Heart failure is an increasingly common condition resulting in high rates of morbidity and mortality. For patients who have heart failure and reduced ejection fraction, randomized clinical trials demonstrate consistent mortality benefit from angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, direct-acting vasodilators, beta blockers, and aldosterone antagonists. Additionally, some data show benefits from two new classes of drugs: angiotensin receptor blocker/neprilysin inhibitor and sinus node modulator. Diuretics and digoxin can be used as needed for symptom control. Statins are not recommended solely for treatment of heart failure. Implantable cardioverter-defibrillators and biventricular pacemakers improve mortality and function in selected patients. For patients who have been hospitalized for heart failure, disease management programs and telemonitoring can reduce hospitalizations and mortality.
American adults 40 years and older have about a 20% lifetime risk of developing heart failure.1 The diagnosis is associated with significant morbidity and mortality. Over the past three decades, the prevalence of heart failure in the United States has continued to increase, largely because the population is aging, and older individuals are at higher risk of developing the disease.2
WHAT IS NEW ON THIS TOPIC: HEART FAILURE
Based on limited high-quality evidence, American College of Cardiology/American Heart Association/Heart Failure Society of America guidelines recommend ARB/neprilysin inhibitor therapy as a first-line alternative to ACE inhibitors for those with symptomatic heart failure who are not hypotensive or intolerant of angiotensin system antagonists.
Disease management programs and telemonitoring can reduce hospitalizations and mortality, especially for patients who have previously been hospitalized for heart failure.
ACE = angiotensin-converting enzyme; ARB = angiotensin receptor blocker.
SORT: KEY RECOMMENDATIONS FOR PRACTICE

| Clinical recommendation | Evidence rating | References |
|---|---|---|
| The combination of sacubitril/valsartan (Entresto) may be an alternative to angiotensin-converting enzyme inhibitors in patients with heart failure. | B | 13 |
| Aldosterone antagonists and beta blockers decrease mortality in patients with symptomatic heart failure. | A | 5–10 |
| The sinus modulator ivabradine (Corlanor) decreases the risk of cardiovascular death or hospitalization in patients with sinus rhythm, with a heart rate of more than 70 beats per minute, and who are taking the tolerated or target dosage of a beta blocker. | B | 14 |
| Consider referral for device therapy (implantable cardioverter-defibrillators and cardiac resynchronization therapy) in patients with reduced ejection fraction and symptomatic heart failure or ischemic cardiomyopathy whose life expectancy is more than one year. | A | 22–24, 42 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Clinical trials of patients with left ventricular systolic dysfunction, also called heart failure with reduced ejection fraction, have demonstrated that angiotensin-converting enzyme (ACE) inhibitors,3 angiotensin receptor blockers (ARBs),4 beta blockers,5–7 aldosterone antagonists,8–10 and in selected populations isosorbide dinitrate/hydralazine (Bidil)11,12 reduce all-cause mortality and hospitalizations for heart failure. More recent clinical trials have demonstrated benefit from an ARB/neprilysin inhibitor and from a sinus node modulator.13,14
Overall adjusted hospitalization rates for Medicare patients with heart failure dropped almost 30% between 1998 and 2008, partly because of application of these effective therapies.15 Also, risk-adjusted one-year mortality declined from 31.7% to 29.6%. These benefits occurred even though an analysis of 137 cardiology practices found only 80% of eligible patients were on ACE inhibitors or ARBs, 86% were on beta blockers, and 34% were on aldosterone antagonists.16
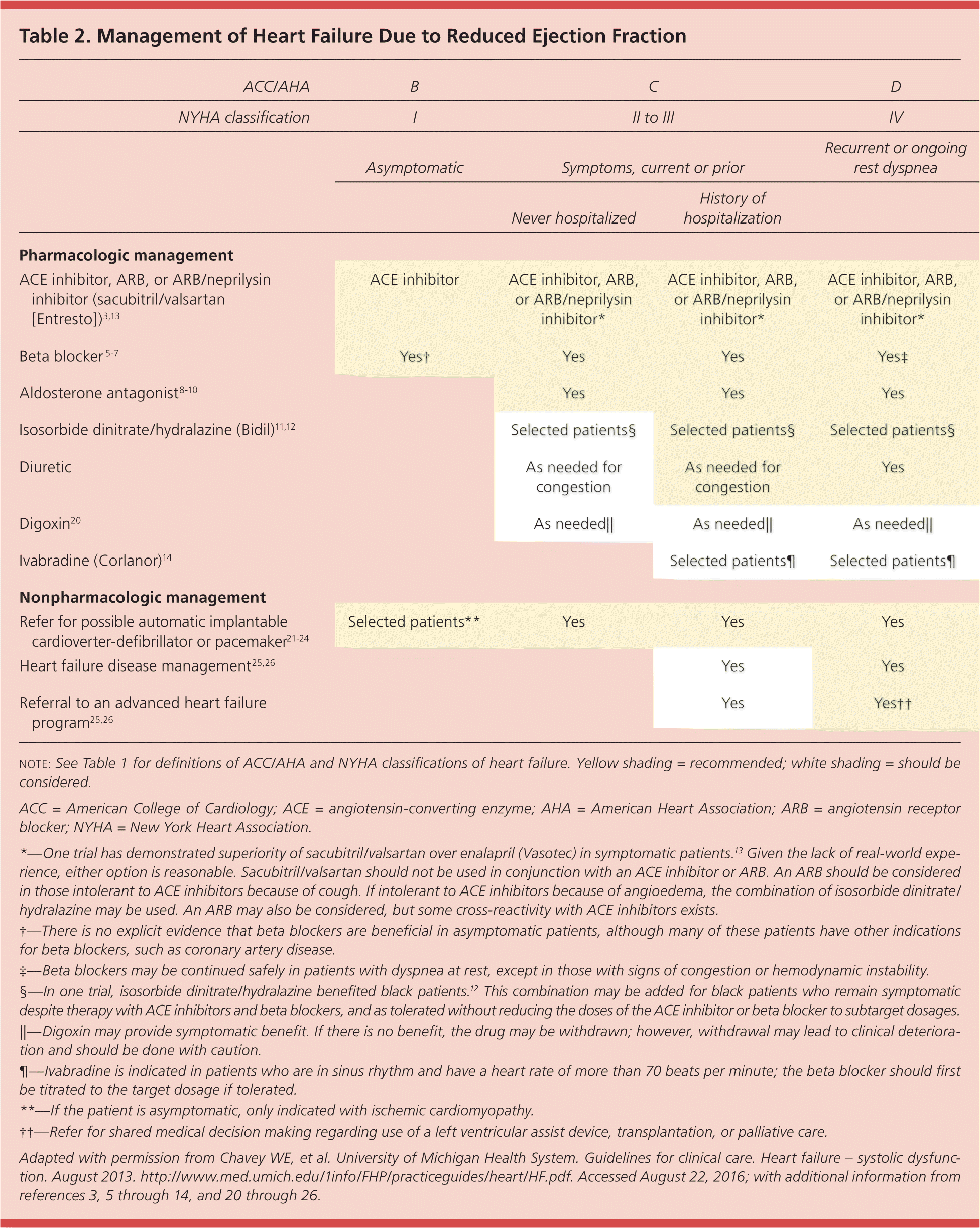
This article provides a symptom-based strategy for the treatment of heart failure with reduced ejection fraction. Symptoms of heart failure, often classified by the New York Heart Association (NYHA) system and occasionally by the American College of Cardiology/American Heart Association (ACC/AHA) system, are summarized in Table 1.17 Patients with symptoms and a history of hospitalization for heart failure are at significantly higher risk of subsequent mortality and recurrent hospitalization for heart failure. American Family Physician has published articles on the prevention and diagnosis of heart failure and the management of heart failure due to preserved ejection fraction.18,19 Electrophysiologic devices (defibrillators and biventricular pacing for resynchronization) and advanced heart failure therapies (mechanical assist devices, cardiac transplantation) are beyond the scope of this article. Table 2 summarizes heart failure symptom progression and suggested management. 3,5–14,20–27
Table 1. Functional Classification Systems for Heart Failure

| New York Heart Association classification |
| I: Asymptomatic |
| II: Minor symptoms, symptoms with modest exertion |
| III: Moderate symptoms, symptoms with minor exertion |
| IV: Symptoms at rest |
| American College of Cardiology/American Heart Association classification |
| A: At risk of heart failure but without structural disease |
| B: Structural heart failure but without symptoms |
| C: Structural heart failure with current or prior symptoms |
| D: Symptoms at rest |
Information from reference 17.
Table 2. Management of Heart Failure Due to Reduced Ejection Fraction
| ACC/AHA | B | C | D | |
|---|---|---|---|---|
| NYHA classification | I | II to III | IV | |
| Asymptomatic | Symptoms, current or prior | Recurrent or ongoing rest dyspnea | ||
| Never hospitalized | History of hospitalization | |||
| Pharmacologic management | ||||
| ACE inhibitor, ARB, or ARB/neprilysin inhibitor (sacubitril/valsartan [Entresto])3,13 | ACE inhibitor | ACE inhibitor, ARB, or ARB/neprilysin inhibitor* | ACE inhibitor, ARB, or ARB/neprilysin inhibitor* | ACE inhibitor, ARB, or ARB/neprilysin inhibitor* |
| Beta blocker 5–7 | Yes† | Yes | Yes | Yes‡ |
| Aldosterone antagonist8–10 | Yes | Yes | Yes | |
| Isosorbide dinitrate/hydralazine (Bidil)11,12 | Selected patients§ | Selected patients§ | Selected patients§ | |
| Diuretic | As needed for congestion | As needed for congestion | Yes | |
| Digoxin20 | As needed|| | As needed|| | As needed|| | |
| Ivabradine (Corlanor)14 | Selected patients¶ | Selected patients¶ | ||
| Nonpharmacologic management | ||||
| Refer for possible automatic implantable cardioverter-defibrillator or pacemaker21–24 | Selected patients** | Yes | Yes | Yes |
| Heart failure disease management25,26 | Yes | Yes | ||
| Referral to an advanced heart failure program25,26 | Yes | Yes†† | ||
note: See Table 1 for definitions of ACC/AHA and NYHA classifications of heart failure. Yellow shading = recommended; white shading = should be considered.
ACC = American College of Cardiology; ACE = angiotensin-converting enzyme; AHA = American Heart Association; ARB = angiotensin receptor blocker; NYHA = New York Heart Association.
*—One trial has demonstrated superiority of sacubitril/valsartan over enalapril (Vasotec) in symptomatic patients.13Given the lack of real-world experience, either option is reasonable. Sacubitril/valsartan should not be used in conjunction with an ACE inhibitor or ARB. An ARB should be considered in those intolerant to ACE inhibitors because of cough. If intolerant to ACE inhibitors because of angioedema, the combination of isosorbide dinitrate/hydralazine may be used. An ARB may also be considered, but some cross-reactivity with ACE inhibitors exists.
†—There is no explicit evidence that beta blockers are beneficial in asymptomatic patients, although many of these patients have other indications for beta blockers, such as coronary artery disease.
‡—Beta blockers may be continued safely in patients with dyspnea at rest, except in those with signs of congestion or hemodynamic instability.
§—In one trial, isosorbide dinitrate/hydralazine benefited black patients.12 This combination may be added for black patients who remain symptomatic despite therapy with ACE inhibitors and beta blockers, and as tolerated without reducing the doses of the ACE inhibitor or beta blocker to subtarget dosages.
||—Digoxin may provide symptomatic benefit. If there is no benefit, the drug may be withdrawn; however, withdrawal may lead to clinical deterioration and should be done with caution.
¶—Ivabradine is indicated in patients who are in sinus rhythm and have a heart rate of more than 70 beats per minute; the beta blocker should first be titrated to the target dosage if tolerated.
**—If the patient is asymptomatic, only indicated with ischemic cardiomyopathy.
††—Refer for shared medical decision making regarding use of a left ventricular assist device, transplantation, or palliative care.
Adapted with permission from Chavey WE, et al. University of Michigan Health System. Guidelines for clinical care. Heart failure – systolic dysfunction. August 2013. http://www.med.umich.edu/1info/FHP/practiceguides/heart/HF.pdf. Accessed August 22, 2016; with additional information from references 3, 5 through 14, and 20 through 26.
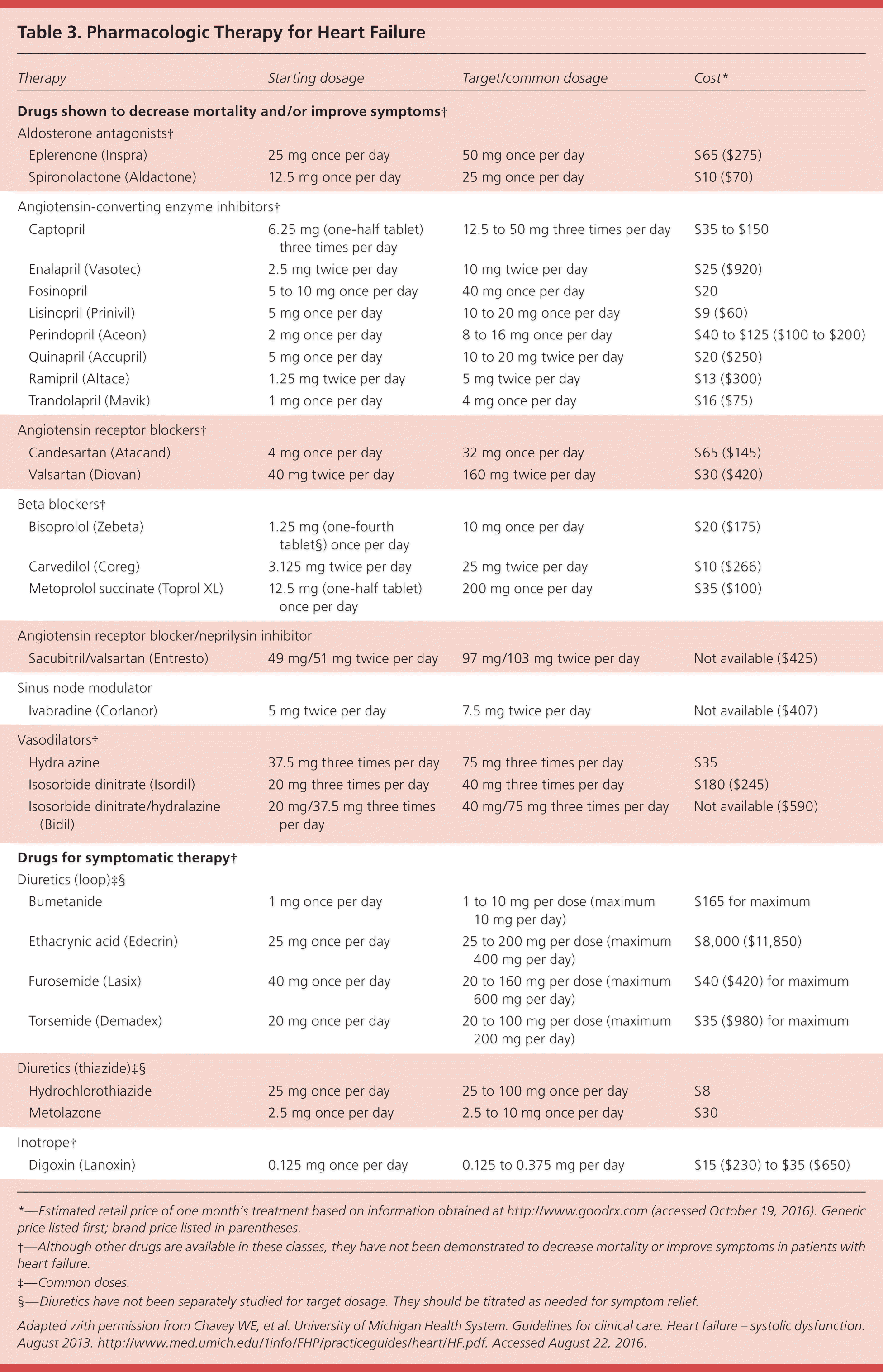
Pharmacologic Treatment Options
Table 3. Pharmacologic Therapy for Heart Failure
| Therapy | Starting dosage | Target/common dosage | Cost* | |
|---|---|---|---|---|
| Drugs shown to decrease mortality and/or improve symptoms† | ||||
| Aldosterone antagonists† | ||||
| Eplerenone (Inspra) | 25 mg once per day | 50 mg once per day | $65 ($275) | |
| Spironolactone (Aldactone) | 12.5 mg once per day | 25 mg once per day | $10 ($70) | |
| Angiotensin-converting enzyme inhibitors† | ||||
| Captopril | 6.25 mg (one-half tablet) three times per day | 12.5 to 50 mg three times per day | $35 to $150 | |
| Enalapril (Vasotec) | 2.5 mg twice per day | 10 mg twice per day | $25 ($920) | |
| Fosinopril | 5 to 10 mg once per day | 40 mg once per day | $20 | |
| Lisinopril (Prinivil) | 5 mg once per day | 10 to 20 mg once per day | $9 ($60) | |
| Perindopril (Aceon) | 2 mg once per day | 8 to 16 mg once per day | $40 to $125 ($100 to $200) | |
| Quinapril (Accupril) | 5 mg once per day | 10 to 20 mg twice per day | $20 ($250) | |
| Ramipril (Altace) | 1.25 mg twice per day | 5 mg twice per day | $13 ($300) | |
| Trandolapril (Mavik) | 1 mg once per day | 4 mg once per day | $16 ($75) | |
| Angiotensin receptor blockers† | ||||
| Candesartan (Atacand) | 4 mg once per day | 32 mg once per day | $65 ($145) | |
| Valsartan (Diovan) | 40 mg twice per day | 160 mg twice per day | $30 ($420) | |
| Beta blockers† | ||||
| Bisoprolol (Zebeta) | 1.25 mg (one-fourth tablet§) once per day | 10 mg once per day | $20 ($175) | |
| Carvedilol (Coreg) | 3.125 mg twice per day | 25 mg twice per day | $10 ($266) | |
| Metoprolol succinate (Toprol XL) | 12.5 mg (one-half tablet) once per day | 200 mg once per day | $35 ($100) | |
| Angiotensin receptor blocker/neprilysin inhibitor | ||||
| Sacubitril/valsartan (Entresto) | 49 mg/51 mg twice per day | 97 mg/103 mg twice per day | Not available ($425) | |
| Sinus node modulator | ||||
| Ivabradine (Corlanor) | 5 mg twice per day | 7.5 mg twice per day | Not available ($407) | |
| Vasodilators† | ||||
| Hydralazine | 37.5 mg three times per day | 75 mg three times per day | $35 | |
| Isosorbide dinitrate (Isordil) | 20 mg three times per day | 40 mg three times per day | $180 ($245) | |
| Isosorbide dinitrate/hydralazine (Bidil) | 20 mg/37.5 mg three times per day | 40 mg/75 mg three times per day | Not available ($590) | |
| Drugs for symptomatic therapy† | ||||
| Diuretics (loop)ठ| ||||
| Bumetanide | 1 mg once per day | 1 to 10 mg per dose (maximum 10 mg per day) | $165 for maximum | |
| Ethacrynic acid (Edecrin) | 25 mg once per day | 25 to 200 mg per dose (maximum 400 mg per day) | $8,000 ($11,850) | |
| Furosemide (Lasix) | 40 mg once per day | 20 to 160 mg per dose (maximum 600 mg per day) | $40 ($420) for maximum | |
| Torsemide (Demadex) | 20 mg once per day | 20 to 100 mg per dose (maximum 200 mg per day) | $35 ($980) for maximum | |
| Diuretics (thiazide)ठ| ||||
| Hydrochlorothiazide | 25 mg once per day | 25 to 100 mg once per day | $8 | |
| Metolazone | 2.5 mg once per day | 2.5 to 10 mg once per day | $30 | |
| Inotrope† | ||||
| Digoxin (Lanoxin) | 0.125 mg once per day | 0.125 to 0.375 mg per day | $15 ($230) to $35 ($650) | |
*—Estimated retail price of one month's treatment based on information obtained at http://www.goodrx.com (accessed October 19, 2016). Generic price listed first; brand price listed in parentheses.
†—Although other drugs are available in these classes, they have not been demonstrated to decrease mortality or improve symptoms in patients with heart failure.
‡—Common doses.
§—Diuretics have not been separately studied for target dosage. They should be titrated as needed for symptom relief.
Adapted with permission from Chavey WE, et al. University of Michigan Health System. Guidelines for clinical care. Heart failure – systolic dysfunction. August 2013. http://www.med.umich.edu/1info/FHP/practiceguides/heart/HF.pdf. Accessed August 22, 2016.
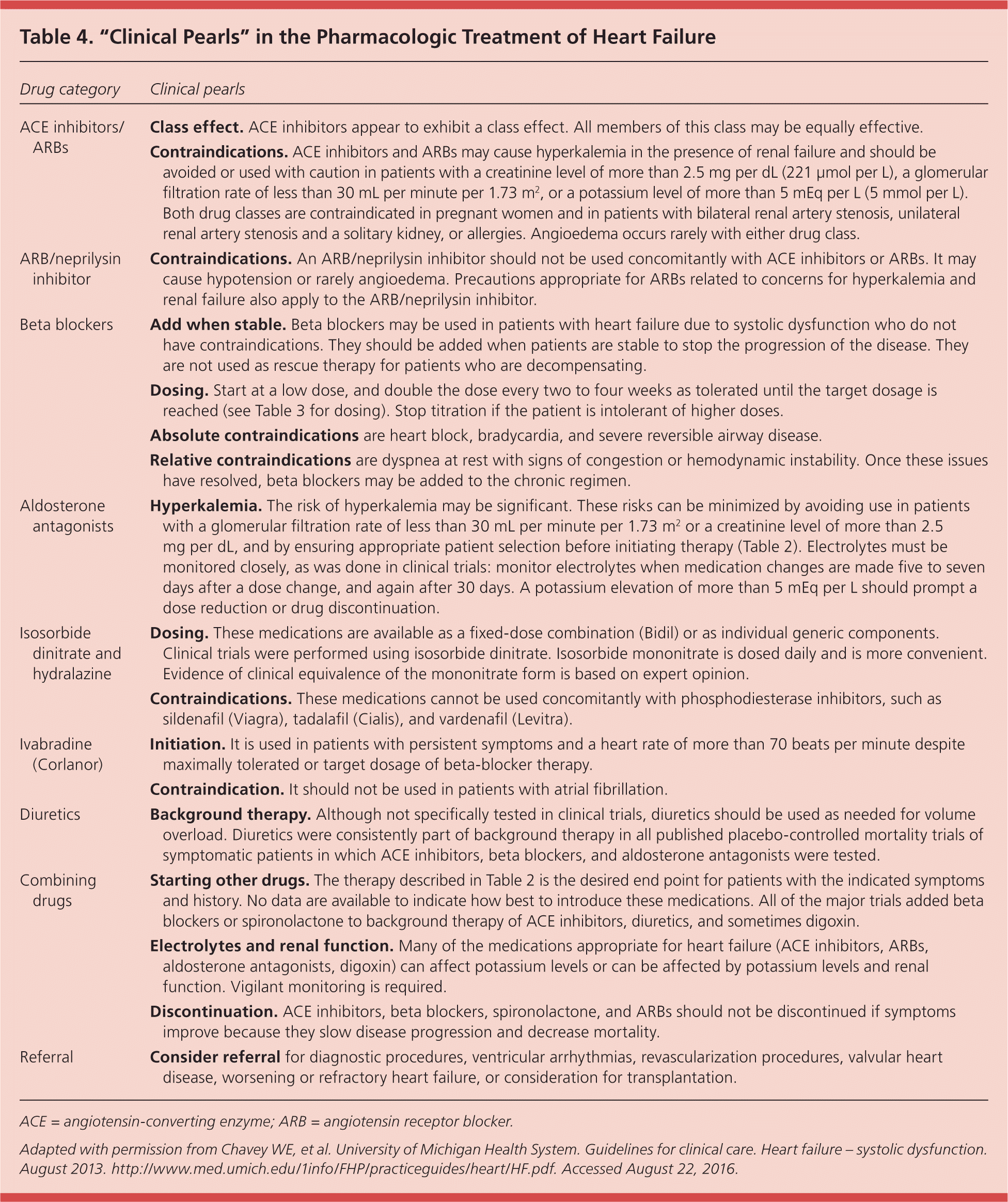
Table 4. “Clinical Pearls” in the Pharmacologic Treatment of Heart Failure
| Drug category | Clinical pearls |
|---|---|
| ACE inhibitors/ARBs | Class effect. ACE inhibitors appear to exhibit a class effect. All members of this class may be equally effective. |
| Contraindications. ACE inhibitors and ARBs may cause hyperkalemia in the presence of renal failure and should be avoided or used with caution in patients with a creatinine level of more than 2.5 mg per dL (221 μmol per L), a glomerular filtration rate of less than 30 mL per minute per 1.73 m2, or a potassium level of more than 5 mEq per L (5 mmol per L). Both drug classes are contraindicated in pregnant women and in patients with bilateral renal artery stenosis, unilateral renal artery stenosis and a solitary kidney, or allergies. Angioedema occurs rarely with either drug class. | |
| ARB/neprilysin inhibitor | Contraindications. An ARB/neprilysin inhibitor should not be used concomitantly with ACE inhibitors or ARBs. It may cause hypotension or rarely angioedema. Precautions appropriate for ARBs related to concerns for hyperkalemia and renal failure also apply to the ARB/neprilysin inhibitor. |
| Beta blockers | Add when stable. Beta blockers may be used in patients with heart failure due to systolic dysfunction who do not have contraindications. They should be added when patients are stable to stop the progression of the disease. They are not used as rescue therapy for patients who are decompensating. |
| Dosing. Start at a low dose, and double the dose every two to four weeks as tolerated until the target dosage is reached (see Table 3 for dosing). Stop titration if the patient is intolerant of higher doses. | |
| Absolute contraindications are heart block, bradycardia, and severe reversible airway disease. | |
| Relative contraindications are dyspnea at rest with signs of congestion or hemodynamic instability. Once these issues have resolved, beta blockers may be added to the chronic regimen. | |
| Aldosterone antagonists | Hyperkalemia. The risk of hyperkalemia may be significant. These risks can be minimized by avoiding use in patients with a glomerular filtration rate of less than 30 mL per minute per 1.73 m2 or a creatinine level of more than 2.5 mg per dL, and by ensuring appropriate patient selection before initiating therapy (Table 2). Electrolytes must be monitored closely, as was done in clinical trials: monitor electrolytes when medication changes are made five to seven days after a dose change, and again after 30 days. A potassium elevation of more than 5 mEq per L should prompt a dose reduction or drug discontinuation. |
| Isosorbide dinitrate and hydralazine | Dosing. These medications are available as a fixed-dose combination (Bidil) or as individual generic components. Clinical trials were performed using isosorbide dinitrate. Isosorbide mononitrate is dosed daily and is more convenient. Evidence of clinical equivalence of the mononitrate form is based on expert opinion. |
| Contraindications. These medications cannot be used concomitantly with phosphodiesterase inhibitors, such as sildenafil (Viagra), tadalafil (Cialis), and vardenafil (Levitra). | |
| Ivabradine (Corlanor) | Initiation. It is used in patients with persistent symptoms and a heart rate of more than 70 beats per minute despite maximally tolerated or target dosage of beta-blocker therapy. |
| Contraindication. It should not be used in patients with atrial fibrillation. | |
| Diuretics | Background therapy. Although not specifically tested in clinical trials, diuretics should be used as needed for volume overload. Diuretics were consistently part of background therapy in all published placebo-controlled mortality trials of symptomatic patients in which ACE inhibitors, beta blockers, and aldosterone antagonists were tested. |
| Combining drugs | Starting other drugs. The therapy described in Table 2 is the desired end point for patients with the indicated symptoms and history. No data are available to indicate how best to introduce these medications. All of the major trials added beta blockers or spironolactone to background therapy of ACE inhibitors, diuretics, and sometimes digoxin. |
| Electrolytes and renal function. Many of the medications appropriate for heart failure (ACE inhibitors, ARBs, aldosterone antagonists, digoxin) can affect potassium levels or can be affected by potassium levels and renal function. Vigilant monitoring is required. | |
| Discontinuation. ACE inhibitors, beta blockers, spironolactone, and ARBs should not be discontinued if symptoms improve because they slow disease progression and decrease mortality. | |
| Referral | Consider referral for diagnostic procedures, ventricular arrhythmias, revascularization procedures, valvular heart disease, worsening or refractory heart failure, or consideration for transplantation. |
ACE = angiotensin-converting enzyme; ARB = angiotensin receptor blocker.
Adapted with permission from Chavey WE, et al. University of Michigan Health System. Guidelines for clinical care. Heart failure – systolic dysfunction. August 2013. http://www.med.umich.edu/1info/FHP/practiceguides/heart/HF.pdf. Accessed August 22, 2016.
ACE INHIBITORS
Since the 1980s, ACE inhibitors have been the cornerstone of therapy for heart failure in patients without contraindications or intolerance. With the exception of isosorbide dinitrate/hydralazine11 and ARBs,4 which demonstrated benefit among those who cannot tolerate ACE inhibitors, all other heart failure therapies have been studied in patients who were already on ACE inhibitors. All ACE inhibitors that have been tested in clinical trials have shown benefit, and thus exhibit a class effect.
ARB/NEPRILYSIN INHIBITOR
The inhibition of neutral endopeptidase (neprilysin), a protease, leads to increasing concentrations of natriuretic and vasoactive peptides.28 Targeted neprilysin inhibition can produce beneficial and deleterious effects.29 The PARADIGM-HF study aimed to assess the impact of the neprilysin inhibitor sacubitril combined with the ARB valsartan (Entresto) among patients with heart failure and reduced ejection fraction.13 This drug company–sponsored trial included 8,842 patients with symptomatic heart failure who received sacubitril/valsartan or the ACE inhibitor enalapril (Vasotec) at a dosage of 10 mg twice daily. The primary outcome was a composite of death from cardiovascular causes or a first hospitalization from heart failure. The trial was stopped early at 27 months (the planned duration was 34 months) because of demonstrated benefit. The primary outcome was reached in 26.5% of patients taking enalapril vs. 21.8% of those taking sacubitril/valsartan (hazard ratio [HR] = 0.80; P < .001; number needed to treat [NNT] = 22). Although it was not the primary outcome, death from cardiovascular causes was reduced (16.5% vs. 13.3%; HR = 0.80; P < .001; NNT = 31), as was the overall death rate (19.8% vs. 17.0%; HR = 0.84; P < .001; NNT = 36). The sacubitril/valsartan combination, the only product of its kind, is now being referred to as an ARB/neprilysin inhibitor.
The estimated yearly cost for sacubitril/valsartan is $4,600.30 The associated cost-effectiveness ratio of $51,000 per quality-adjusted life-year is at the upper range of what is generally considered cost-effective.30 Another U.S. analysis found an incremental cost-effectiveness ratio of $45,000 per quality-adjusted life-year compared with enalapril.31 A theoretical concern is that inhibition of neprilysin might increase β-amyloid peptides associated with Alzheimer disease, but this effect has not yet been shown to be clinically relevant.32
In July 2015, the U.S. Food and Drug Administration approved the use of sacubitril/valsartan in patients with chronic heart failure and reduced ejection fraction to reduce the risk of cardiovascular death and hospitalization for heart failure.33 However, it has been tested in only one study and there has been limited postmarketing surveillance for potential deleterious effects. Based on limited high-quality evidence, ACC/AHA/Heart Failure Society of America (HFSA) guidelines recommend ARB/neprilysin inhibitor therapy as a first-line alternative to ACE inhibitors in patients with symptomatic heart failure who are not hypotensive or intolerant of angiotensin system antagonists.34 The American Academy of Family Physicians did not endorse this guideline because of concerns about its methodology and insufficient evaluation of harms.
BETA BLOCKER
Beta blockers, such as bisoprolol (Zebeta), carvedilol (Coreg), and metoprolol succinate (Toprol XL), are effective at reducing mortality in patients with symptomatic heart failure when combined with ACE inhibitors.5–7 Beta blockers have not been tested in those with NYHA class I (asymptomatic) heart failure. However, because heart failure with reduced ejection fraction is often caused by ischemic heart disease, many patients with NYHA class I heart failure might also benefit from beta blockers. Beta blockers work by blunting the noradrenergic hormonal influences and have long-term benefits. They are not rescue agents, and are best started when patients are stable and have no symptoms at rest. Dosing is important when using beta blockers for heart failure; dosages below the target do not appear to have the same impact.
ALDOSTERONE ANTAGONIST
Eplerenone (Inspra) and spironolactone have been proven beneficial in patients with heart failure when added to ACE inhibitor and beta-blocker therapy.8–10 Eplerenone or spironolactone is indicated in all symptomatic patients with heart failure due to reduced ejection fraction, and three to 14 days post–myocardial infarction in patients with reduced ejection fraction and symptomatic heart failure or concomitant diabetes mellitus.
ARB
ARBs have shown benefit as a substitute in those who cannot take ACE inhibitors.4 They should not be used in patients taking ACE inhibitors.
DIRECT-ACTING VASODILATORS
The direct-acting vasodilator isosorbide dinitrate/hydralazine is an option for those intolerant of ACE inhibitors and ARBs because of renal disease, and is indicated as adjunctive therapy in black patients who remain symptomatic despite treatment with ACE inhibitors. The African-American Heart Failure Trial showed that isosorbide dinitrate/hydralazine added to ACE inhibitor therapy provides incremental mortality benefit in black patients.12 Retrospective evaluations of prior trials suggest that nonblack patients have greater benefit from ACE inhibitors, but black patients have greater benefit from isosorbide dinitrate/hydralazine.35,36 A substudy of the African-American Heart Failure Trial showed that a genetic polymorphism that is more common among blacks is responsible for the difference in outcomes.37 Isosorbide dinitrate/hydralazine is taken three times daily, making compliance a challenge.
DIGOXIN
Digoxin should be considered for those who remain symptomatic despite therapy with all other disease-modifying agents. It works as a positive inotrope but can also be arrhythmogenic. In the only large controlled trial of digoxin in patients with heart failure, digoxin had no impact on mortality but decreased hospitalization rates.20
SINUS NODE MODULATOR
The SHIFT trial was a double-blind randomized controlled trial of ivabradine (Corlanor) vs. placebo that included patients with reduced ejection fraction who had been admitted to the hospital for heart failure within the 12 months before enrollment; were in sinus rhythm; and were taking some combination of an ACE inhibitor (79%), ARB (14%), beta blocker (90%), diuretic (84%), aldosterone antagonist (60%), and glycoside (digoxin, digitoxin [no longer available in the United States]; 22%). In the study, 6,558 patients were followed for a median of 22.9 months, and ivabradine was titrated to a maximum dosage of 7.5 mg twice daily.14 The primary end point was cardiovascular death or hospitalization for decompensated heart failure. Patients in the treatment arm had an 18% lower risk of reaching the primary end point compared with those in the placebo arm (24% vs. 29%; HR = 0.82; P < .0001; NNT = 26), primarily because of a reduction in admissions for heart failure. Further analysis demonstrated that the improved clinical outcomes occurred only in patients with a heart rate of more than 70 beats per minute. Ivabradine provided no benefit in patients with a lower heart rate. The U.S. Food and Drug Administration approved ivabradine for heart failure management in 2015.38
In contrast to the SHIFT study, ivabradine was not effective in a previous trial of patients with reduced ejection fraction and ischemic heart disease, not all of whom had a prior hospitalization for heart failure.39 According to ACC/AHA/HFSA guidelines, ivabradine may be considered in appropriate patients, but the beta blocker should first be titrated to the target dosage if tolerated.34
DIURETICS
Diuretic therapy (loop and thiazide) is essential when needed to manage volume status in patients with heart failure. Diuretics do not provide mortality benefit, so they should be used only when needed to treat congestion.
STATINS
The role of statins in the management of patients with heart failure is unproven. Two large trials failed to demonstrate any benefit from statins among patients with heart failure and reduced ejection fraction, even if the heart failure was due to ischemic heart disease.40,41 The 2013 ACC/AHA guidelines do not recommend statin therapy solely for the treatment of heart failure.21
Device Therapy
Implantable cardioverter-defibrillators improve mortality, and cardiac resynchronization therapy (biventricular pacemakers with or without an automatic implantable cardioverter-defibrillator) can improve symptoms among patients with heart failure.22–24,42 The determination of patient eligibility for device therapy can be complicated and is driven in part by Medicare coverage criteria. Any patient with reduced ejection fraction and symptomatic heart failure or ischemic cardiomyopathy whose life expectancy is more than one year may be referred to electrophysiology for possible device therapy. 22–24,42
Disease Management
Disease management programs and telemonitoring can reduce hospitalizations and mortality, especially in patients who were previously hospitalized for heart failure.25,26 Key components include heart failure education emphasizing self-care and adherence to medications, face-to-face contact after hospital discharge, mechanisms for postdischarge medication adjustment, and patient hotlines or other ways of quickly reaching clinicians.43
This article updates a previous article on this topic by Chavey, Bleske, Van Harrison, Hogikyan, Kesterson, and Nicklas.44
Data Sources: A literature review was generated from Essential Evidence Plus, which included relevant POEMs, Cochrane reviews, diagnostic test data, and a PubMed search for the best available evidence. Search date: August 12, 2015.
The authors thank Ellen Patrick for her assistance with the preparation of the manuscript.

