Testosterone deficiency, or male hypogonadism, is a clinical syndrome that can be defined as persistently low serum testosterone levels in the setting of symptoms consistent with testosterone deficiency. Studies suggest that testosterone replacement therapy may improve sexual function, depressive symptoms, bone density, and lean body mass. Evidence is conflicting regarding its effect on cardiovascular events and mortality. Although prior studies suggested that testosterone replacement therapy increased the risk of cardiovascular disease, a large, randomized trial showed that it does not increase the risk of myocardial infarction or stroke, even in patients at high risk. After a detailed discussion of the potential benefits and risks through shared decision-making, testosterone replacement therapy should be considered for men with testosterone deficiency to correct selected symptoms and induce and maintain secondary sex characteristics. Treatment method should take into consideration patient preference, pharmacokinetics, potential for medication interactions, formulation-specific adverse effects, treatment burden, and cost. Clinicians should monitor men receiving testosterone replacement therapy for symptom improvement, potential adverse effects, and adherence. Serum testosterone, hematocrit, and prostate-specific antigen levels should be measured at baseline and at least annually in men 40 years or older receiving testosterone replacement therapy. (Am Fam Physician. 2024;109(6):543-549. Copyright © 2024 American Academy of Family Physicians.)
Testosterone deficiency (TD), or male hypogonadism, is a clinical syndrome that can be defined as persistently low serum testosterone levels in the setting of symptoms consistent with TD.1 Biochemical male hypogonadism specifically refers to low serum testosterone levels with or without symptoms. The global prevalence of TD ranges from 10% to 40%, with an increased rate in patients 45 to 50 years and older.2 The prevalence of biochemical male hypogonadism increases with age, ranging from 12% among men 50 to 59 years of age to 49% among those 80 years and older.3 This article focuses on the diagnosis and management of primary hypogonadism in cisgender men and other patients with male gonads. Secondary hypogonadism is typically associated with hypothalamic or pituitary disease and is outside the scope of this review. The use of testosterone as gender-affirming masculinizing hormone therapy was discussed in a previous American Family Physician article.4
WHAT'S NEW ON THIS TOPIC

| In a 2023 randomized trial involving men 45 to 80 years of age with hypogonadism and preexisting or elevated risk of cardiovascular disease, testosterone replacement therapy was noninferior to placebo for the risk of major adverse cardiac events. This primary outcome occurred in 7.0% of the treatment group and in 7.3% of the placebo group. |
| In men 65 years and older with symptomatic testosterone deficiency, increasing total testosterone concentrations to the mid-normal range for 1 year had a moderate benefit in sexual function and some benefit for mood and depressive symptoms, but no benefit for vitality or walking distance. |
SORT: KEY RECOMMENDATIONS FOR PRACTICE
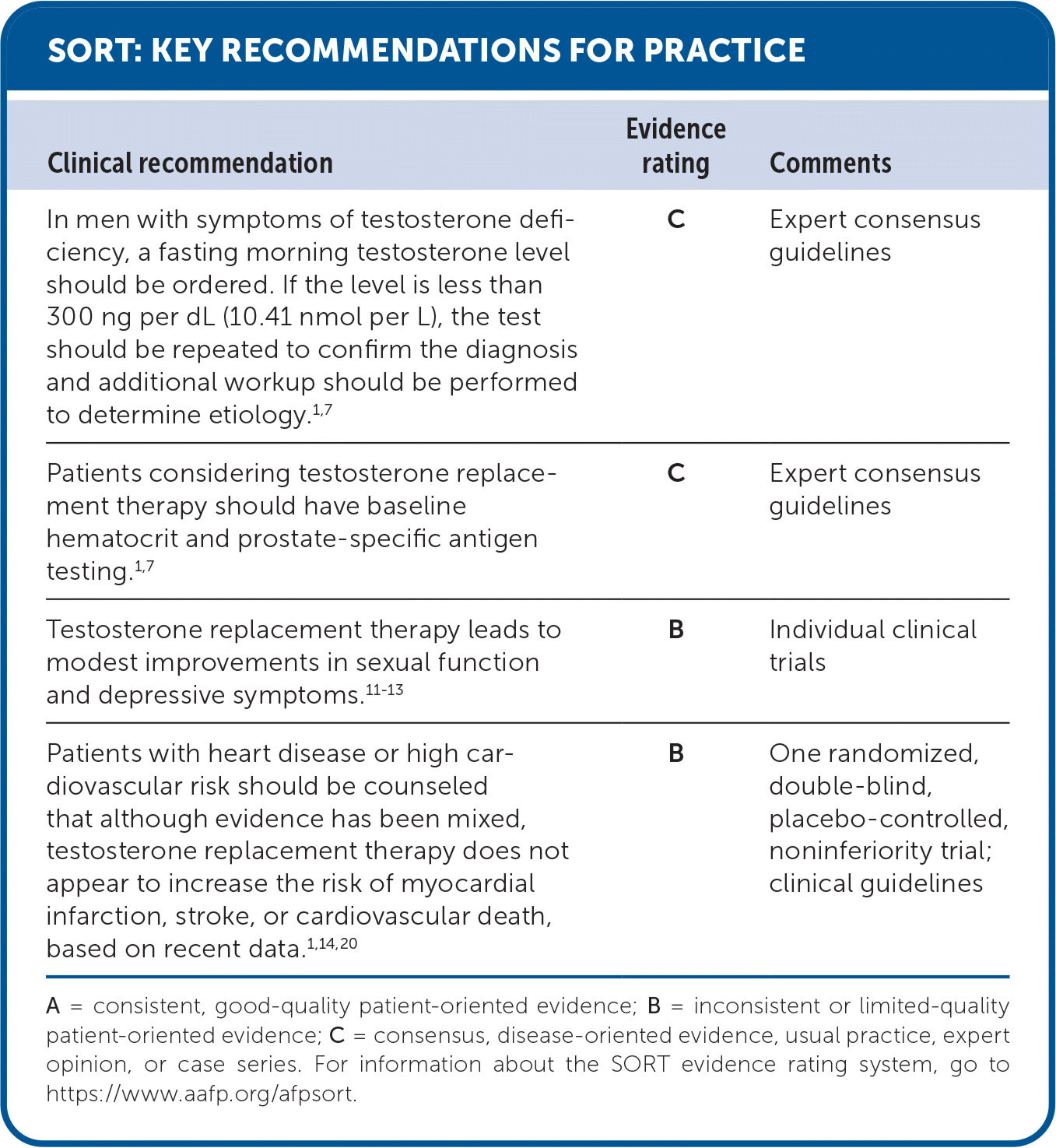
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Diagnosis
The Endocrine Society recommends diagnosing testicular hypogonadism only in men with signs and symptoms consistent with TD (Table 15 ) and unequivocally and consistently low serum testosterone levels.1 Validated tools such as the ADAM (Androgen Deficiency in the Aging Male) questionnaire can guide clinicians toward an accurate symptomatic diagnosis.6
TABLE 1. Symptoms and Signs Suggestive of Androgen Deficiency in Men

| A. More-specific symptoms and signs |
| Incomplete or delayed sexual development, eunuchoidism |
| Reduced sexual desire (libido) and activity |
| Decreased spontaneous erections |
| Breast discomfort, gynecomastia |
| Loss of body (axillary and pubic) hair, reduced shaving |
| Very small (especially < 5 mL) or shrinking testes |
| Inability to father children, low or zero sperm count |
| Height loss, low-trauma fracture, low bone mineral density |
| Hot flushes, sweats |
| B. Other less-specific symptoms and signs |
| Decreased energy, motivation, initiative, and self-confidence |
| Feeling sad or blue, depressed mood, dysthymia |
| Poor concentration and memory |
| Sleep disturbance, increased sleepiness |
| Mild anemia (normochromic, normocytic, in the female range) |
| Reduced muscle bulk and strength |
| Increased body fat, body mass index |
| Diminished physical or work performance |
Reprinted with permission from Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline [published correction appears in J Clin Endocrinol Metab. 2021;106(7):e2848]. J Clin Endocrinol Metab. 2010;95(6):2537.
The Endocrine Society and the American Urological Association recommend measuring fasting morning serum total testosterone (TT) concentrations and confirming a diagnosis of TD by repeat measurement.1,7 Both organizations define TD as serum TT less than 300 ng per dL (10.41 nmol per L).
In men who meet symptomatic and biochemical criteria for TD with serum TT under 300 ng per dL, additional diagnostic evaluation is recommended to determine potential causes of androgen deficiency (Figure 11,7,8 ). Serum measurements of TT should be repeated to confirm the diagnosis of TD. A baseline hematocrit level greater than 48% is a contraindication due to the risk of developing worsening erythrocytosis. Hematocrit levels greater than 54% during therapy warrant discontinuation and referral to hematology. If luteinizing hormone levels are low or low-normal, prolactin should be measured to evaluate for pituitary adenoma. An elevated luteinizing hormone measurement with two confirmed serum TT results less than 300 ng per dL confirms TD. Upon diagnosis, patients 40 years and older should have a baseline prostate-specific antigen test, as recommended by the American Urological Association and Endocrine Society. If the prostate-specific antigen level is normal (i.e., less than 4 ng per mL [4 mcg per L]), or the patient does not need a test based on age, testosterone replacement therapy (TRT) can be considered.
FIGURE 1.

Evaluation of male hypogonadism.
Adapted with permission from Petering RC, Brooks NA. Testosterone therapy: review of clinical applications [published correction appears in Am Fam Physician. 2019;100(7):393]. Am Fam Physician. 2017;96(7):443, with additional information from references 1 and 7.
Potential Benefits
Studies suggest that benefits of TRT include improvement in sexual function, depressive symptoms, bone density, and lean body mass. Evidence is inconclusive regarding effects on cardiovascular events and mortality.1
CARDIOVASCULAR EVENTS AND MORTALITY
A retrospective cohort study of 83,010 men with TD in the U.S. Department of Veterans Affairs health care system compared men who received TRT with untreated men and found an association with decreased mortality risk in the treated group (10.3%) vs. the untreated group (20.7%; P < .001%).9 Participants were categorized into three groups: (G1) TRT with normalization of serum TT levels, (G2) TRT without normalization of serum TT levels, and (G3) no TRT. All-cause mortality (hazard ratio [HR] = 0.44; 95% CI, 0.42 to 0.46), risk of myocardial infarction (MI; HR = 0.76; 95% CI, 0.63 to 0.93), and incidence of ischemic stroke (HR = 0.64; 95% CI, 0.43 to 0.96) were significantly lower in G1 vs. G3. All-cause mortality (HR = 0.53; 95% CI, 0.50 to 0.55), risk of MI (HR = 0.82; 95% CI, 0.71 to 0.95), and incidence of ischemic stroke (HR = 0.70; 95% CI, 0.51 to 0.96) were significantly lower in G1 vs. G2 with no difference in MI or stroke risk between G2 and G3. However, these potential benefits of TRT have not been replicated in randomized trials.
In a placebo-controlled, double-blind, parallel-group randomized trial, 156 men with a mean age of 67.6 years were randomized to daily treatment with TRT or placebo for 3 years with dose titration to yield TT levels between 500 and 900 ng per dL (17.35 and 31.23 nmol per L).10 Comorbid conditions included hypertension (42%), obesity (27%), diabetes mellitus (15%), and cardiovascular disease (15%). TRT did not correlate with a significant difference in rates of change in carotid artery intima-media thickness or coronary artery calcium scores. Moreover, it did not improve overall sexual function or health-related quality of life.
SEXUAL AND PHYSICAL FUNCTION AND MOOD
Studies have shown beneficial effects of TRT on sexual function and mood in men with hypogonadism.11,12 In a coordinated set of seven double-blind, placebo-controlled trials, men 65 years and older with serum TT levels less than 275 ng per dL (9.54 nmol per L) and symptoms of TD were randomized to receive TRT or placebo for 1 year, with each patient participating in one or more of three trials.13 Each of the trials assessed sexual function, physical function, or vitality. Increasing serum TT concentrations to the mid-normal range for 1 year showed moderate benefit in sexual function and some benefit for mood and depressive symptoms, but no benefit for vitality or walking distance. Given limited evidence, the American College of Physicians recommends that clinicians discuss initiating TRT in men with TD only to improve sexual function but not for concerns related to energy, vitality, physical function, or cognition.14
BONE DENSITY AND LEAN BODY MASS
In a placebo-controlled, double-blind trial with 211 participants, patients with TD had significantly greater increases than the placebo group in mean spine trabecular volumetric bone mineral density (vBMD), spine peripheral vBMD, hip trabecular and peripheral vBMD, and mean estimated strength of spine trabecular bone, spine peripheral bone, and hip trabecular and peripheral bone.15
In a single-center, randomized, double-blind, placebo-controlled study, 274 men 65 years and older with biochemical hypogonadism were randomized to treatment with transdermal testosterone or placebo. Lean body mass increased and fat mass decreased in the treatment group by 1.08 ± 1.8 kg (P < .001) and 0.9 ± 1.6 kg (P = .02), respectively.16
Potential Harms
TRT may increase the risk of erythrocytosis. However, there is no evidence that TRT increases the risk of prostate cancer. Although previous studies suggested that TRT increased the risk of cardiovascular disease, a more recent trial did not demonstrate an increased risk of MI or stroke.
A trial studied 209 community-dwelling men with serum TT levels of 100 to 350 ng per dL (3.47 to 12.15 nmol per L), a mean age of 74 years, limitations in mobility, and a high prevalence of hypertension, diabetes, hyperlipidemia, and obesity. Participants were randomized to daily treatment with testosterone gel or placebo for 6 months.17 Patients in the treatment group experienced increases in leg and chest press strength, but the trial was stopped early because they also experienced increased risk of cardiovascular events (HR = 2.4; P = .05).
A retrospective cohort study of a health care insurance claims database evaluated the risk of acute nonfatal MI following an initial TRT prescription (n = 55,593).18 The incidence rate of MI in the 90 days following the initial prescription was compared with the rate in the year before the initial prescription. The overall rate of nonfatal MI in the TRT group of 55,593 men increased in all ages from 3.48 per 1,000 patient-years to 4.75 per 1,000 patient-years.
Subsequent to these studies, the U.S. Food and Drug Administration began requiring label changes for all prescription testosterone products to reflect the possible increased risk of MI and stroke, stating that the benefit and safety of these medications have not been established for the treatment of low testosterone levels due to aging, even if a man's symptoms seem related to low testosterone.19
To further evaluate the cardiovascular safety of TRT, the TRAVERSE (Testosterone Replacement Therapy for Assessment of Long-term Vascular Events and Efficacy Response in Hypogonadal Men) study conducted a multicenter, randomized, double-blind, placebo-controlled, noninferiority trial with 5,246 men 45 to 80 years of age with high risk of or preexisting cardiovascular disease, symptoms of hypogonadism, and two fasting testosterone measurements of less than 300 ng per dL.20 In men with hypogonadism and elevated risk of or preexisting cardiovascular disease, TRT was found to be noninferior to placebo regarding risk of major adverse cardiac events.1,14,20 Each day, patients received transdermal 1.62% testosterone gel, adjusted to maintain TT levels between 350 and 750 ng per dL (12.15 and 26.03 nmol per L), or placebo gel for a mean duration of 22 months. The primary outcome of first occurrence of any component of a composite of death from cardiovascular causes, nonfatal MI, or nonfatal stroke occurred in 7.0% of the treatment group and in 7.3% of the placebo group (HR = 0.96; 95% CI, 0.78 to1.17; P < .001 for noninferiority). A statistically higher incidence of atrial fibrillation (3.5% vs. 2.4%), acute kidney injury (2.3% vs. 1.5%), and pulmonary embolism (0.9% vs. 0.5%) was observed in the treatment group compared with the placebo group.
Treatment Options and Monitoring
After a detailed discussion of the potential benefits and risks of therapy through shared decision-making, TRT should be considered for men with TD to correct selected symptoms and induce and maintain secondary sex characteristics (e.g., facial hair, muscle mass). The Endocrine Society guidelines recommend against TRT for men who are planning to conceive in the near future or those with certain conditions (Table 2).1
TABLE 2. Contraindications to Testosterone Replacement Therapy

| Breast cancer |
| Elevated hematocrit (i.e., > 48%) |
| Fertility planning |
| Myocardial infarction or stroke within the past 6 months |
| Prostate cancer or a palpable prostate nodule or induration |
| Prostate-specific antigen level > 3 ng per mL (3 mcg per L) in men at increased risk of prostate cancer (e.g., African American men, men who have a first-degree relative with prostate cancer) without urologic evaluation |
| Prostate-specific antigen level > 4 ng per mL (4 mcg per L) |
| Severe lower urinary tract symptoms |
| Thrombophilia |
| Uncontrolled congestive heart failure |
| Untreated severe obstructive sleep apnea |
Information from reference 1.
According to the Endocrine Society guidelines, the goal of TRT should be to restore TT concentrations to the mid-normal range (300 to 900 ng per dL) and improve symptoms of TD. Treatment with any of the approved formulations should take into consideration patient preference, pharmacokinetics, potential for medication interactions, formulation-specific adverse effects, treatment burden, and insurance coverage vs. patient cost1 (Table 31,21–28 ). Clinicians should monitor men receiving TRT for symptom improvement, potential adverse effects, adherence, and measurement of serum TT and hematocrit concentrations at 3 to 6 months and then annually. Prostate cancer risk should be evaluated via measurement of prostate-specific antigen after initiating TRT and then at least annually.1
TABLE 3. Testosterone Replacement Therapy Methods
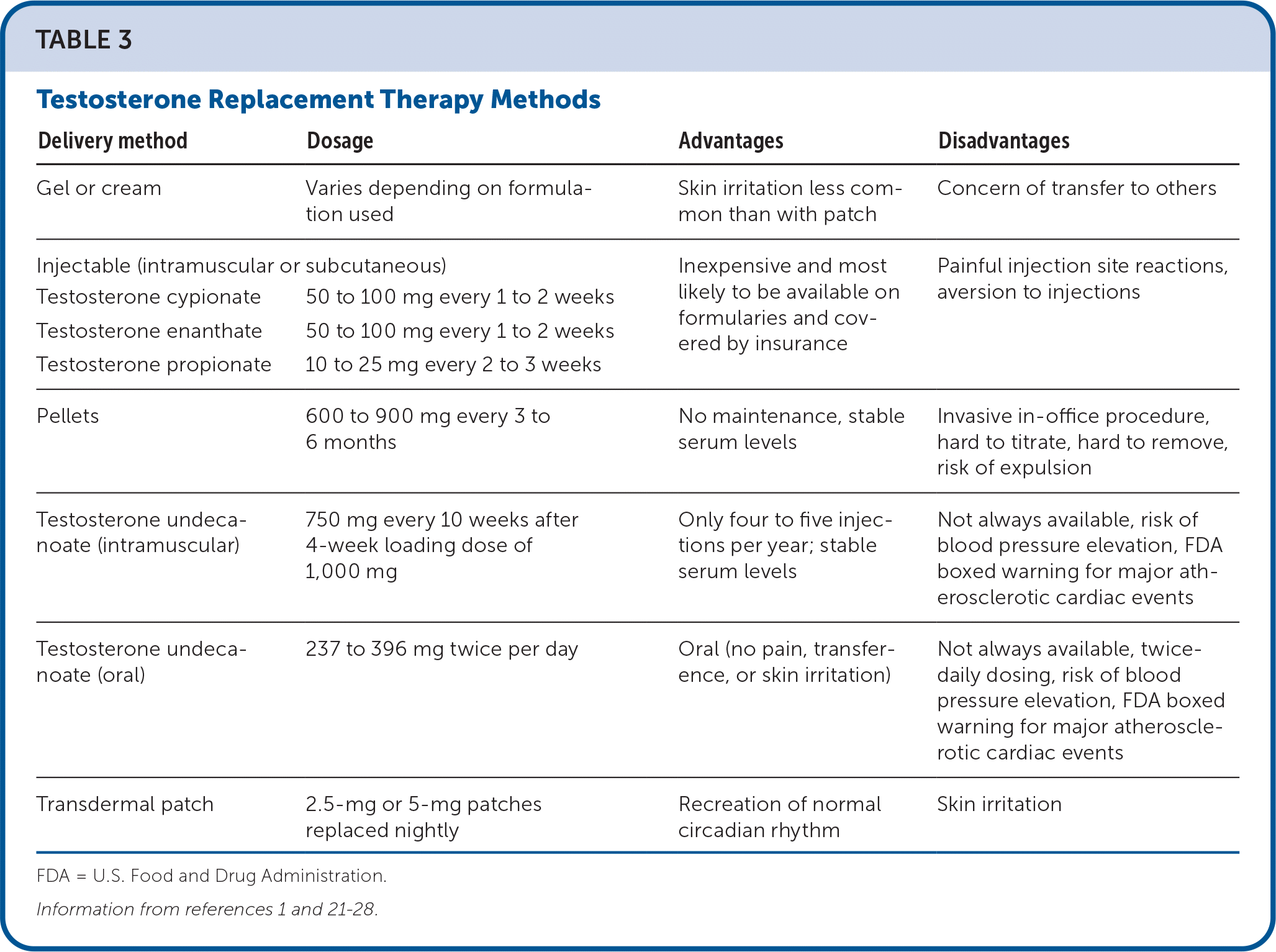
| Delivery method | Dosage | Advantages | Disadvantages |
|---|---|---|---|
| Gel or cream | Varies depending on formulation used | Skin irritation less common than with patch | Concern of transfer to others |
| Injectable (intramuscular or subcutaneous) | Inexpensive and most likely to be available on formularies and covered by insurance | Painful injection site reactions, aversion to injections | |
| Testosterone cypionate Testosterone enanthate Testosterone propionate | 50 to 100 mg every 1 to 2 weeks 50 to 100 mg every 1 to 2 weeks 10 to 25 mg every 2 to 3 weeks | ||
| Pellets | 600 to 900 mg every 3 to 6 months | No maintenance, stable serum levels | Invasive in-office procedure, hard to titrate, hard to remove, risk of expulsion |
| Testosterone undecanoate (intramuscular) | 750 mg every 10 weeks after 4-week loading dose of 1,000 mg | Only four to five injections per year; stable serum levels | Not always available, risk of blood pressure elevation, FDA boxed warning for major atherosclerotic cardiac events |
| Testosterone undecanoate (oral) | 237 to 396 mg twice per day | Oral (no pain, transference, or skin irritation) | Not always available, twice-daily dosing, risk of blood pressure elevation, FDA boxed warning for major atherosclerotic cardiac events |
| Transdermal patch | 2.5-mg or 5-mg patches replaced nightly | Recreation of normal circadian rhythm | Skin irritation |
FDA = U.S. Food and Drug Administration.
This article updates previous articles on this topic by Fugh-Berman,21 Petering and Brooks,8 and Margo and Winn.29
Data Sources: A PubMed search was conducted using the key terms testosterone, testosterone deficiency, male hypogonadism, testosterone replacement therapy, guidelines, benefits, and adverse effects. Related to potential benefits and adverse effects, the terms sexual function, sexual dysfunction, depression, bone density, prostate cancer, cardiovascular disease, and stroke were searched in relation to testosterone. The search included society guidelines, meta-analyses, randomized controlled trials, clinical trials, and reviews. The U.S. Preventive Services Task Force, the Cochrane Database of Systematic Reviews, Essential Evidence Plus, and the Agency for Healthcare Research and Quality were reviewed. Original research studies cited within society guidelines were also reviewed. Search dates: January through June 2023 and April 2024.

