Pancreatic cancer is relatively uncommon and carries a poor prognosis because patients often develop signs or symptoms at a late stage of illness. Patients with a family history, especially those with genetic syndromes, are at a significantly increased risk of pancreatic cancer. Modifiable risk factors include smoking, heavy alcohol use, and obesity. Although patients at increased risk should be screened, screening is not recommended for asymptomatic people at average risk. The differential diagnosis for a symptomatic patient is broad, including gastroesophageal reflux disease, gastritis, peptic ulcer disease, chronic pancreatitis, biliary dyskinesia, cholelithiasis, gastroparesis, or constipation. Initial serologic testing should include transaminase and bilirubin levels, and in patients with midepigastric pain, lipase levels. Pancreas-protocol, contrast-enhanced abdominal computed tomography is the imaging test of choice. Carbohydrate antigen 19-9 is the most studied cancer marker and moderately accurate in patients suspected of having cancer; however, the positive predictive value is 0.9% in asymptomatic patients. Treatment includes neoadjuvant or adjuvant chemotherapy and surgery if the cancer is resectable. The treatment approach is best determined by a multidisciplinary, high-volume center. For a patient undergoing chemotherapy, nutritional and psychosocial support and palliation of symptoms should be goals during treatment.
Pancreatic cancer accounts for 3% of all cancer diagnoses and 7% of cancer-related deaths in the United States; pancreatic ductal adenocarcinoma makes up 95% of pancreatic cancers.1 This article provides a summary and review of the best available patient-oriented evidence for pancreatic cancer.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
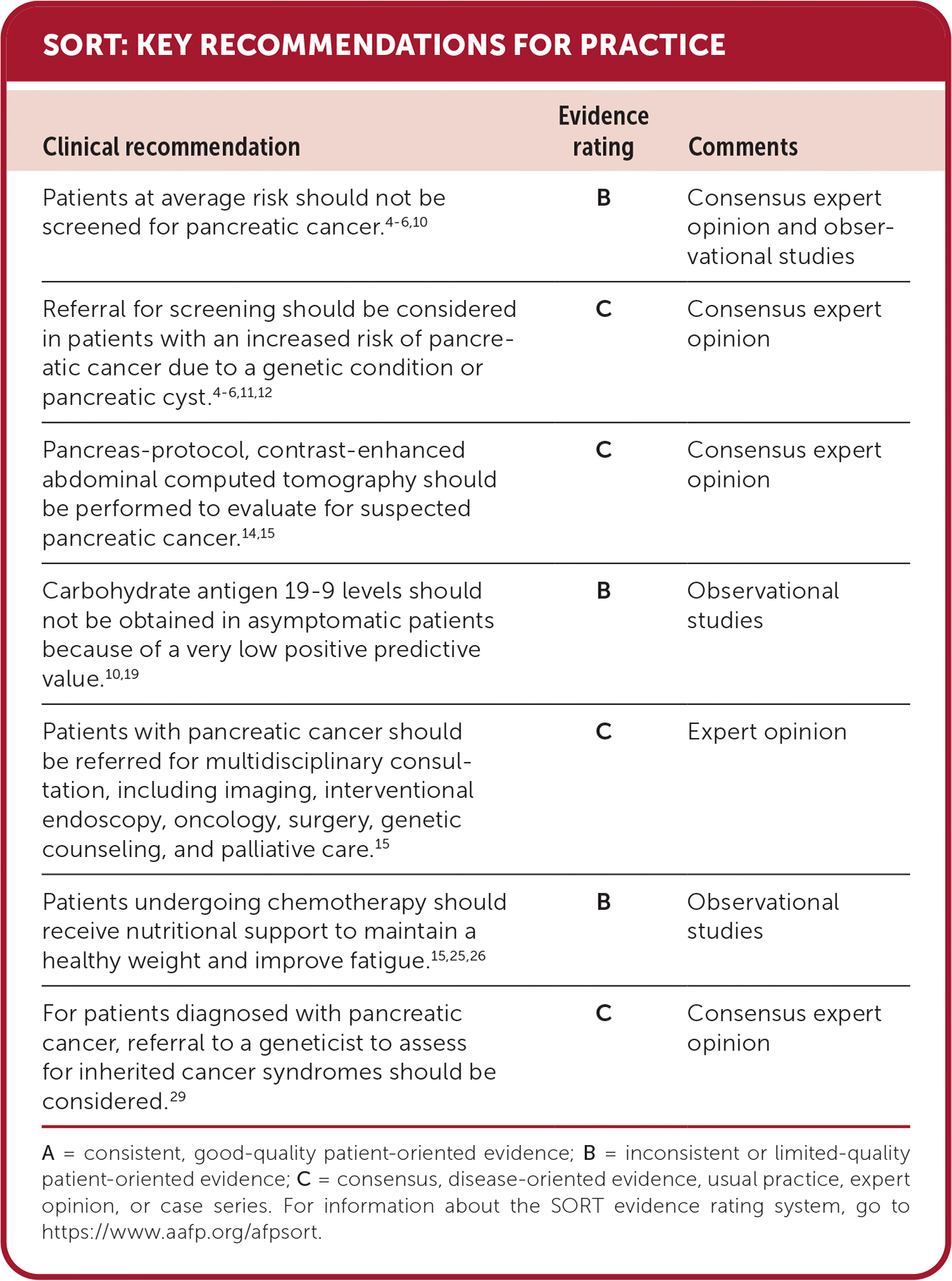
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Epidemiology
- In 2023, approximately 64,050 people were diagnosed with pancreatic cancer in the United States.1 The lifetime risk of developing pancreatic cancer is 1.7%.2
- Increasing age is a risk factor for pancreatic cancer, with a median age of 70 years at diagnosis; 90% of patients are diagnosed at 55 years or older.3
- Approximately 85% of pancreatic cancers are sporadic, 10% are familial, and 5% are due to an identifiable genetic syndrome.4
- Modifiable and nonmodifiable risk factors are summarized in Table 1.3–9
TABLE 1. Risk Factors for Pancreatic Cancer
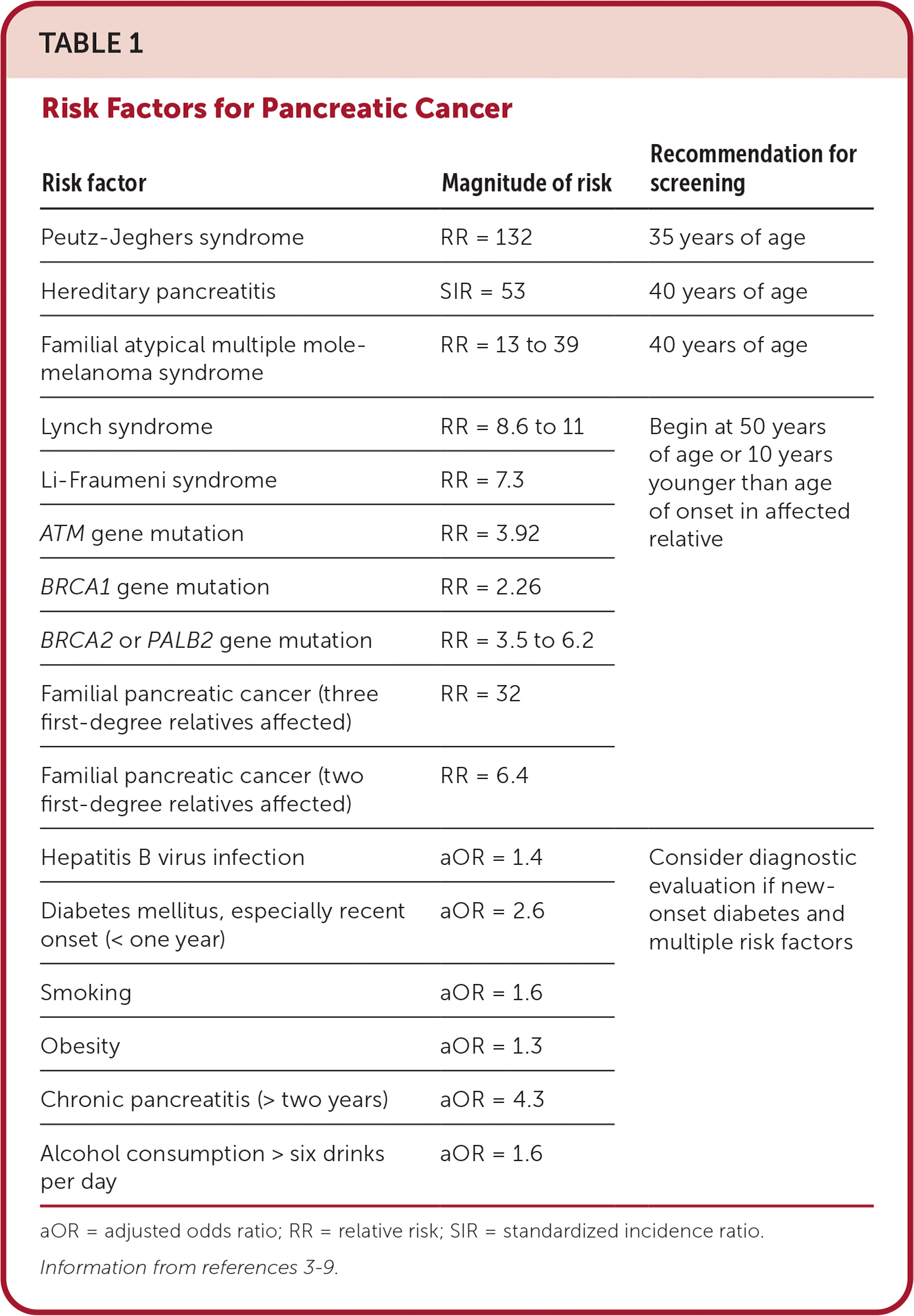
| Risk factor | Magnitude of risk | Recommendation for screening |
|---|---|---|
| Peutz-Jeghers syndrome | RR = 132 | 35 years of age |
| Hereditary pancreatitis | SIR = 53 | 40 years of age |
| Familial atypical multiple mole-melanoma syndrome | RR = 13 to 39 | 40 years of age |
| Lynch syndrome | RR = 8.6 to 11 | Begin at 50 years of age or 10 years younger than age of onset in affected relative |
| Li-Fraumeni syndrome | RR = 7.3 | |
| ATM gene mutation | RR = 3.92 | |
| BRCA1 gene mutation | RR = 2.26 | |
| BRCA2 or PALB2 gene mutation | RR = 3.5 to 6.2 | |
| Familial pancreatic cancer (three first-degree relatives affected) | RR = 32 | |
| Familial pancreatic cancer (two first-degree relatives affected) | RR = 6.4 | |
| Hepatitis B virus infection | aOR = 1.4 | Consider diagnostic evaluation if new-onset diabetes and multiple risk factors |
| Diabetes mellitus, especially recent onset (< one year) | aOR = 2.6 | |
| Smoking | aOR = 1.6 | |
| Obesity | aOR = 1.3 | |
| Chronic pancreatitis (> two years) | aOR = 4.3 | |
| Alcohol consumption > six drinks per day | aOR = 1.6 |
aOR = adjusted odds ratio; RR = relative risk; SIR = standardized incidence ratio.
Screening and Prevention
- The U.S. Preventive Services Task Force and multiple specialty societies recommend that asymptomatic adults at average risk for pancreatic cancer should not be screened.4–6,10
- Specialty guidelines recommend screening patients with a genetic syndrome and an increased risk of pancreatic cancer and those at high familial risk.4–6,11
- Screening for pancreatic cancer in patients at increased risk, including those with a pancreatic cyst, is complex and should be performed at an expert center.4–6,12
- Guidelines from the American Society for Gastrointestinal Endoscopy recommend that patients with a high-risk genetic syndrome be screened annually with endoscopic ultrasonography, contrast-enhanced 1.5 Tesla magnetic resonance imaging (MRI), or alternating endoscopic ultra-sonography and MRI.6
- Uncontrolled studies of high-risk patients with screen-detected cancers demonstrate higher three-year survival rates than patients with symptomatic cancers (57% vs. 8.9%), although this may be because of lead-time bias. There is no evidence from randomized trials that screening high-risk individuals reduces mortality.11
- Blood tests, such as the Galleri and the IMMRay PanCan-d tests, lack prospective, independent validation and are not recommended to screen for pancreatic cancer.13
Diagnosis
- The differential diagnosis of pancreatic cancer includes gastroesophageal reflux disease, gastritis, peptic ulcer disease, chronic pancreatitis, biliary dyskinesia, cholelithiasis, gastroparesis, or constipation.
- Initial serologic testing typically includes transaminase and bilirubin levels, and in patients with midepigastric pain, lipase levels.
SIGNS AND SYMPTOMS
- Signs and symptoms indicating advanced disease may include unintended weight loss, abdominal discomfort with radiation to the back, nausea, or vomiting.1
- Biliary obstruction may also occur, causing jaundice, pale stools, or dark urine.
- New or worsening diabetes mellitus may be a sign of pancreatic cancer.
DIAGNOSTIC TESTING
- The choice of diagnostic test depends on clinical suspicion for pancreatic cancer and the need for therapeutic intervention. Figure 1 summarizes one approach for the diagnosis of suspected pancreatic cancer.14
- Pancreas-protocol, contrast-enhanced abdominal computed tomography is the imaging test of choice for the evaluation of pancreatic cancer, and it is necessary if transabdominal ultrasonography is nondiagnostic.14,15
- Transabdominal ultrasonography is an acceptable initial imaging test, especially for the evaluation of jaundice, but it is operator dependent and may not be sensitive for small pancreatic lesions (less than 3 cm).16
- MRI with magnetic resonance cholangiopancreatography is 84% sensitive and 97% specific for cystic lesions or detecting ductal narrowing due to gallstones or mass effect and visualization of the entire pancreas.15,16
- Endoscopic retrograde cholangiopancreatography is preferred in patients with obstructive cholestasis in need of stenting, although it is more expensive than ultrasonography or computed tomography and carries risks of pancreatitis, bleeding, and cholangitis.16
- Endoscopic ultrasonography is one of the most accurate methods for detecting pancreatic cancer, especially tumors less than 3 cm or cystic lesions.6,12
- Fine-needle aspiration can be guided by endoscopic ultrasonography and provides a tissue diagnosis with less risk of complication than endoscopic retrograde cholangiopancreatography.12
- Carbohydrate antigen 19-9 levels greater than 37.0 U per mL are 72% sensitive (95% CI, 71% to 73%) and 86% specific (95% CI, 85% to 86%) for the diagnosis of pancreatic cancer in symptomatic patients.17 The positive and negative likelihood ratios are 5.1 and 0.32, respectively, suggesting only moderate accuracy.
- Carbohydrate antigen 19-9 levels may be elevated by noncancerous pancreatic or bilious diseases, and 10% of the population cannot synthesize the antigen, which produces a false-negative test result.18
- In a series of 71,040 asymptomatic patients, 1,063 patients (1.5%) had elevated carbohydrate antigen 19-9 levels and four of those had pancreatic cancer, yielding a positive predictive value of 0.9%.19 Therefore, obtaining carbohydrate antigen 19-9 levels in asymptomatic patients is not recommended.10
FIGURE 1.
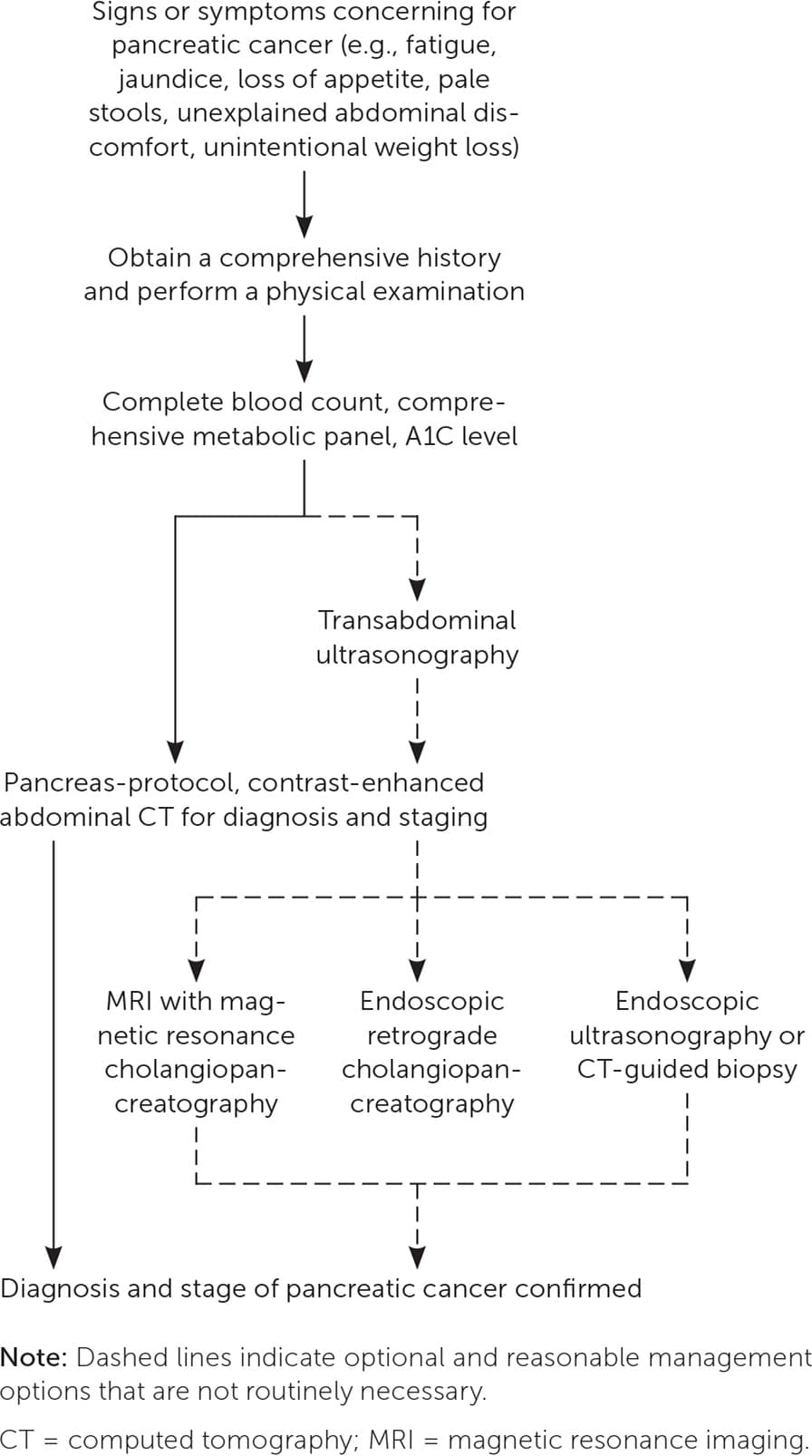
Algorithm for the diagnosis of suspected pancreatic cancer.
Adapted with permission from Kanji ZS, Gallinger S. Diagnosis and management of pancreatic cancer. CMAJ. 2013;185(14):1222.
Treatment
- Treatment depends on the stage of pancreatic cancer at the time of diagnosis, which identifies patients for whom surgery and curative intent are possible (i.e., locally resectable and no metastases).15
- Decisions about resectability and treatment approach should involve multidisciplinary consultation at high-volume centers.15
- Palliative care consultation was associated with less aggressive care at the end of life. Patients received less chemotherapy within 14 days of death and had fewer intensive care and hospital admissions and emergency department visits.20
DRUG THERAPY
- Systemic chemotherapy is used in all stages of pancreatic cancer and may be given before surgery (neoadjuvant) or after surgery (adjuvant) in patients with resectable disease.15
- Consider referring patients to participate in a clinical trial.15
- Specific chemotherapy regimens depend on the stage of pancreatic cancer and performance status of the patient. Folfirinox (fluorouracil/leucovorin/oxaliplatin/irinotecan) is preferred.15
- Gemcitabine-based monotherapy or combination regimens are also first-line, especially in patients with poor performance status.15
- Common adverse effects include neutropenia (46%), diarrhea (13%), thrombocytopenia (9%), and sensory neuropathy (9%), although these can be mitigated with a modified regimen.21
- Nutritional and psychosocial support and palliation of symptoms should be goals for patients receiving chemotherapy.15
SURGERY
- Up-front surgical resection (before adjuvant therapy) may be appropriate for patients with few comorbidities, good performance status, and no vascular involvement of the tumor; however, fewer than 20% of patients are surgical candidates.1
- Routine preoperative biliary drainage for patients with obstructive jaundice increases morbidity with no effect on mortality or length of hospitalization.22
RADIATION
- The decision to include radiation therapy should be individualized and based on goals of treatment; randomized trials and meta-analyses do not demonstrate a survival benefit when added to standard treatments.15
- National Comprehensive Cancer Network guidelines suggest that radiation therapy be included in the treatment plan depending on the stage of disease. It can be used for palliation of symptoms, as an adjunct to chemotherapy, and for the treatment of patients who are not candidates for systemic chemotherapy.15
- Radiation therapy should not be used in patients with metastatic disease that is progressing, unless it is used for palliative purposes.15
COMPLEMENTARY MEDICINE
- Antioxidants do not protect against gastrointestinal cancers and increase mortality.23
- In patients with advanced pancreatic cancer, mistletoe extract, given subcutaneously three times weekly, improved outcomes, including pain, fatigue, appetite loss, and insomnia, on six functional scales and seven of nine symptom scales.24 The study was not masked; therefore, patients may have experienced a placebo effect associated with repeated injections.
LIFESTYLE AND BEHAVIORAL INTERVENTIONS
- Dietary habits may impact chemotherapeutic metabolism; cytochrome P450 inhibitors such as grapefruit, oranges, pomegranates, cabbage, and onions should be avoided.25
- Improvement in modifiable risk factors (e.g., obesity, smoking, alcohol use, physical inactivity) may reduce the risk of pancreatic cancer.
- Oral nutritional supplementation during chemotherapy can mitigate weight loss and improve fatigue.15,25,26
REFERRAL, CONSULTATION, AND HOSPITALIZATION
- Patients with pancreatic cancer will require a multidisciplinary team, which has been shown to decrease socioeconomic disparities in treatment and improve survival.27
- Referral for a celiac plexus block can reduce the use of opioids and improve pain control.28
- Consider referring patients with pancreatic cancer to a geneticist to assess for inherited cancer syndromes.29
Prognosis
- For all stages combined, the five-year relative survival rate is 11%1,2 (Table 22,15 ).
- Diagnosis at earlier stages is associated with greater survival; the 10-year survival rate for stage 0 pancreatic cancer is 93%, and the five-year survival rate for stage 1A cancers is 38%.1
- Lymph node or distant metastasis is the most important prognostic factor; the five-year survival rate is 44% for local disease, 16% for regional disease, and 3% for meta-static disease.2
- Only 12% of patients will be staged with localized disease at the time of diagnosis.2
- Posttreatment normalization or a decrease of greater than 50% in carbohydrate antigen 19-9 levels is associated with a favorable prognosis, whereas an increasing level is concerning for disease progression.15,30
TABLE 2. Staging of Pancreatic Cancer
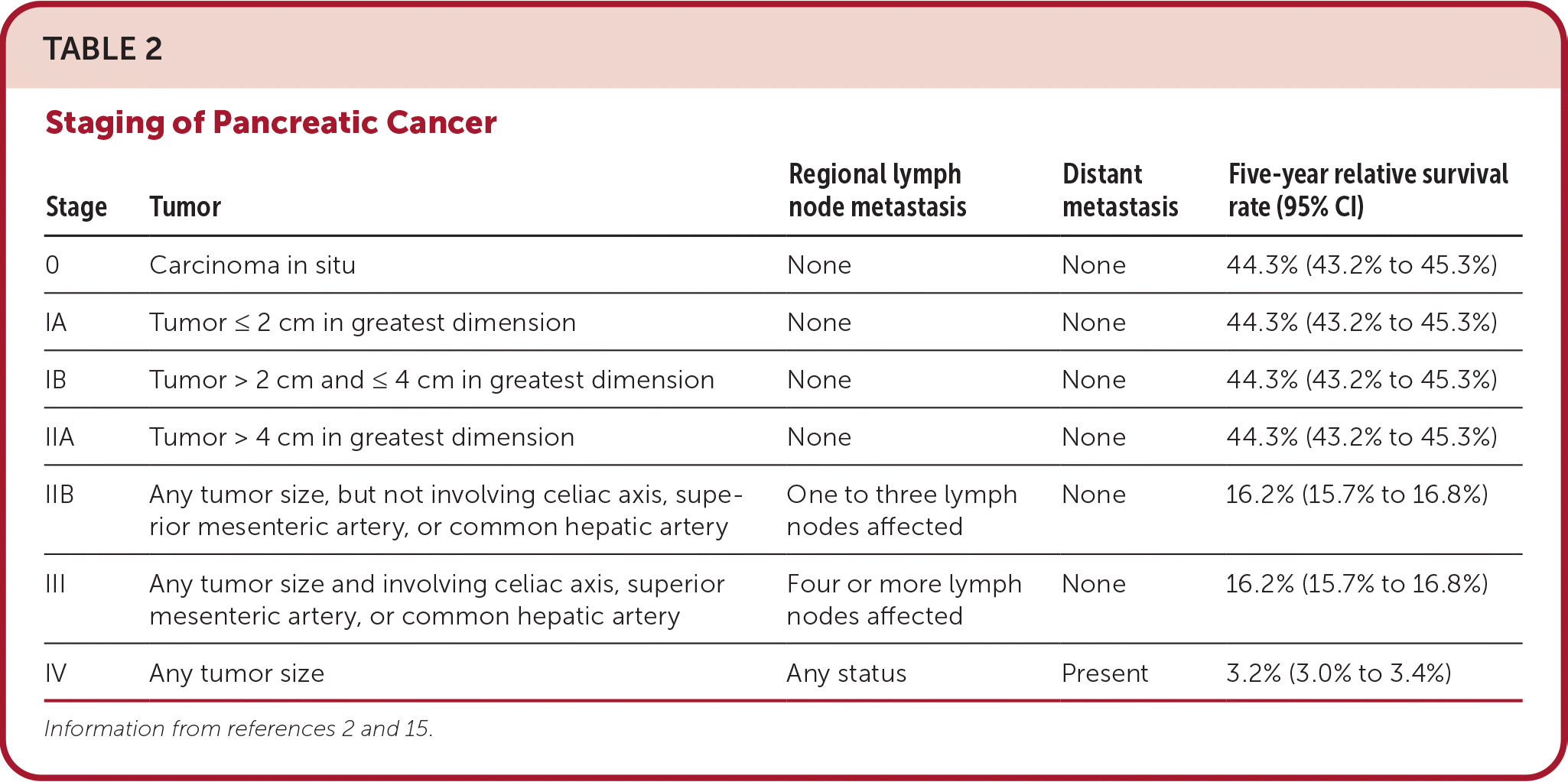
| Stage | Tumor | Regional lymph node metastasis | Distant metastasis | Five-year relative survival rate (95% CI) |
|---|---|---|---|---|
| 0 | Carcinoma in situ | None | None | 44.3% (43.2% to 45.3%) |
| IA | Tumor ≤ 2 cm in greatest dimension | None | None | 44.3% (43.2% to 45.3%) |
| IB | Tumor > 2 cm and ≤ 4 cm in greatest dimension | None | None | 44.3% (43.2% to 45.3%) |
| IIA | Tumor > 4 cm in greatest dimension | None | None | 44.3% (43.2% to 45.3%) |
| IIB | Any tumor size, but not involving celiac axis, superior mesenteric artery, or common hepatic artery | One to three lymph nodes affected | None | 16.2% (15.7% to 16.8%) |
| III | Any tumor size and involving celiac axis, superior mesenteric artery, or common hepatic artery | Four or more lymph nodes affected | None | 16.2% (15.7% to 16.8%) |
| IV | Any tumor size | Any status | Present | 3.2% (3.0% to 3.4%) |
This article updates previous articles on this topic by De La Cruz, et al.,31 and Freelove and Walling.32
Data Sources: A literature search was conducted in PubMed using the term pancreatic cancer and filtering results by systematic review, meta-analysis, and randomized controlled trial. The search was repeated using the MeSH term pancreatic neoplasms. Articles published in the past five years were prioritized for relevancy. This article was based on literature cited in Essential Evidence Plus, the Cochrane database, recently published InfoPOEMs, and a PubMed search using the Clinical Queries database for the term pancreatic adenocarcinoma. Search dates: August 2022, and April, September, and December 2023.

