Key Points for Practice
• Risk stratification tools, such as the Oakland score, have some evidence of helping determine which patients with lower GI bleeding do not need hospitalization, but clinical judgment is still needed.
• For hemodynamically significant lower GI bleeding, CT angiography can accurately locate the bleeding source.
• For hemodynamically stable patients, with or without continued lower GI bleeding, performing colonoscopy within 14 days of presentation is as effective as performing it within 24 hours.
• Transcatheter embolization within 90 minutes of positive CT angiography results can stop bleeding 98% of the time, although 30-day mortality is 13%.
From the AFP Editors
Acute lower gastrointestinal (GI) bleeding, with hematochezia or bright red blood from the rectum, is cause for more than 100,000 hospital admissions annually. The American College of Gastroenterology (ACG) has published updated guidelines for the management of acute lower GI bleeding.
Risk Stratification
Risk stratification tools such as the Oakland score (https://www.mdcalc.com/calc/10042/oakland-score-safe-discharge-lower-gi-bleed) can be used to assist physicians in determining which patients may be suited for outpatient management. The Oakland score and other similar tools have demonstrated small-scale validity without randomized controlled trials or large-scale data verification. Physicians should primarily use clinical judgment when risk-stratifying patients with lower GI bleeding.
Evaluation
In patients with hemodynamically significant bleeding, computed tomography (CT) angiography can identify the source and associated vasculature with a sensitivity of approximately 90%. Nearly 80% of patients with negative CT angiography results will have no rebleeding and require no further evaluation.
Colonoscopy is the standard for diagnostic testing in hemodynamically stable patients with lower GI bleeding. In a European cohort, colonoscopy led to a definitive or presumptive source in 79% of patients. For hemodynamically stable patients without continued bleeding, moderate-quality evidence supports completing a colonoscopy within 14 days of spontaneous hemostasis. For these patients, performing a colonoscopy in the first 24 hours after presentation does not improve outcomes. No bowel preparation appears superior to any other for colonoscopy after lower GI bleeding.
Patients who have had a colonoscopy within the past 12 months may not need repeat testing if bleeding has subsided and a suspected bleeding source, such as diverticulosis, was identified.
Transfusion
Low-quality evidence supports a restrictive transfusion approach at a threshold hemoglobin level of less than 7 g per dL (70 g per L) in hemo-dynamically stable patients. In a retrospective analysis, restrictive vs. nonrestrictive transfusion strategies demonstrated similar mortality and rebleeding rates. A higher threshold of 8 g per dL (80 g per L) can be considered in patients with known cardiovascular disease or acute coronary syndrome.
Management
Transcatheter arteriography is nine times more likely to detect the bleeding source and enable embolization therapy when performed within 90 minutes of a positive angiogram compared with delayed arteriography. Embolization therapy has a 98% success rate, but it can cause an ulcer or bowel infarction in nearly 5% of patients. Even after embolization, 30-day rebleeding and mortality rates are 16% and 13%, respectively.
Despite higher colonoscopy treatment rates after a positive CT angiogram result, routine use of CT angiography before colonoscopy does not reduce mortality or rebleeding.
Anticoagulant Medications
Endoscopic procedures can generally be performed safely in patients with an international normalized ratio of 2.5 or less. Although the evidence is limited, use of reversal agents is discouraged unless the patient remains hemodynamically unstable despite resuscitation.
For severe lower GI bleeding, the target platelet level should be greater than 30 × 103 per μL (30 × 109 per L) and a threshold greater than 50 × 103 per μL (50 × 109 per L) is needed before endoscopy. Transfusing to higher platelet levels increases mortality without improving rebleeding risk.
Because rebleeding risk appears small, the ACG recommends that patients taking aspirin for secondary prevention of cardiac disease continue therapy. For dual antiplatelet therapy in patients with a recent cardiac stent, a multidisciplinary team should compare risks.
Antifibrinolytic agents such as tranexamic acid should be avoided in lower GI bleeding because the evidence shows increased rates of thromboembolism and seizure without any improvement in outcomes.
Following hospitalization for lower GI bleeding, the ACG recommends that patients discontinue aspirin for primary cardiovascular prevention and nonsteroidal anti-inflammatory drug treatment after diverticular bleeds. In contrast, aspirin for secondary cardiovascular prevention and anticoagulation taken following venous thromboembolism or ischemic stroke should be continued. For other anticoagulation therapy, a multidisciplinary team should compare risks.
G-TRUST SCORECARD
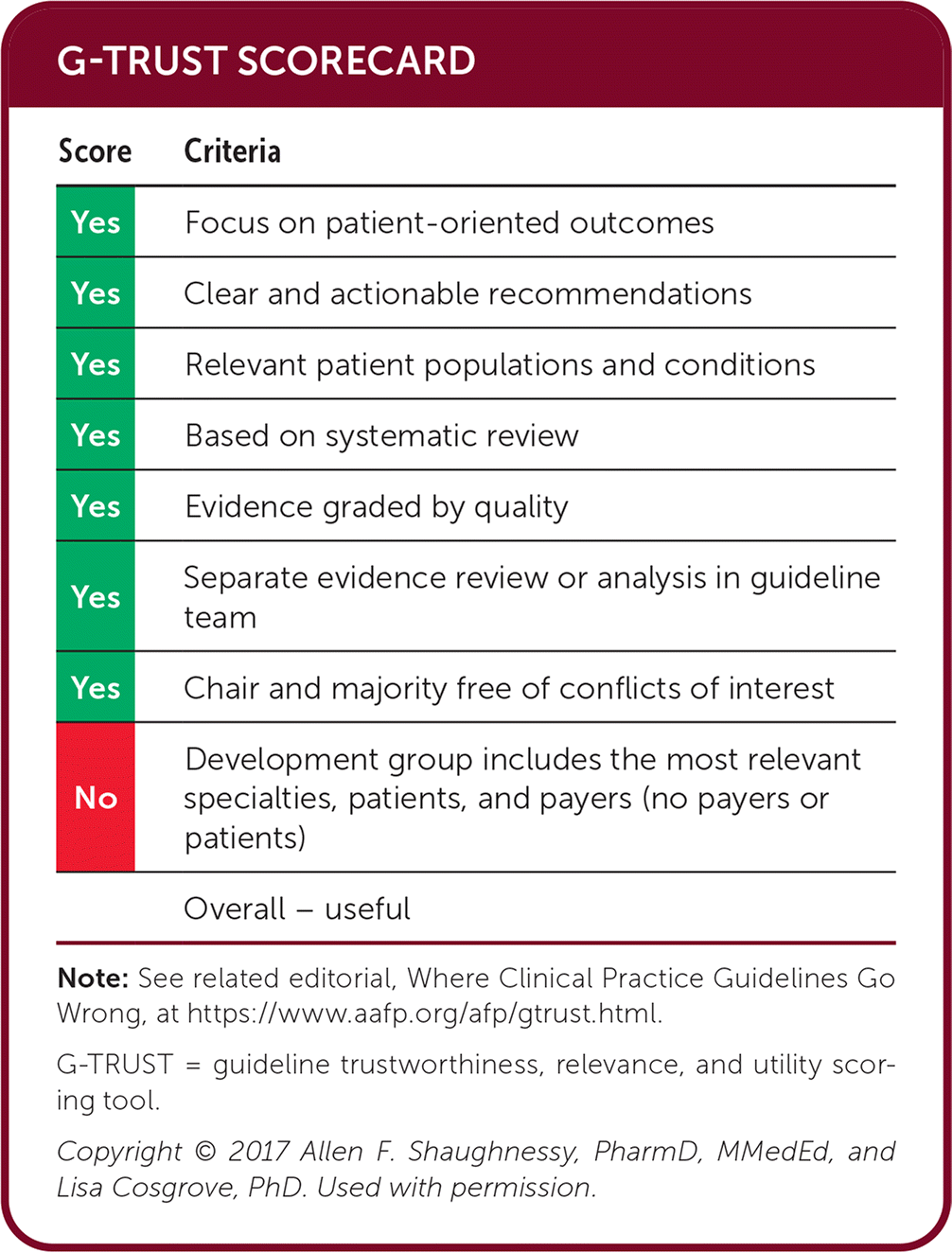
| Score | Criteria |
|---|---|
| Yes | Focus on patient-oriented outcomes |
| Yes | Clear and actionable recommendations |
| Yes | Relevant patient populations and conditions |
| Yes | Based on systematic review |
| Yes | Evidence graded by quality |
| Yes | Separate evidence review or analysis in guideline team |
| Yes | Chair and majority free of conflicts of interest |
| No | Development group includes the most relevant specialties, patients, and payers (no payers or patients) |
| Overall – useful |
Note: See related editorial, Where Clinical Practice Guidelines Go Wrong, at https://www.aafp.org/afp/gtrust.html.
G-TRUST = guideline trustworthiness, relevance, and utility scoring tool.
Copyright © 2017 Allen F. Shaughnessy, PharmD, MMedEd, and Lisa Cosgrove, PhD. Used with permission.
Guideline source: American College of Gastroenterology
Published source: Sengupta N, Feuerstein JD, Jairath V, et al. Management of patients with acute lower gastrointestinal bleeding: an updated ACG guideline. Am J Gastroenterol. 2023;118(2):208–231.

