Key Clinical Issue
What are the potential benefits and adverse effects of medications used to treat alcohol use disorder (AUD) in adults in outpatient settings?
Evidence-Based Answer
Oral naltrexone (Revia), 50 mg daily, reduces alcohol consumption across multiple outcomes, and once-daily dosing offers relative ease of use. (Strength of Recommendation [SOR]: C, disease-oriented evidence.) Acamprosate and topiramate also have evidence of benefit for reducing alcohol consumption; however, acamprosate has a higher pill burden, and topiramate has a less desirable adverse effect profile. (SOR: C, disease-oriented evidence.) There is lower strength of evidence for reducing alcohol consumption when using injectable naltrexone (Vivitrol), baclofen, or gabapentin. (SOR: C, disease-oriented evidence.) Evidence remains inadequate for use of disulfiram compared with placebo for reducing alcohol consumption.1 Current data are insufficient to understand the effect of any of the pharmacotherapies for AUD on health outcomes (e.g., mortality, quality of life, function, accidents, injury).
Practice Pointers
Excessive alcohol use is a leading cause of preventable death, contributing to more than 178,000 deaths in the United States annually per the latest data from 2020–2021.2 Screening and brief intervention for unhealthy alcohol use are recommended by the U.S. Preventive Services Task Force for adults, including pregnant people.3 National surveillance data from 2022 suggest that 29.5 million people in the United States 12 years or older (10.5%) met criteria for AUD in the past year, but only 2.1% of those received medication to treat it.4 In 2023, the Agency for Healthcare Research and Quality published an update to their 2014 systematic review of outpatient pharmacologic treatment for AUD.1
Medications for AUD are generally studied with psychosocial cointervention; thus, this summary reflects effects attributed to medications beyond nonpharmacologic interventions and placebo. Few studies have directly compared any of these medications. The data are limited surrounding medication effectiveness in special populations, specific to primary care, or relative to longer-term health outcomes (e.g., quality of life, accidents, injury, mortality).
Acamprosate, disulfiram, oral naltrexone, and the long-acting injectable formulation of naltrexone are approved by the U.S. Food and Drug Administration for treatment of AUD. Baclofen, gabapentin, ondansetron, prazosin, topiramate, and varenicline (Chantix) have also been studied for off-label use. Of these medications, oral naltrexone and acamprosate have the strongest and most consistent evidence and should be considered first-line treatments for AUD in primary care.
Oral naltrexone, an opioid antagonist, reduces return to heavy drinking, percentage of drinking days, and percentage of heavy-drinking days. Studies of oral naltrexone also show a nonsignificant trend toward reduction in return to any drinking. Acamprosate, an N-methyl-d-aspartate (NMDA) receptor modulator, reduces return to any drinking and percentage of total drinking days. Studies comparing naltrexone with acamprosate have not established the superiority of one medication over the other (prevent return to any drinking: oral naltrexone, number needed to treat [NNT] = 18 vs. acamprosate, NNT = 11; prevent return to heavy drinking: oral naltrexone, NNT = 11 [data not available for acamprosate]).5 No new comparative studies were identified since the 2014 Agency for Healthcare Research and Quality review.
Considerations when selecting acamprosate or naltrexone include dosing regimen, contraindications, and potential adverse effects (Table 1). Note that oral naltrexone is metabolized through the liver and acamprosate is metabolized by the kidneys.
TABLE 1. Comparison of First-Line Treatments Acamprosate and Oral Naltrexone for Alcohol Use Disorder

| Considerations | Acamprosate | Naltrexone (Revia) |
|---|---|---|
| Dosing regimen | Two 333-mg pills, three times daily* | One 50-mg pill, once daily |
| Contraindications | Patients with kidney failure | Patients who use, or will need to use, opioids† Patients with liver failure, acute hepatitis Use caution in patients with other liver diseases, depending on potential risks and benefits |
| Potential adverse effects | Anxiety, diarrhea, vomiting | Dizziness, nausea, vomiting |
*—Dosing should be adjusted for patients with moderate kidney impairment.
†—Can precipitate withdrawal in those with physiologic opioid dependence.
Previous evidence on injectable naltrex -one's effectiveness was limited6; however, a 2021 randomized controlled trial in a population of individuals experiencing homelessness showed reduction in total drinking days and heavy-drinking days.7 Injectable naltrexone could be a good option for patients who have difficulty with daily medication adherence. Disulfiram causes accumulation of acetaldehyde during alcohol consumption, creating adverse effects of flushing, nausea, vomiting, and heart rate and blood pressure changes. Importantly, available evidence suggests that disulfiram does not reduce alcohol consumption outcomes and therefore is not recommended as treatment for AUD, particularly given the risk for negative events if alcohol use occurs while taking the medication.1
Medications not approved by the U.S. Food and Drug Administration that have some effectiveness in AUD management include topiramate, baclofen, and gabapentin. Topiramate demonstrated reduction in total drinking days, heavy-drinking days, and number of drinks per drinking day; however, common adverse effects include anorexia, concentration changes, dizziness, paresthesia, and psycho-motor slowing. Studies with lower strength of evidence show reduction in return to any drinking when taking baclofen as well as reduction in return to heavy drinking when taking gabapentin.
A guide for physicians about medications for AUD is available from the Substance Abuse and Mental Health Services Administration. Additionally, a resource guide for AUD from the American Society of Addiction Medicine is available.
CLINICAL BOTTOM LINE
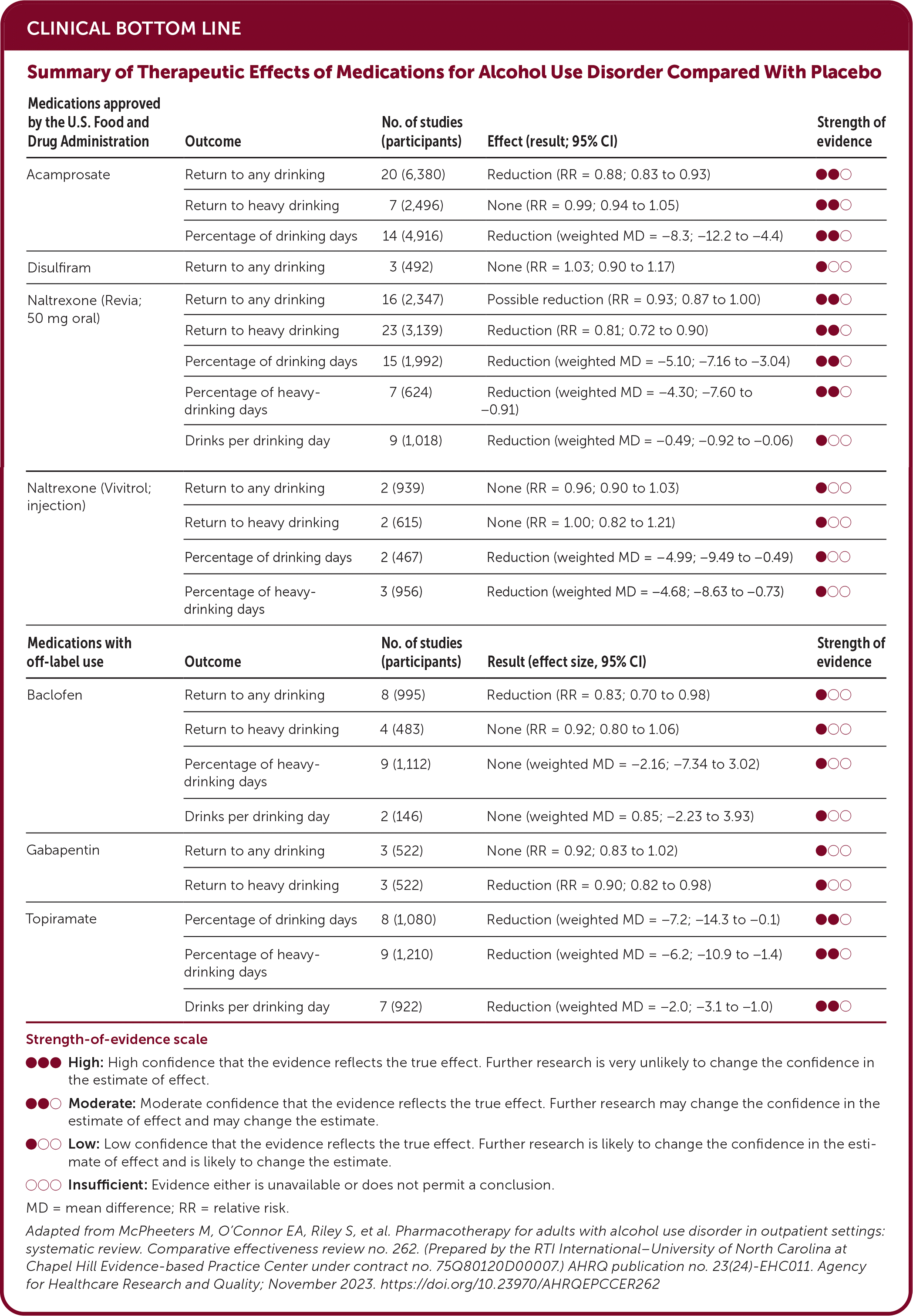
| Medications approved by the U.S. Food and Drug Administration | Outcome | No. of studies (participants) | Effect (result; 95% CI) | Strength of evidence |
| Acamprosate | Return to any drinking | 20 (6,380) | Reduction (RR = 0.88; 0.83 to 0.93) | • • ○ |
| Return to heavy drinking | 7 (2,496) | None (RR = 0.99; 0.94 to 1.05) | • • ○ | |
| Percentage of drinking days | 14 (4,916) | Reduction (weighted MD = −8.3; −12.2 to −4.4) | • • ○ | |
| Disulfiram | Return to any drinking | 3 (492) | None (RR = 1.03; 0.90 to 1.17) | • ○ ○ |
| Naltrexone (Revia; 50 mg oral) | Return to any drinking | 16 (2,347) | Possible reduction (RR = 0.93; 0.87 to 1.00) | • • ○ |
| Return to heavy drinking | 23 (3,139) | Reduction (RR = 0.81; 0.72 to 0.90) | • • ○ | |
| Percentage of drinking days | 15 (1,992) | Reduction (weighted MD = −5.10; −7.16 to −3.04) | • • ○ | |
| Percentage of heavy-drinking days | 7 (624) | Reduction (weighted MD = −4.30; −7.60 to −0.91) | • • ○ | |
| Drinks per drinking day | 9 (1,018) | Reduction (weighted MD = −0.49; −0.92 to −0.06) | • ○ ○ | |
| Naltrexone (Vivitrol; injection) | Return to any drinking | 2 (939) | None (RR = 0.96; 0.90 to 1.03) | • ○ ○ |
| Return to heavy drinking | 2 (615) | None (RR = 1.00; 0.82 to 1.21) | • ○ ○ | |
| Percentage of drinking days | 2 (467) | Reduction (weighted MD = −4.99; −9.49 to −0.49) | • ○ ○ | |
| Percentage of heavy-drinking days | 3 (956) | Reduction (weighted MD = −4.68; −8.63 to −0.73) | • ○ ○ | |
| Medications with off-label use | Outcome | No. of studies (participants) | Result (effect size, 95% CI) | Strength of evidence |
| Baclofen | Return to any drinking | 8 (995) | Reduction (RR = 0.83; 0.70 to 0.98) | • ○ ○ |
| Return to heavy drinking | 4 (483) | None (RR = 0.92; 0.80 to 1.06) | • ○ ○ | |
| Percentage of heavy-drinking days | 9 (1,112) | None (weighted MD = −2.16; −7.34 to 3.02) | • ○ ○ | |
| Drinks per drinking day | 2 (146) | None (weighted MD = 0.85; −2.23 to 3.93) | • ○ ○ | |
| Gabapentin | Return to any drinking | 3 (522) | None (RR = 0.92; 0.83 to 1.02) | • ○ ○ |
| Return to heavy drinking | 3 (522) | Reduction (RR = 0.90; 0.82 to 0.98) | • ○ ○ | |
| Topiramate | Percentage of drinking days | 8 (1,080) | Reduction (weighted MD = −7.2; −14.3 to −0.1) | • • ○ |
| Percentage of heavy-drinking days | 9 (1,210) | Reduction (weighted MD = −6.2; −10.9 to −1.4) | • • ○ | |
| Drinks per drinking day | 7 (922) | Reduction (weighted MD = −2.0; −3.1 to −1.0) | • • ○ | |
Strength-of-evidence scale
• • • High: High confidence that the evidence reflects the true effect. Further research is very unlikely to change the confidence in the estimate of effect.
• • ○ Moderate: Moderate confidence that the evidence reflects the true effect. Further research may change the confidence in the estimate of effect and may change the estimate.
• ○ ○ Low: Low confidence that the evidence reflects the true effect. Further research is likely to change the confidence in the estimate of effect and is likely to change the estimate.
○ ○ ○ Insufficient: Evidence either is unavailable or does not permit a conclusion.
MD = mean difference; RR = relative risk.
Adapted from McPheeters M, O'Connor EA, Riley S, et al. Pharmacotherapy for adults with alcohol use disorder in outpatient settings: systematic review. Comparative effectiveness review no. 262. (Prepared by the RTI International–University of North Carolina at Chapel Hill Evidence-based Practice Center under contract no. 75Q80120D00007.) AHRQ publication no. 23(24)-EHC011. Agency for Healthcare Research and Quality; November 2023. https://doi.org/10.23970/AHRQEPCCER262
Editor's Note: American Family Physician SOR ratings are different from the AHRQ Strength-of-Evidence ratings.

