickness and capillary size.5 Treatment in infants younger than one year is effective and avoids the need for general anesthesia that is needed for treatment in older children.6,7 The most common adverse effects of treatment are transient purpura, localized edema, crusting, and blistering, but long-term complications such as pigmentary changes and scarring are rare.8
INFANTILE HEMANGIOMAS
Infantile hemangiomas are the most common benign vascular tumor in children, occurring in up to 5% of infants and 10% of premature infants. They are more common in infants with low birth weight, female sex, twin gestation, and premature birth.9,10 Clinical presentation at birth varies from bright strawberry-red lesions to pale patches or telangiectasias surrounded by pallor 11 (Figure 2). Hemangiomas proliferate rapidly between one and three months of age and typically stop growing by five months of age. Involution starts by 12 months of age and lesions disappear by four years of age in 80% of children.10
FIGURE 2.
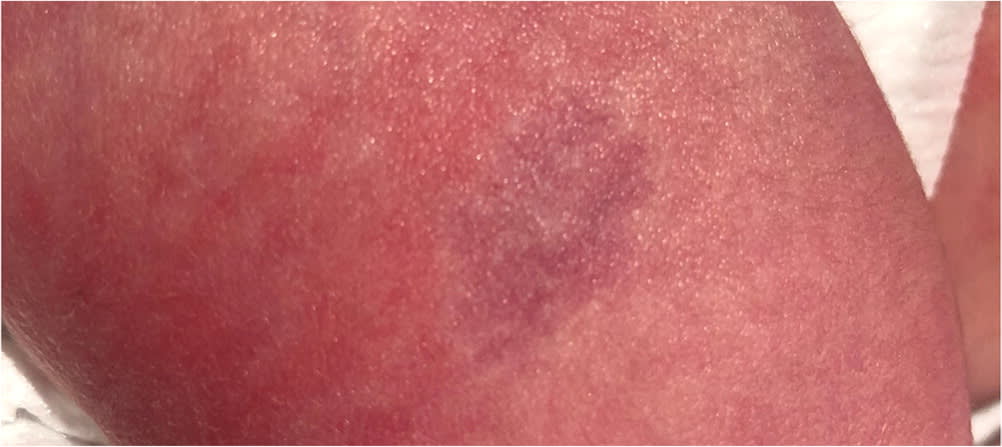
Infantile hemangioma presenting as telangiectasias surrounded by skin pallor.
Most hemangiomas are benign and do not require treatment. High-risk features are those that increase potential for life-threatening complications, functional impairment, ulceration, underlying abnormalities, permanent scarring, or alteration of anatomic landmarks. Hemangiomas on the face or anterior neck can be associated with functional impairment of feeding or vision or, if they are associated with airway hemangiomas, life-threatening complications. The presence of five or more hemangiomas increases the likelihood of hepatic hemangioma, which rarely can lead to high-output heart failure. Hemangiomas in the perineum, gluteal cleft, or lumbosacral area can be associated with occult spinal dysraphism or LUMBAR (lower body hemangioma, urogenital abnormalities/ulceration, myelopathy, bony deformities, anorectal malformations/arterial anomalies, and rectal anomalies) syndrome. Hemangiomas with a diameter greater than 5 cm that involve the face or scalp can be an indicator of PHACE (posterior fossa anomalies, hemangioma, arterial anomalies, cardiac anomalies, and eye anomalies) syndrome. Hemangiomas located over the axilla, much of the face, or diaper area have the potential for permanent scarring or alteration of key anatomic landmarks. Urgent referral to a hemangioma specialist is recommended if any high-risk features are present.10 The Infantile Hemangioma Referral Score screening tool (https://www.ihscoring.com) reliably identifies infantile hemangiomas that require referral, can be used quickly by primary care physicians, and has good interrater reliability between expert and nonexpert users.12,13
Imaging should be performed if there is concern for associated anatomic abnormalities. Abdominal Doppler ultrasonography is indicated in children with five or more cutaneous hemangiomas or if there is concern for LUMBAR syndrome. Infants should be evaluated for occult spinal dysraphism if hemangiomas are present in the perineum, gluteal cleft, or lumbosacral area. Magnetic resonance angiography of the head and neck should be performed if there is concern for PHACE syndrome.10
Success rates are significantly higher if treatment is started before 10 weeks of age, and current guidelines recommend starting by one month of age.10,14 Systemic beta blockers are the treatment of choice, specifically propranolol, 2 to 3 mg per kg per day.10 This is supported by moderate-quality evidence, with a number needed to treat of 2 vs. placebo for clearance of hemangiomas.15 Propranolol is superior to systemic atenolol in the rate of complete response; however, propranolol is associated with significantly more adverse effects.16 Treatment should be initiated in the inpatient setting for infants younger than five weeks' corrected gestational age or in older infants with poor social support or cardiovascular or respiratory comorbidities.10 Adverse effects of propranolol include changes in sleep, acrocyanosis, bronchial irritation, gastrointestinal symptoms, hypotension, bradycardia, or hypoglycemia.9,10 Propranolol administration with or after feeding and withholding the medication during times of reduced intake or vomiting can lower the risk of treatment-associated hypoglycemia. Systemic or intralesional corticosteroids and topical timolol are alternatives to propranolol.10
Melanocytic Lesions
Melanocytic or pigmented lesions are caused by displaced melanocytes, such as those in dermal melanocytosis, or atypical growth of melanocytes, such as those in congenital melanocytic nevi.
DERMAL MELANOCYTOSIS
Dermal melanocytosis is a benign, hyperpigmented macule primarily found in the lumbosacral and gluteal regions (Figure 3). Single or multiple macules that are blueish gray in color may be present and should be documented during physical examination. No treatment is required. Parents can be reassured that dermal melanocytosis will fade by two years of age and may disappear by puberty.17
FIGURE 3.
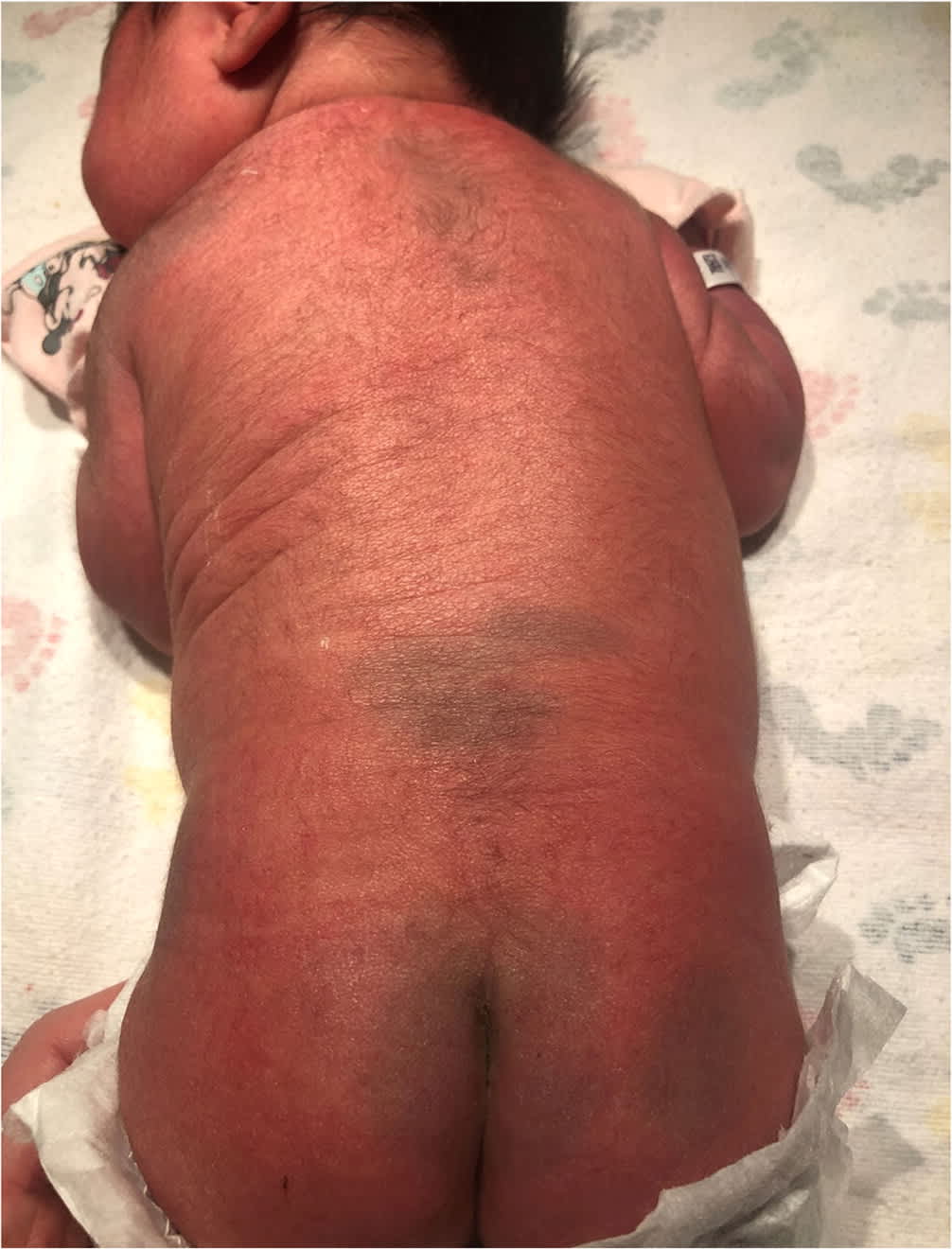
Dermal melanocytosis is characterized by benign, hyperpigmented macules primarily found in the lumbosacral and gluteal regions.
CONGENITAL MELANOCYTIC NEVI
Congenital melanocytic nevi are pigmented lesions that affect up to 1 out of 17 newborns. Lesions vary in size from a few millimeters to several centimeters, may be flat or palpable, and most commonly occur on the trunk, extremities, or face. The most common locations are the lower back and thigh area. Color may be uniform or variable within the same lesion but is predominantly shades of black or brown18 (Figure 4). Locations and sizes of congenital melanocytic nevi should be clearly documented during physical examination. Lesions tend to increase in size as the child ages, and the rate of increase depends on the location. Lesions on the head will likely double in size, whereas those on the trunk and extremities will likely triple in size.19
FIGURE 4.

Congenital melanocytic nevus on the lower extremity.
The primary concern with a congenital melanocytic nevus is the risk of melanoma. Lifetime risk of melanoma in any congenital melanocytic nevus is up to 2.2%; however, risk increases with projected adult size. Solitary lesions with a projected size of less than 20 cm may be observed, with referral for any change in color, texture, size, bleeding, or pain. Immediate dermatologist referral is recommended for a projected size of greater than 20 cm or with multiple congenital melanocytic nevi.18 Historically, surgical removal has been recommended; however, removal does not eliminate the risk of melanoma.18–20
Markers of Spinal Dysraphism
Occult spinal dysraphisms are skin-covered developmental abnormalities of the nervous system. Early recognition of skin markers of occult spinal dysraphisms and evaluation and surgical correction in affected newborns can prevent irreversible neurologic damage before neurologic impairment is clinically apparent.21 Most cases of occult spinal dysraphisms are associated with midline lumbosacral skin abnormalities such as atypical dimples, hypertrichosis, capillary hemangiomas, subcutaneous masses, including lipoma, or a caudal appendage21 (Figure 5). Features of atypical sacral dimples include dimples that are larger than 5 mm, located more than 25 mm from the anus or off the midline, or with no visible skin at the base.21,22 A solitary dimple is a low-risk finding.22 Lumbosacral skin findings other than simple, solitary dimples, especially two or more skin findings, suggest an increased risk of occult spinal dysraphisms.20,22,23
FIGURE 5.
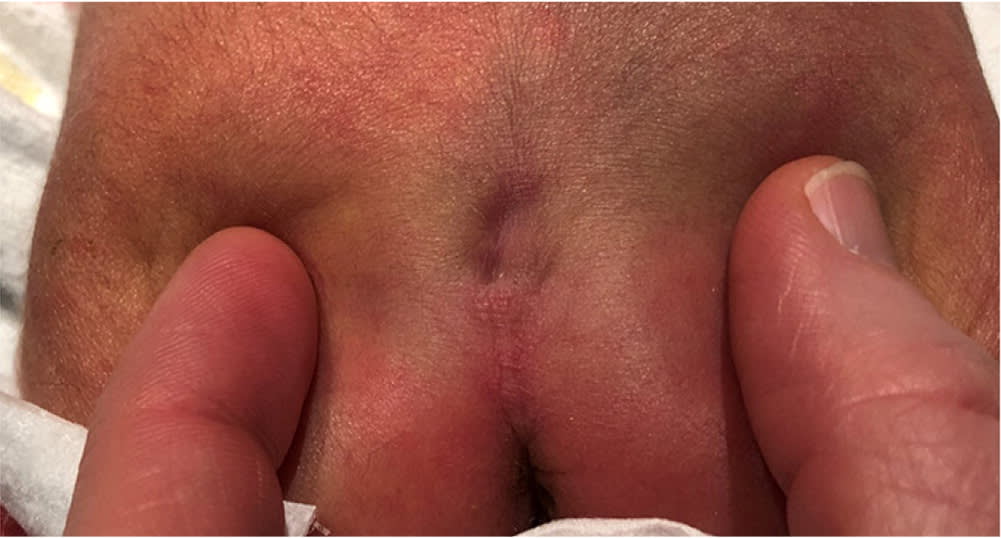
Cutaneous stigmata of occult spinal dysraphism. This newborn has two simple dimples and a small, palpable, subcutaneous mass below the more caudal dimple. There is also an unrelated dermal melanocytosis.
Ultrasonography and magnetic resonance imaging (MRI) can be used to evaluate newborns with markers of occult spinal dysraphisms (Table 1).20,21,23–25 Although ultrasonography is more difficult to obtain in newborns after three months of age due to ossification of the vertebral bodies, MRI requires the use of sedation.21,22 Ultrasonography is cost-effective for initial evaluation in patients with a solitary atypical dimple, has good diagnostic agreement with MRI, and appropriately identifies patients with spinal lesions that may require intervention.23–25 In patients with multiple dimples or cutaneous findings other than dimples that infer a higher risk of a surgical lesion, evaluation with MRI may be a more time efficient and cost-effective approach; however, in resource-limited settings, ultrasonography is appropriate as initial imaging.21,23,25 Newborns should be referred to neurosurgery for any abnormal imaging findings or before imaging if there is discharge from any dimple.21
TABLE 1. Evaluation of Cutaneous Markers of Occult Spinal Dysraphism
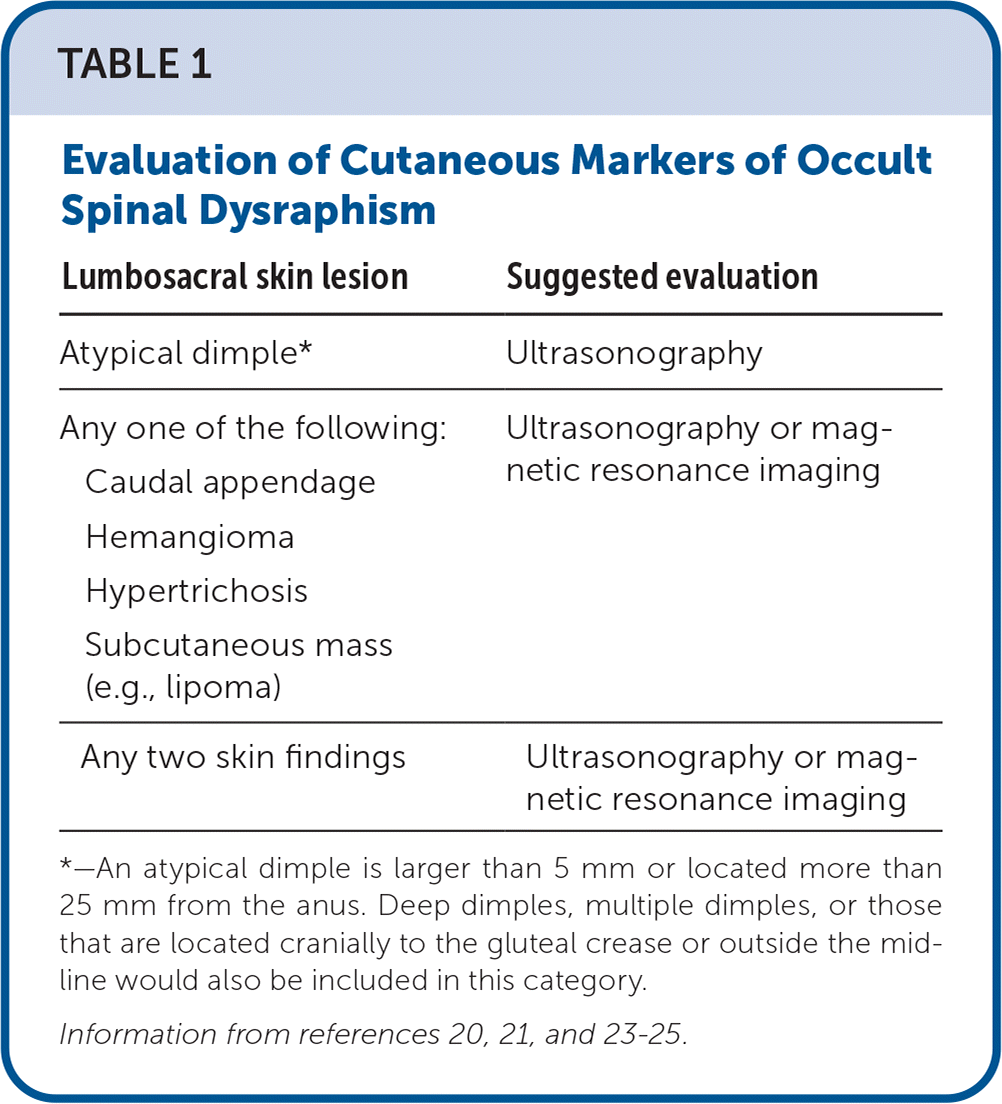
| Lumbosacral skin lesion | Suggested evaluation |
|---|---|
| Atypical dimple* | Ultrasonography |
| Any one of the following: | Ultrasonography or magnetic resonance imaging |
| Caudal appendage | |
| Hemangioma | |
| Hypertrichosis | |
| Subcutaneous mass (e.g., lipoma) | |
| Any two skin findings | Ultrasonography or magnetic resonance imaging |
*—An atypical dimple is larger than 5 mm or located more than 25 mm from the anus. Deep dimples, multiple dimples, or those that are located cranially to the gluteal crease or outside the mid-line would also be included in this category.
This article updates a previous article on this topic by McLaughlin, et al.20
Data Sources: A PubMed search was completed using the terms nevus simplex, nevus flammeus, infantile hemangiomas, dermal melanocytosis, congenital melanocytic nevi, markers of spinal dysraphism, and key terms for diagnosis and management. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. The Cochrane database, UpToDate, Essential Evidence Plus, and the TRIP database were also searched. Search dates: November 2022 to February 2023, May to June 2023, and January 2024.
The authors thank the patients’ families who allowed their newborns to be photographed for this article.

