Type 2 diabetes mellitus is a chronic disease that is increasing in global prevalence. An individualized approach to pharmacotherapy should consider costs, benefits beyond glucose control, and adverse events. Metformin is the first-line therapy due to its low cost and effectiveness. Sulfonylureas and thiazolidinediones are additional low-cost oral hypoglycemic classes available in the United States; however, evidence shows variability in weight gain and hypoglycemia. Thiazolidinediones increase fluid retention and are not recommended in patients with New York Heart Association class III or IV heart failure. Newer medications, including glucagon-like peptide-1 receptor agonists and sodium-glucose cotransporter-2 inhibitors, have demonstrated weight loss, reduced cardiovascular events, decreased renal disease, and improved all-cause morbidity and mortality. Sodium-glucose cotransporter-2 inhibitors are recommended for people with known cardiovascular disease, heart failure, and chronic kidney disease but carry an increased risk of urinary tract and mycotic infections. Glucagon-like peptide-1 receptor agonists are contraindicated in patients with active multiple endocrine neoplasia type 2 or a personal or family history of medullary thyroid carcinoma; adverse effects include gastrointestinal upset and pancreatitis. Dipeptidyl-peptidase-4 inhibitors have a low risk of hypoglycemia but may increase the risk of pancreatitis and require a renal dose adjustment. Public and private programs to increase access to newer hypoglycemic medications are increasing; however, there are limitations to access, particularly for uninsured and underinsured people.
More than 38 million people in the United States have diabetes mellitus; 90% to 95% have type 2.1 In 2022, medical and economic costs of $412.9 billion in the United States were attributed to diabetes.2 In the next 10 years, estimates predict a near doubling of the number of affected people in the United States, with a greater prevalence among low-income populations.3,4 People with annual incomes below the poverty level have the highest prevalence of a diabetes diagnosis and complication rates.1 Food insecurity, food deserts, and lack of access to safe exercise spaces in lower socioeconomic areas complicate efforts to prevent and control type 2 diabetes.5,6 Racial and ethnic disparities also exist. A total of 14.5% of American Indians and Alaska Natives, 12.1% of non-Hispanic Blacks, 11.7% of Hispanics, 9.1% of non-Hispanic Asians, and 6.9% of non-Hispanic Whites are diagnosed with diabetes; however, millions more people are undiagnosed.7–9
WHAT'S NEW ON THIS TOPIC
| In 2016, the U.S. Food and Drug Administration revised the metformin label to reflect its safety in people with an estimated glomerular filtration rate of 30 mL per minute per 1.73 m2 or greater. When the estimated glomerular filtration rate is between 30 and 45 mL per minute per 1.73 m2, a maximum daily dosage of 1,000 mg and close monitoring of renal function are recommended. |
| A large randomized clinical trial showed that empagli flozin (Jardiance), a sodium-glucose cotransporter-2 inhibitor, reduced a composite outcome of myocardial infarction, stroke, and cardiovascular death in people with established arteriosclerotic cardiovascular disease compared with placebo. |
| A 2023 systematic review found that glucagon-like peptide-1 receptor agonists and sodium-glucose cotransporter-2 inhibitors reduce cardiovascular-related deaths, nonfatal myocardial infarction, hospital admissions, end-stage renal disease, and all-cause mortality. |
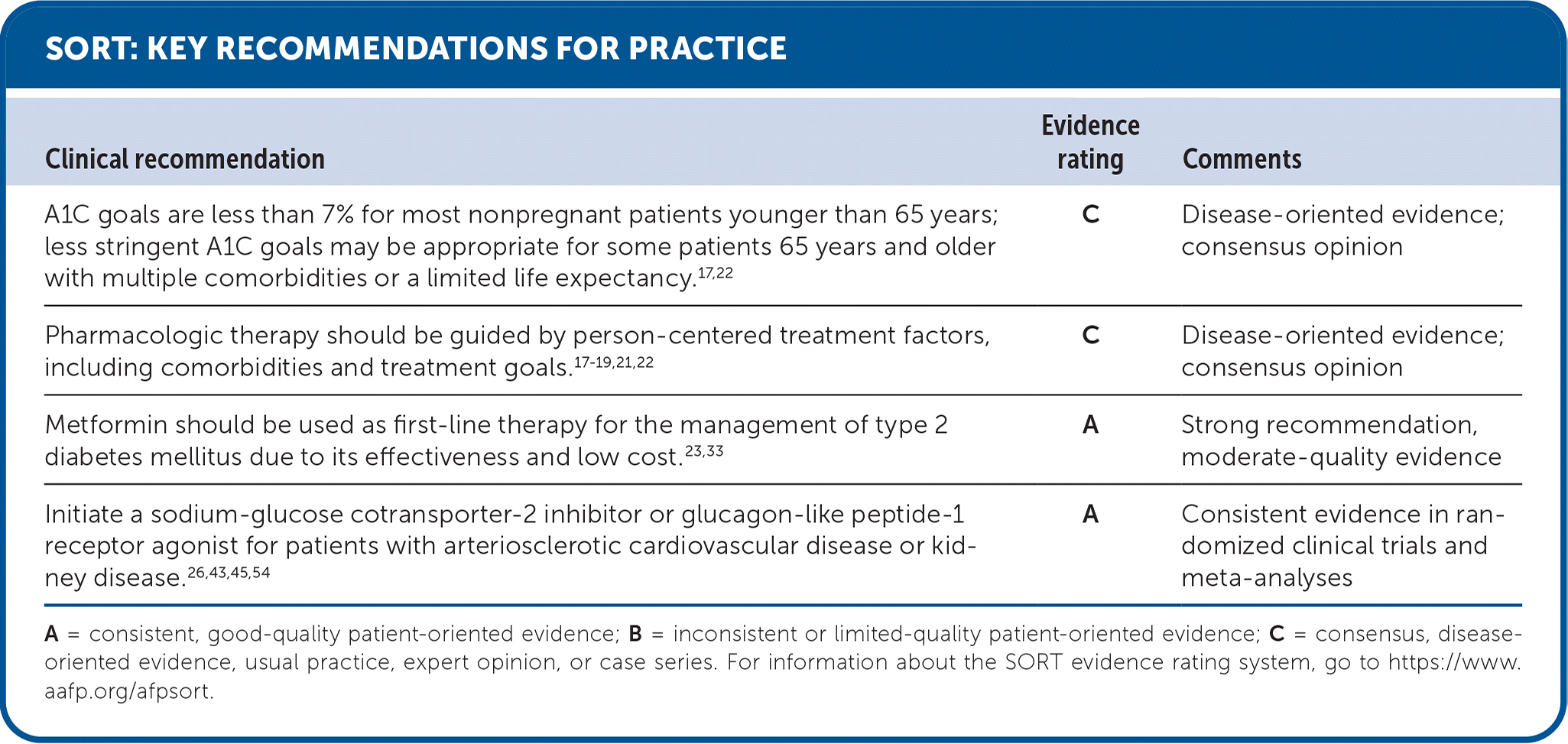
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | Comments |
|---|---|---|
| A1C goals are less than 7% for most nonpregnant patients younger than 65 years; less stringent A1C goals may be appropriate for some patients 65 years and older with multiple comorbidities or a limited life expectancy.17,22 | C | Disease-oriented evidence; consensus opinion |
| Pharmacologic therapy should be guided by person-centered treatment factors, including comorbidities and treatment goals.17–19,21,22 | C | Disease-oriented evidence; consensus opinion |
| Metformin should be used as first-line therapy for the management of type 2 diabetes mellitus due to its effectiveness and low cost.23,33 | A | Strong recommendation, moderate-quality evidence |
| Initiate a sodium-glucose cotransporter-2 inhibitor or glucagon-like peptide-1 receptor agonist for patients with arteriosclerotic cardiovascular disease or kidney disease.26,43,45,54 | A | Consistent evidence in randomized clinical trials and meta-analyses |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Social determinants of health (i.e., the conditions in which individuals are born, work, and live) are strongly associated with diabetes outcomes.7,10 Education, income, occupation, and environment affect the ability to prevent and control diabetes.7 To improve health equity, federal programs, including the Affordable Care Act and Medicaid, have expanded access to care for more people with diabetes. Patient assistance programs have also increased access to newer, more expensive medications.7,11
Prevention and Nonpharmacologic Management
The goal of diabetes care is to optimize quality of life by preventing macrovascular and microvascular complications, including cardiovascular, renal, neurologic, ophthalmic, and gastrointestinal disorders.12
Risk factors for developing diabetes include elevated visceral adiposity, polycystic ovary syndrome, fasting plasma glucose level of 100 mg per dL (5.55 mmol per L) or greater, A1C of 5.7% or greater, a family history of type 2 diabetes, poor dietary habits, and a history of gestational diabetes.13 Dietary modifications alone do not sustain A1C improvement. A meta-analysis of randomized controlled trials demonstrated that a low-carbohydrate diet was associated with significant A1C reduction at 3 months but not at 6 or 12 months and a body mass index reduction at 6 months but not at 12 months.14 Multifaceted programs such as the Diabetes Prevention Program have provided strong evidence for lifestyle modifications to prevent diabetes and control glucose levels for people with type 2 diabetes. Modifications include 150 minutes or more of moderate-intensity exercise or 75 minutes or more of vigorous-intensity exercise per week, weight loss of 7% or greater, and a reduction in dietary fat and caloric intake.15
Patient-Centered Glycemic Control
An A1C is the average blood glucose level over 3 months measured by the percentage of glycosylated red blood cells. Anemia, end-stage renal disease, alcoholism, and certain hemoglobinopathies can affect the accuracy of an A1C.16 The American Diabetes Association recommends a target A1C of less than 7% for healthy adults younger than 65 years and less than 7.5% for healthy adults 65 years and older with intact cognition and functional status.17 Less stringent A1C goals below 8% may be more appropriate for some patients. Several clinical trials found that severe hypoglycemia is a marker for a high absolute risk of cardiovascular events and mortality.17–20 Individualized therapy is based on factors including the risk of hypoglycemia, drug-drug interactions, adverse effects, disease duration, life expectancy, comorbidities, established vascular complications, patient preference and resources, and the patient's support system.17–19,21,22 In some patients, continuous glucose monitoring may improve glycemic control and reduce hypoglycemia by providing real-time data on blood glucose levels.20
Pharmacologic Management
Eight classes of hypoglycemic medications (Table 123,24 and Table 221,25–29 ) are discussed in this review. Figure 1 provides an approach to prescribing type 2 diabetes medications based on patient risk factors.12,23,26,28,30 Unless contraindicated, metformin is the first-line therapy.23 Accessibility is the next consideration. For example, if a patient has an indication for a newer medication (e.g., sodium-glucose cotransporter-2 [SGLT-2] inhibitors: microalbuminuria, chronic kidney disease, coronary artery disease; glucagon-like peptide-1 [GLP-1] receptor agonists: coronary artery disease), it is critical to ensure that the patient can obtain the medication through insurance or be assisted in applying for a program to cover the cost.17,31,32 Otherwise, lower-cost medications should be considered. Treatment regimens should support weight management goals.23 If noninsulin therapy is maximized or exhausted and glycemic control is not achieved, insulin should be considered.3
TABLE 2. Characteristics of Hypoglycemic Medications by Drug Class
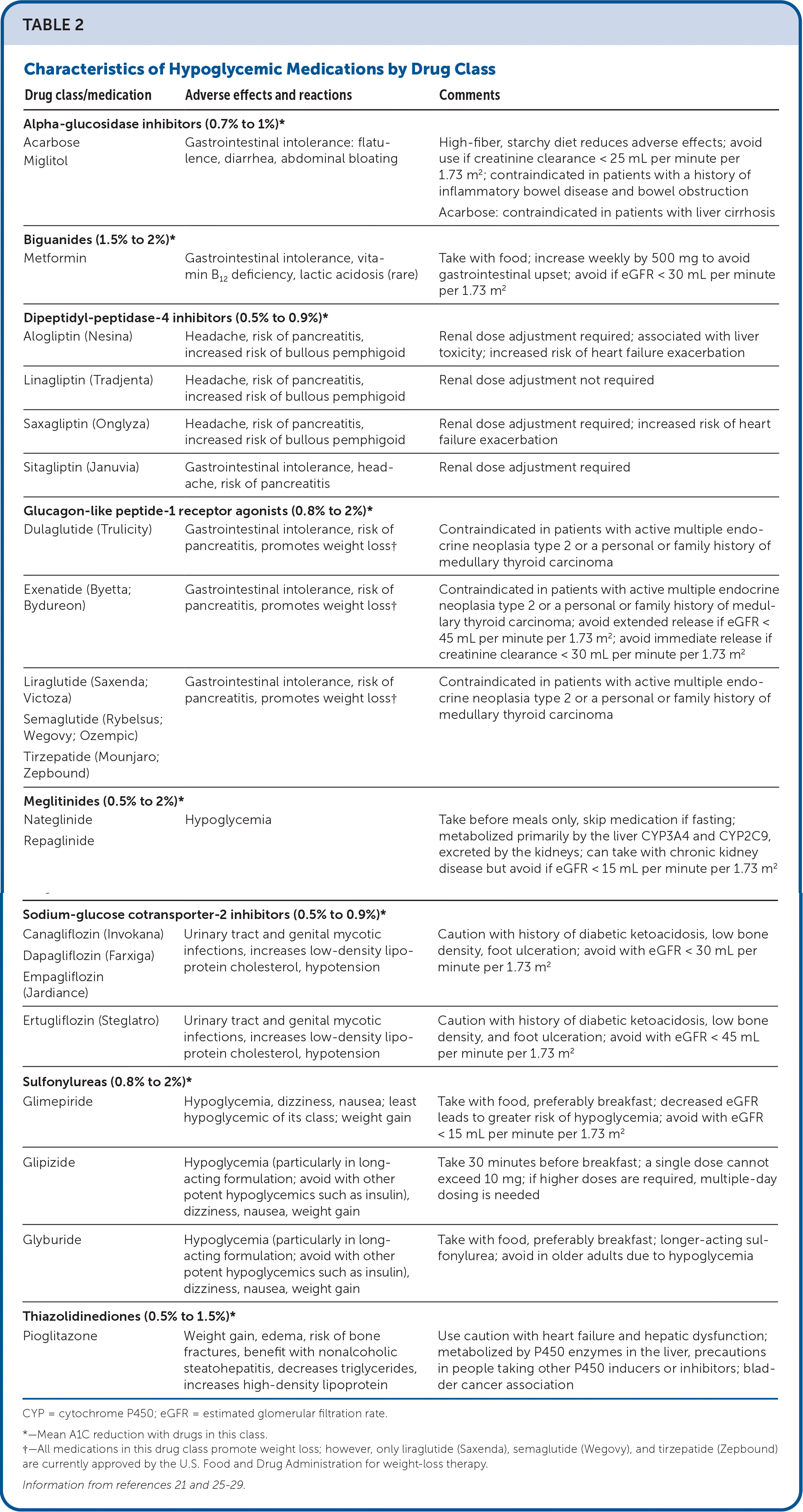
| Drug class/medication | Adverse effects and reactions | Comments |
|---|---|---|
| Alpha-glucosidase inhibitors (0.7% to 1%)* | ||
| Acarbose | Gastrointestinal intolerance: flatulence, diarrhea, abdominal bloating | High-fiber, starchy diet reduces adverse effects; avoid use if creatinine clearance < 25 mL per minute per 1.73 m2; contraindicated in patients with a history of inflammatory bowel disease and bowel obstruction Acarbose: contraindicated in patients with liver cirrhosis |
| Miglitol | ||
| Biguanides (1.5% to 2%)* | ||
| Metformin | Gastrointestinal intolerance, vitamin B12 deficiency, lactic acidosis (rare) | Take with food; increase weekly by 500 mg to avoid gastrointestinal upset; avoid if eGFR < 30 mL per minute per 1.73 m2 |
| Dipeptidyl-peptidase-4 inhibitors (0.5% to 0.9%)* | ||
| Alogliptin (Nesina) | Headache, risk of pancreatitis, increased risk of bullous pemphigoid | Renal dose adjustment required; associated with liver toxicity; increased risk of heart failure exacerbation |
| Linagliptin (Tradjenta) | Headache, risk of pancreatitis, increased risk of bullous pemphigoid | Renal dose adjustment not required |
| Saxagliptin (Onglyza) | Headache, risk of pancreatitis, increased risk of bullous pemphigoid | Renal dose adjustment required; increased risk of heart failure exacerbation |
| Sitagliptin (Januvia) | Gastrointestinal intolerance, headache, risk of pancreatitis | Renal dose adjustment required |
| Glucagon-like peptide-1 receptor agonists (0.8% to 2%)* | ||
| Dulaglutide (Trulicity) | Gastrointestinal intolerance, risk of pancreatitis, promotes weight loss† | Contraindicated in patients with active multiple endocrine neoplasia type 2 or a personal or family history of medullary thyroid carcinoma |
| Exenatide (Byetta; Bydureon) | Gastrointestinal intolerance, risk of pancreatitis, promotes weight loss† | Contraindicated in patients with active multiple endocrine neoplasia type 2 or a personal or family history of medullary thyroid carcinoma; avoid extended release if eGFR < 45 mL per minute per 1.73 m2; avoid immediate release if creatinine clearance < 30 mL per minute per 1.73 m2 |
| Liraglutide (Saxenda; Victoza) Semaglutide (Rybelsus; Wegovy; Ozempic) Tirzepatide (Mounjaro; Zepbound) | Gastrointestinal intolerance, risk of pancreatitis, promotes weight loss† | Contraindicated in patients with active multiple endocrine neoplasia type 2 or a personal or family history of medullary thyroid carcinoma |
| Meglitinides (0.5% to 2%)* | ||
| Nateglinide Repaglinide | Hypoglycemia | Take before meals only, skip medication if fasting; metabolized primarily by the liver CYP3A4 and CYP2C9, excreted by the kidneys; can take with chronic kidney disease but avoid if eGFR < 15 mL per minute per 1.73 m2 |
| Sodium-glucose cotransporter-2 inhibitors (0.5% to 0.9%)* | ||
| Canagliflozin (Invokana) Dapagliflozin (Farxiga) Empagliflozin (Jardiance) | Urinary tract and genital mycotic infections, increases low-density lipoprotein cholesterol, hypotension | Caution with history of diabetic ketoacidosis, low bone density, foot ulceration; avoid with eGFR < 30 mL per minute per 1.73 m2 |
| Ertugliflozin (Steglatro) | Urinary tract and genital mycotic infections, increases low-density lipoprotein cholesterol, hypotension | Caution with history of diabetic ketoacidosis, low bone density, and foot ulceration; avoid with eGFR < 45 mL per minute per 1.73 m2 |
| Sulfonylureas (0.8% to 2%)* | ||
| Glimepiride | Hypoglycemia, dizziness, nausea; least hypoglycemic of its class; weight gain | Take with food, preferably breakfast; decreased eGFR leads to greater risk of hypoglycemia; avoid with eGFR < 15 mL per minute per 1.73 m2 |
| Glipizide | Hypoglycemia (particularly in long-acting formulation; avoid with other potent hypoglycemics such as insulin), dizziness, nausea, weight gain | Take 30 minutes before breakfast; a single dose cannot exceed 10 mg; if higher doses are required, multiple-day dosing is needed |
| Glyburide | Hypoglycemia (particularly in long-acting formulation; avoid with other potent hypoglycemics such as insulin), dizziness, nausea, weight gain | Take with food, preferably breakfast; longer-acting sulfonylurea; avoid in older adults due to hypoglycemia |
| Thiazolidinediones (0.5% to 1.5%)* | ||
| Pioglitazone | Weight gain, edema, risk of bone fractures, benefit with nonalcoholic steatohepatitis, decreases triglycerides, increases high-density lipoprotein | Use caution with heart failure and hepatic dysfunction; metabolized by P450 enzymes in the liver, precautions in people taking other P450 inducers or inhibitors; bladder cancer association |
CYP = cytochrome P450; eGFR = estimated glomerular filtration rate.
*—Mean A1C reduction with drugs in this class.
†—All medications in this drug class promote weight loss; however, only liraglutide (Saxenda), semaglutide (Wegovy), and tirzepatide (Zepbound) are currently approved by the U.S. Food and Drug Administration for weight-loss therapy.
FIGURE 1.
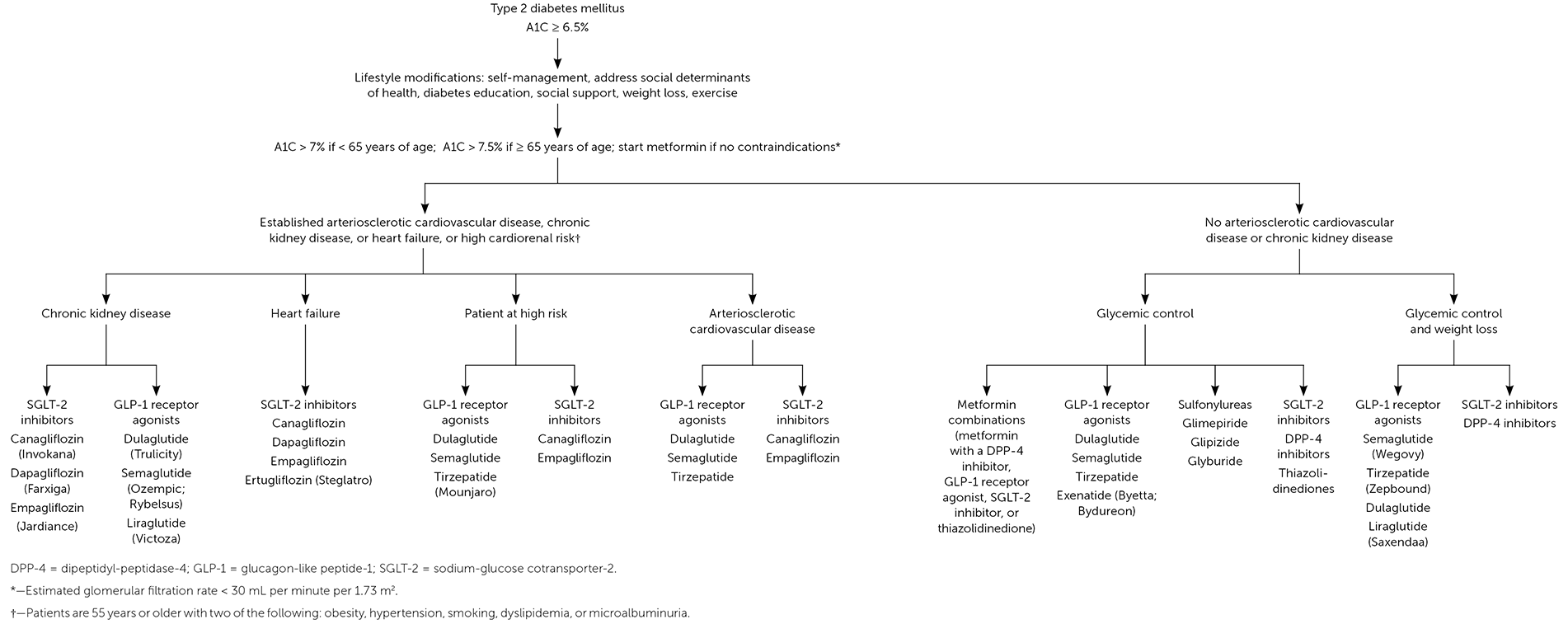
Approach to type 2 diabetes mellitus treatment stratified by risk factors.
ALPHA-GLUCOSIDASE INHIBITORS
The primary mechanism of action of alpha-glucosidase inhibitors is to inhibit the alpha-glucosidase enzyme, which is found in the small intestine cells brush border that catalyzes complex carbohydrates. This action reduces postprandial hyperglycemia, although patients must have concurrent food intake to receive its effects. Therapy should be initiated at the lowest effective dose and titrated slowly every two to four weeks.29 Alpha-glucosidase inhibitors are excreted renally and contraindicated in people with a serum creatinine of 2.0 mg per dL (176.8 μmol per L) or greater.29 The alpha-glucosidase inhibitor, acarbose, increases serum transaminase levels and should not be used in patients with liver cirrhosis.24
BIGUANIDES
Metformin is the preferred first-line oral blood glucose–lowering medication to manage type 2 diabetes.33 Metformin decreases hepatic glucose production and intestinal absorption of glucose to improve insulin sensitivity and is effective, safe, and inexpensive; data suggest that it decreases the risk of cardiovascular events and death.33–35 The principal adverse effect of metformin is gastrointestinal intolerance, including bloating, nausea, abdominal discomfort, and diarrhea, which can be reduced by slow titration and concurrent food intake. Patients with risk factors for lactic acidosis, including concurrent use of carbonic anhydrase inhibitors and those 65 years or older with recent use of iodinated contrast, undergoing surgery, or in a hypoxic state, should be monitored during treatment.33,36 Metformin is cleared by renal filtration; caution must be taken in the setting of chronic kidney disease. In 2016, the U.S. Food and Drug Administration (FDA) revised the metformin label to reflect its safety in people with an estimated glomerular filtration rate (eGFR) of 30 mL per minute per 1.73 m2 or greater. When the eGFR is between 30 and 45 mL per minute per 1.73 m2, a maximum daily dosage of 1,000 mg and close monitoring of renal function are recommended.33–35 Metformin is contraindicated in patients with an eGFR of less than 30 mL per minute per 1.73 m2 and acute or chronic metabolic acidosis. Metformin is also contraindicated for patients with hepatic impairment and unstable heart failure.33–35
DIPEPTIDYL-PEPTIDASE-4 INHIBITORS
Dipeptidyl-peptidase-4 (DPP-4) inhibitors block DPP-4, an enzyme that degrades incretin peptides GLP-1 and -2. DPP-4 inhibitors activate glucose-dependent insulinotropic polypeptides to stimulate beta cells to secrete insulin.37 They are weight-neutral and have a low risk of hypoglycemia. DPP-4 inhibitors are often combined with other hypoglycemics (e.g., metformin, thiazolidinediones), but use with GLP-1 receptor agonists does not provide additive glucose-lowering effects.24,38,39 An association between DPP-4 inhibitor use and bullous pemphigoid and other dermatoses has been observed; they have also been associated with pancreatitis in clinical trials, although causality has not been established.30,40 Unlike SGLT-2 inhibitors and GLP-1 receptor agonists, DPP-4 inhibitors have not demonstrated improved cardiovascu lar outcomes. Sitagliptin (Januvia), saxagliptin (Onglyza), and alogliptin (Nesina) did not show significant differences in cardiovascular events between treatment and placebo groups.27,41,42 In April 2016, the FDA issued a boxed warning that saxagliptin and alogliptin may increase the risk of heart failure, particularly in people with preexisting heart failure or renal impairment.28
GLP-1 RECEPTOR AGONISTS
GLP-1 receptor agonists are medications approved to treat diabetes and obesity. Glucagon-like peptides and glucose-dependent insulinotropic poly peptides stimulate insulin secre -tion after glucose ingestion via the incretin effect, a natural process that may be decreased or absent in patients with type 2 diabetes.25,43 GLP-1 receptor agonists are subcutaneous injectable formulations, except for semaglutide, which is also available in an oral form (Rybelsus), and orforglipron and danuglipron, which are currently being reviewed for oral formulations.25,44–47 Increasing data support the effectiveness of GLP-1 receptor agonists and SGLT-2 inhibitors. A 2018 systematic review (816 trials; n = 471,038) found that GLP-1 receptor agonists and SGLT-2 inhibitors reduced cardiovascular-related deaths, nonfatal myocardial infarction, hospital admissions, end-stage renal disease, and all-cause mortality.26 Another study showed that GLP-1 receptor agonists decreased nonfatal stroke rates, whereas SGLT-2 inhibitors demonstrated greater effectiveness in decreasing end-stage renal disease.26,48 GLP-1 receptor agonists also promote weight loss (mean loss = 6.4 lb [2.9 kg]).26,46 Adverse effects and potential adverse events include gastrointestinal upset, pancreatitis, and cholelithiasis/cholecystitis.46,49 Liraglutide (Saxenda), semaglutide, and dulaglutide (Trulicity) are also associated with an increased risk of diabetic retinopathy.50
MEGLITINIDES
Meglitinides directly stimulate the pancreatic beta cells via sulfonylurea receptor-1, sulfonylurea receptor-1A, and sulfonylurea receptor-1B, causing insulin release.51 Meglitinides have a short half-life and a rapid onset of action. This may be beneficial for patients who eat once per day because it can be taken once with the largest meal to reduce postprandial hyperglycemia. Repaglinide and nateglinide are the medications in this class in the United States. The most common adverse effects are weight gain and hypoglycemia.24
SGLT-2 INHIBITORS
SGLT-2 inhibitors decrease blood glucose by increasing urinary excretion of glucose. They are expressed in the nephron's proximal tubule, mediating 90% of the filtered glucose reabsorption. By blocking glucose reabsorption, SGLT-2 inhibitors increase urinary excretion and decrease plasma glucose levels.24,52 Dosing adjustments are required in renal insufficiency, and the glucose-lowering effect of SGLT-2 inhibitors decreases as the eGFR declines.35,53 A large randomized clinical trial (n = 7,020) showed that empagliflozin (Jardiance) reduced a composite outcome of myocardial infarction, stroke, and cardiovascular death in people with established arteriosclerotic cardiovascular disease compared with placebo.43,54 Because of these benefits, SGLT-2 inhibitors are indicated in chronic kidney disease and heart failure management and in patients with diabetes who have microalbuminuria, chronic kidney disease, or chronic heart failure.45 SGLT-2 inhibitors also promote weight loss (mean = 1.5 to 7.7 lb [0.68 to 3.49 kg]), reduce potassium in hyperkalemia, and increase magnesium in hypomagnesemia.55 The FDA issued a boxed warning that SGLT-2 inhibitors may lead to euglycemic ketoacidosis and are not recommended for people with a history of diabetic ketoacidosis; patients should be educated on the ketoacidosis risk.23,53,54,56
SULFONYLUREAS
Sulfonylureas are insulin-secretagogues that directly stimulate insulin release from pancreatic beta cells, requiring these cells to function.30 Sulfonylureas also minimally improve insulin resistance in tissues.30 Sulfonylureas do not decrease all-cause mortality, but they are low cost and may offer one of the few options for patients to achieve glycemic control. Glimepiride, glyburide, and glipizide are the most prescribed sulfonylureas in the United States. Hypoglycemia is the most common adverse effect and may be worse in older adults, patients with renal dysfunction, and those concurrently using insulin.30 There are significant variations with different sulfonylureas for weight gain and hypoglycemia. Some studies suggest little to no weight gain with glimepiride and that glyburide causes a greater risk of hypoglycemia due to a longer half-life.24,55 Most adverse effects, including cardiovascular events, occur at doses beyond clinical effectiveness.55 If there is a need for glycemic control at one-half of the maximum dosage, an alternative agent should be considered.55
THIAZOLIDINEDIONES
Thiazolidinediones improve glycemic control by enhancing insulin sensitivity in muscle and adipose tissue and inhibiting hepatic glucose production.24 Thiazolidinediones substantially decrease insulin resistance, particularly when combined with other agents.24 The most common adverse effects are weight gain and fluid retention. Physicians should avoid or minimally dose thiazolidinediones for people with New York Heart Association class III or IV heart failure. Thiazolidinediones have an FDA boxed warning for increased risk of bone fractures, primarily affecting distal upper and lower limbs, and should be avoided in patients with osteopenia or osteoporosis.24 There are conflicting data about the association of bladder cancer with thiazolidinedione use.57,58 Pioglitazone is an effective treatment for patients with coexisting nonalcoholic steatohepatitis.59
Patient Assistance Programs
Most patients with type 2 diabetes are resource-limited.1,60 Patient assistance programs sponsored by pharmaceutical manufacturers, public funding, and nonprofit organizations provide financial assistance to increase medication access.61 Successful enrollment is associated with improved glycemic control.62 Educating clinicians on medication cost barriers (including barriers for patients with insurance) is critical to increasing access to newer medications.63 Increasingly robust programs, such as the Michigan Collaborative for Type 2 Diabetes, continue to reduce medication barriers by incorporating quality initiatives to promote patient and clinician awareness.11,63,64
This article updates previous articles on this topic by George, et al.24; Luna and Feinglos65; and Riddle.66
Data Sources: A PubMed search was conducted in Clinical Queries, the Cochrane database, and Essential Evidence Plus. Studies included meta-analyses, randomized controlled trials, clinical trials, and reviews from the past 10 years. Key terms searched were type 2 diabetes mellitus, oral pharmacotherapy, noninsulin therapy, and medical management. The authors ensured that each investigation containing an individual characteristic classification variable in the analyses had the variables of measurement, a justified research question for the inclusion, and clear definitions of study variables. Search dates: April 15 to 20, 2023, and February 9, 2024.

