Diabetic peripheral neuropathy occurs in up to 50% of patients with diabetes mellitus and increases the risk of diabetic foot ulcers and infections. Consistent screening and clear communication are essential to decrease disparities in assessment of neuropathic symptoms and diagnosis. Physicians should address underlying risk factors such as poor glycemic control, vitamin B12 deficiency, elevated blood pressure, and obesity to reduce the likelihood of developing neuropathy. First-line drug therapy for painful diabetic peripheral neuropathy includes duloxetine, gabapentin, amitriptyline, and pregabalin; however, these medications do not restore sensation to affected extremities. Evidence for long-term benefit and safety of first-line treatment options is lacking. Second-line drug therapy includes nortriptyline, imipramine, venlafaxine, carbamazepine, oxcarbazepine, topical lidocaine, and topical capsaicin. Periodic, objective monitoring of medication response is critical because patients may not obtain desired pain reduction, adverse effects are common, and serious adverse effects can occur. Opioids should generally be avoided. Nondrug therapies with low- to moderate-quality evidence include exercise and neuromodulation with spinal cord stimulation or transcutaneous electrical nerve stimulation. Peripheral transcutaneous electrical nerve stimulation is well tolerated and inexpensive, but benefits are modest. Other treatments, such as acupuncture, alpha-lipoic acid, acetyl-L-carnitine, cannabidiol, and onabotulinumtoxinA need further study in patients with diabetic peripheral neuropathy.
Diabetic peripheral neuropathy is the most common complication of diabetes mellitus, affecting up to 50% of patients with diabetes.1 It commonly presents as distal symmetric polyneuropathy with a stocking-and-glove distribution and a loss of sensation that may be painful or asymptomatic, starting in the toes and moving proximally. Diabetic peripheral neuropathy may result in a loss of protective sensation and cascade into the development of foot ulcers, wounds, infections, and tissue necrosis.2,3
SORT: KEY RECOMMENDATIONS FOR PRACTICE
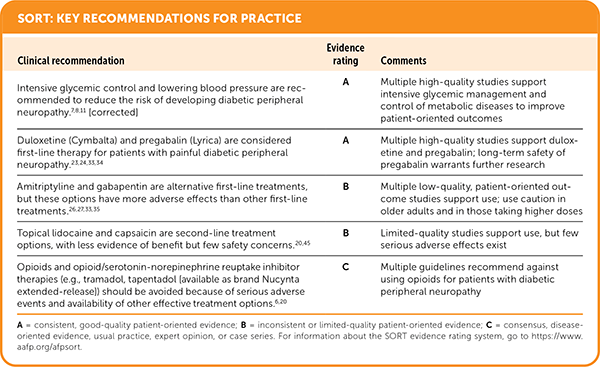
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
BEST PRACTICES IN NEUROLOGY

| Recommendation | Sponsoring organization |
|---|---|
| Do not prescribe neuropathic pain agents for painless neuropathy. | American Podiatric Medical Association |
| Do not routinely use B vitamin supplements for the treatment of polyneuropathy or neuropathic pain unless a deficiency exists. | American Association of Neuromuscular and Electrodiagnostic Medicine |
Note: For supporting citations and to search Choosing Wisely recommendations relevant to primary care, see https://www.aafp.org/pubs/afp/collections/choosing-wisely.html.
Primary Prevention
Prevention of diabetic peripheral neuropathy is essential because there is no cure for neuropathy; treatments should focus on symptom management and slowing disease progression.2,4–6 Reducing the risk of amputations and protecting extremities from trauma continue to be paramount.3,6 Glycemic control remains the primary mechanism to prevent diabetic peripheral neuropathy, with greater benefit from early optimization of glucose control. In patients with type 1 diabetes, intensive glycemic control with an A1C goal of approximately 7% reduces the risk of symptomatic neuropathy by up to 60%.7 Only modest reductions in neuropathy risk occur with intensive glycemic control in patients with type 2 diabetes.8 Assessment for vitamin B12 deficiency may be valuable because a deficiency increases the risk of neuropathy, may lead to a misdiagnosis of diabetic peripheral neuropathy, and can be treated with supplementation. Initial vitamin B12 levels should be measured in individuals with absorption issues (e.g., after bariatric surgery), with autoimmune disease, who eat a strict vegetarian diet, or who take metformin.9,10 Maintaining a systolic blood pressure of 140 mm Hg or lower can decrease the development of diabetic peripheral neuropathy.11 [corrected]
Screening
The American Diabetes Association Diabetes Standards of Care guidelines recommend diabetic peripheral neuropathy screening with monofilament testing, but this practice has poor sensitivity for detecting peripheral neuropathy.6,12 Diabetic peripheral neuropathy is a clinical diagnosis and a diagnosis of exclusion, and other assessment factors (e.g., neurologic, skin, vascular, foot deformities) should be considered.4,6 Additionally, a patient's cultural background should be considered when screening because Black and Hispanic patients report difficulty communicating with physicians about their symptoms and have a lower rate of diabetic peripheral neuropathy diagnosis.13 Further research may highlight drivers of these disparities, but communication in a patient's preferred language and verification of understanding are key.
Secondary Prevention
Multiple tools are available for assessing limitations secondary to neuropathy; the Overall Neuropathy Limitations Scale is one of the available tools that can assist with management decisions. Primary prevention efforts (e.g., intensive glycemic control, comorbidity management) may reduce long-term pain; however, patients with rapid, significant glucose improvements may paradoxically experience short-term increases in pain from treatment-induced neuropathy of diabetes.6,14
Increased emphasis on foot care and preventing diabetic foot ulcers and amputations should occur.3 Individuals at high risk (e.g., patients receiving dialysis, having Charcot foot or other structural foot deformities, having a history of ulcers or peripheral arterial disease) should be referred to foot care specialists; these specialists can advise the patient about specialized footwear, which may be valuable.3 Individuals with foot ulcerations should receive prompt treatment and frequent surveillance to prevent progression.3,6
Correction of underlying risk factors should be addressed to minimize the need to treat diabetic peripheral neuropathy. Treatment with vitamin B12 is beneficial in individuals with a deficiency but not in individuals with adequate levels.9 For patients with severe obesity (body mass index 35 kg per m2 or greater), weight loss interventions may improve quality-of-life scores.15 Tobacco cessation should also be a priority to slow the progression of neuropathy and reduce other complications of diabetes.16
Treatment
A multimodal treatment plan should be developed for individuals with painful diabetic peripheral neuropathy. Reducing pain is the primary objective because current therapy does not reverse previous nerve damage.6 Four drug treatments have been approved by the U.S. Food and Drug Administration for diabetic peripheral neuropathy: duloxetine (Cymbalta), pregabalin (Lyrica), tapentadol (available as brand Nucynta extended-release), and capsaicin patch (Qutenza). Many treatments, however, have been used successfully off-label. Nondrug therapy comprises neuromodulatory therapy and exercise.2,6,17 Combination treatment with first-line therapies from different drug classes may be reasonable for patients who have inadequate pain management while taking monotherapy.18 Figure 1 provides guidance for treatment in patients with diabetic peripheral neuropathy.2,6,17,19–22
FIGURE 1.
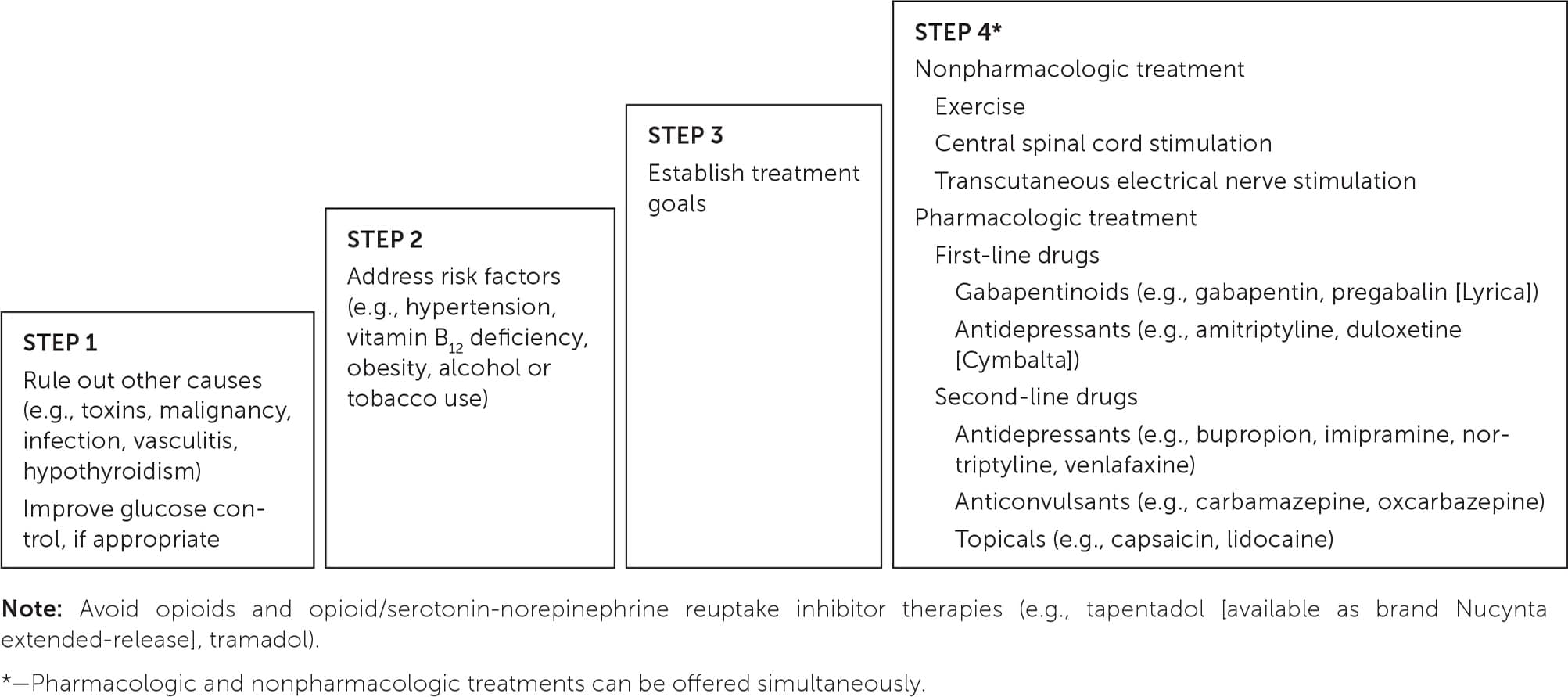
Managing pain from diabetic peripheral neuropathy.
ANTIDEPRESSANTS
Duloxetine, a serotonin-norepinephrine reuptake inhibitor, is first-line therapy and the most studied antidepressant for patients with diabetic peripheral neuropathy. A 2023 Cochrane review concluded that it is the only antidepressant with adequate data in patients with chronic pain.23 With duloxetine, the risk ratio (RR) was 1.73 (95% CI, 1.44 to 2.08) for a 50% or greater reduction in pain at 12 weeks.24 The benefit of duloxetine plateaus at 40 to 60 mg per day, with no greater effect at 120 mg per day and no effect at 20 mg per day.24 Duloxetine may make several conditions worse (e.g., uncontrolled hypertension, cardiovascular disease, hepatic impairment) or have more potent effects in patients with renal insufficiency, requiring dose adjustment, frequent monitoring, or drug avoidance.25
In a few head-to-head trials, duloxetine has been compared with pregabalin and amitriptyline in patients with diabetic peripheral neuropathy. In a 12-week study with 804 participants, more individuals taking duloxetine had a 50% or greater reduction in pain (38%) than those taking pregabalin (26%; RR = 1.46; 95% CI, 1.19 to 1.80).24 Similar rates of at least a 50% reduction in pain were seen with duloxetine (59%) and amitriptyline (55%) in a six-week crossover study of 62 participants.24 Dry mouth was a more common adverse effect when taking amitriptyline vs. duloxetine (55% vs. 24%; P < .01).26
Amitriptyline is another antidepressant commonly prescribed for painful diabetic peripheral neuropathy, although its magnitude of effect remains unclear. A 2015 Cochrane review noted that amitriptyline studies were of weak quality and had high risk of bias.27 Although the studies are flawed, amitriptyline should remain as a first-line treatment, with continued monitoring for effectiveness and adverse effects.27
Use of other antidepressants may be reasonable, but evidence of benefit for patients with painful diabetic peripheral neuropathy is limited. Data for venlafaxine and milnacipran (available as brand Savella) comprise low-quality strials showing limited benefits.28,29 As a result, these treatments should be reserved for patients who are unable to tolerate or have poor response to other options. Similarly, because of weak, low-quality evidence, use of nortriptyline and imipramine should be more reserved considering the questionable benefits and significant adverse-effect burden.30,31 Bupropion is another antidepressant with one low-quality trial in a six-week study that shows its use could improve pain scores.32
ANTICONVULSANTS
Pregabalin and gabapentin are first-line treatment options for diabetic peripheral neuropathy, with pregabalin being slightly preferred.33 Higher pregabalin doses are more effective; a 50% reduction in pain occurred in 31% of patients taking 300 mg daily vs. 41% of patients taking 600 mg daily.34 The overall quality of data supporting gabapentin is weaker but showed a similar finding that higher doses (at least 1,200 mg per day) are needed to obtain a 50% reduction in pain in 38% of patients.6,35 Few head-to-head studies have been conducted, but a 2022 study with 100 patients showed that pregabalin had less sleep interference and a greater difference in global impressions of change compared with gabapentin.33
Initially starting with lower doses of pregabalin and gabapentin and taking doses at bedtime are reasonable to avoid oversedation2,34,35 (Table 12,19,24,25,32,34–38 ). Reduced doses to minimize adverse effects may be needed in patients with renal insufficiency and in older individuals.2,19,25
TABLE 1. Medications for Treating Patients With Painful Diabetic Peripheral Neuropathy
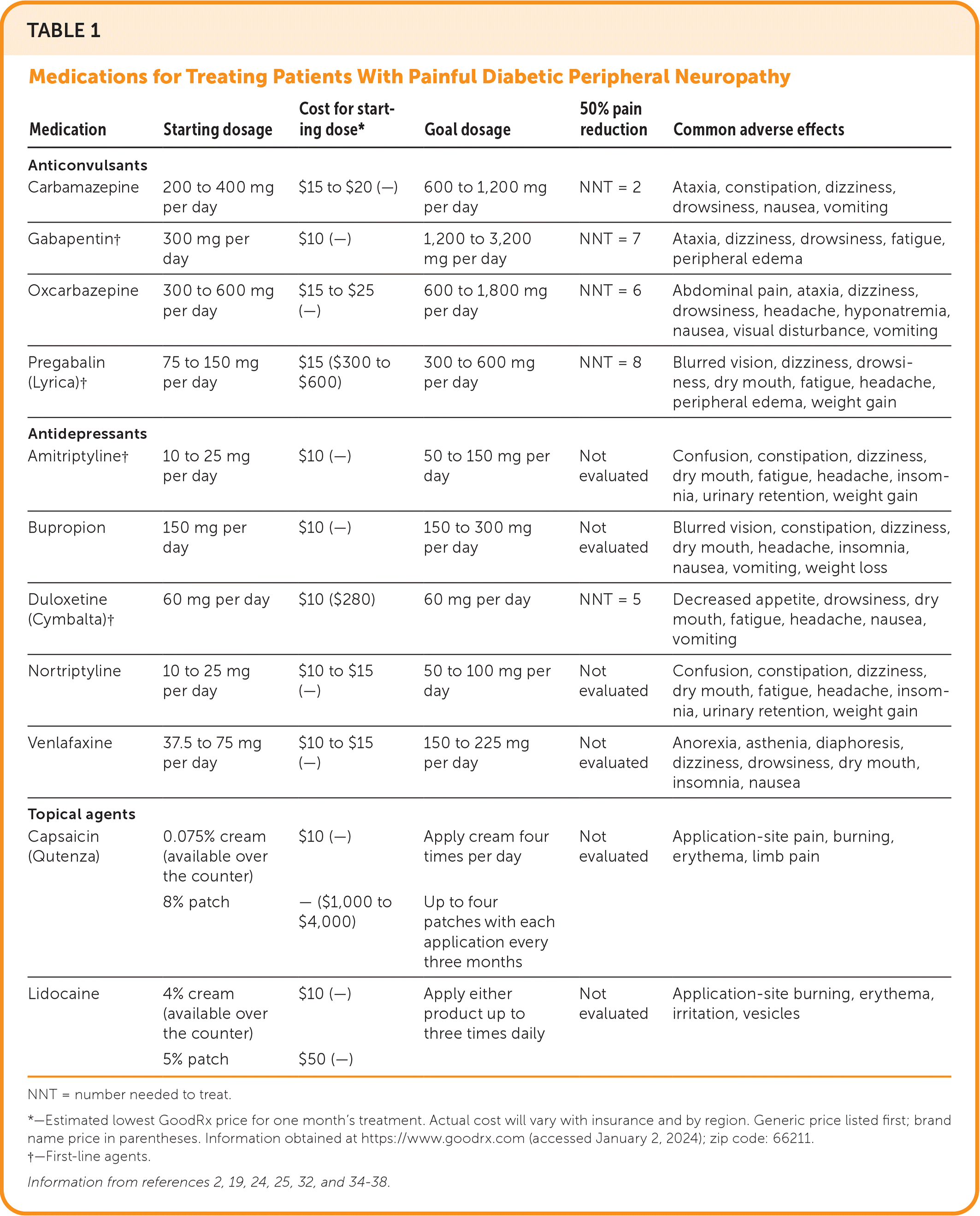
| Medication | Starting dosage | Cost for starting dose* | Goal dosage | 50% pain reduction | Common adverse effects |
|---|---|---|---|---|---|
| Anticonvulsants | |||||
| Carbamazepine | 200 to 400 mg per day | $15 to $20 (—) | 600 to 1,200 mg per day | NNT = 2 | Ataxia, constipation, dizziness, drowsiness, nausea, vomiting |
| Gabapentin† | 300 mg per day | $10 (—) | 1,200 to 3,200 mg per day | NNT = 7 | Ataxia, dizziness, drowsiness, fatigue, peripheral edema |
| Oxcarbazepine | 300 to 600 mg per day | $15 to $25 (—) | 600 to 1,800 mg per day | NNT = 6 | Abdominal pain, ataxia, dizziness, drowsiness, headache, hyponatremia, nausea, visual disturbance, vomiting |
| Pregabalin (Lyrica)† | 75 to 150 mg per day | $15 ($300 to $600) | 300 to 600 mg per day | NNT = 8 | Blurred vision, dizziness, drowsiness, dry mouth, fatigue, headache, peripheral edema, weight gain |
| Antidepressants | |||||
| Amitriptyline† | 10 to 25 mg per day | $10 (—) | 50 to 150 mg per day | Not evaluated | Confusion, constipation, dizziness, dry mouth, fatigue, headache, insomnia, urinary retention, weight gain |
| Bupropion | 150 mg per day | $10 (—) | 150 to 300 mg per day | Not evaluated | Blurred vision, constipation, dizziness, dry mouth, headache, insomnia, nausea, vomiting, weight loss |
| Duloxetine (Cymbalta)† | 60 mg per day | $10 ($280) | 60 mg per day | NNT = 5 | Decreased appetite, drowsiness, dry mouth, fatigue, headache, nausea, vomiting |
| Nortriptyline | 10 to 25 mg per day | $10 to $15 (—) | 50 to 100 mg per day | Not evaluated | Confusion, constipation, dizziness, dry mouth, fatigue, headache, insomnia, urinary retention, weight gain |
| Venlafaxine | 37.5 to 75 mg per day | $10 to $15 (—) | 150 to 225 mg per day | Not evaluated | Anorexia, asthenia, diaphoresis, dizziness, drowsiness, dry mouth, insomnia, nausea |
| Topical agents | |||||
| Capsaicin (Qutenza) | 0.075% cream (available over the counter) 8% patch | $10 (—) — ($1,000 to $4,000) | Apply cream four times per day Up to four patches with each application every three months | Not evaluated | Application-site pain, burning, erythema, limb pain |
| Lidocaine | 4% cream (available over the counter) 5% patch | $10 (—) $50 (—) | Apply either product up to three times daily | Not evaluated | Application-site burning, erythema, irritation, vesicles |
NNT = number needed to treat.
*—Estimated lowest GoodRx price for one month's treatment. Actual cost will vary with insurance and by region. Generic price listed first; brand name price in parentheses. Information obtained at https://www.goodrx.com (accessed January 2, 2024); zip code: 66211.
†—First-line agents.
A 2022 propensity-matched, retrospective study has called into question the safety of long-term use of gabapentin and pregabalin.39 In the study with 210,064 patients with diabetic peripheral neuropathy, patients taking gabapentin or pregabalin had a higher risk of cardiovascular events at five years compared with individuals not using either drug. Cardiovascular events included pulmonary embolism (hazard ratio [HR] = 1.50), peripheral vascular disease (HR = 1.37), stroke (HR = 1.31), myocardial infarction (HR = 1.25), and heart failure (HR = 1.14).39 Confounding variables could have contributed to these results, and further studies are warranted to ensure that pregabalin and gabapentin are safe long-term treatments.
Besides gabapentinoids, carbamazepine and oxcarbazepine have shown improvements in diabetic peripheral neuropathy pain. Use of carbamazepine and oxcarbazepine should be reserved for patients with pain refractory to first-line options because of concerns with tolerability, serious adverse effects, and increased monitoring demands. Follow-up within one to two weeks and then every few months for repeat laboratory testing is critical for minimizing adverse effects36,37 (Table 12,19,24,25,32,34–38 ). Other anticonvulsants (e.g., lacosamide [Vimpat], lamotrigine, topiramate, valproic acid, zonisamide) have been studied in patients with neuropathy but have not shown adequate benefit.40–44
TOPICAL MEDICATIONS
Topical lidocaine and capsaicin are second-line agents with less robust data but fewer adverse effects. Lidocaine is widely available as a 4% cream or 5% patch that can be applied three times daily. A moderate-quality study (n = 204) comparing lidocaine 5% medicated plaster with pregabalin in patients with diabetic peripheral neuropathy showed comparable rates of patients obtaining at least 50% pain relief (40.4% vs. 37.2%).45 Capsaicin is administered as a 0.075% cream four times a day or as an 8% patch (Qutenza) applied to a painful area every three months. Treatment with capsaicin 0.075% cream has weak evidence of benefit but is more affordable than the 8% patch.6,20,25 The capsaicin 8% patch was studied in one high-quality trial (n = 468) vs. standard care alone, which showed that approximately 10% more participants were much or very much improved at eight to 12 weeks.38 The capsaicin 8% patch can be used only in a physician's office and may increase pain in a minority of patients.25 Lidocaine and capsaicin should not be applied to broken skin.
OPIOIDS
A 2013 Cochrane review (14 trials, n = 845) of studies of 12-week duration or shorter evaluated the use of opioids for treatment of neuropathic pain. Data from these studies demonstrated improvement in pain in 57% of participants receiving an opioid vs. 34% of those receiving a placebo, with a number needed to treat of 6 (95% CI, 3 to 50). However, the evidence was rated as low quality because the studies showed a high risk of bias.46
Because of safety concerns, use of traditional opioids is often avoided in patients with diabetic peripheral neuropathy; however, tramadol and tapentadol are synthetic, centrally acting mu opioid agonists with serotonin-norepinephrine reuptake inhibitor activity, making them theoretically more helpful for treating diabetic peripheral neuropathy pain.2,47,48 A Cochrane review of six double-blind trials (n = 438) of tramadol demonstrated a number needed to treat of 5 (95% CI, 2.9 to 8.8) for one additional participant to experience a 50% pain reduction.47 Extended-release tapentadol (available as brand Nucynta extended-release) was approved by the U.S. Food and Drug Administration based on a 12-week double-blind randomized trial (n = 395) in which treatment demonstrated a 50% reduction in pain in 34.9% of patients (P < .001) vs. placebo.48
Any benefits of opioids should be weighed against long-term harmful effects (e.g., addiction, respiratory depression, tolerance, hyperalgesia). The American Academy of Neurology and the American Diabetes Association guidelines recommend avoiding opioids, including tramadol and tapentadol, for management of diabetic peripheral neuropathy.6,20
OTHER MEDICATIONS
Alpha-lipoic acid, an antioxidant, is a safe and tolerable option for treating diabetic peripheral neuropathy. In a 2022 systematic review of eight studies (n = 1,500), use of alpha-lipoic acid demonstrated mixed results, with three trials showing benefit and five trials showing no notable effect.49 Minimal benefits occurred when alpha-lipoic acid was used orally, whereas intravenous administration showed more consistent pain improvements.2,49
Other treatments have limited evidence of benefit. A 2019 Cochrane review looked at acetyl-L-carnitine in four studies with 907 participants but concluded that the current evidence was inconclusive for supporting the reduction of pain.50 Preliminary studies show that other treatments such as onabotulinumtoxinA (available as brand Botox) and cannabidiol (over the counter) have benefit, but high-quality studies are needed.19
NONPHARMACOLOGIC THERAPY
Exercise can improve pain and function in patients with diabetic peripheral neuropathy. The ideal duration and type of exercise has yet to be identified; aerobic, resistance, and balance exercise have all shown benefits.22 Adapting an individual's exercise regimen may be necessary to improve exercise tolerance and minimize injury concerns.
Several neuromodulatory strategies (e.g., spinal cord stimulation, transcutaneous electrical nerve stimulation) have demonstrated benefit for diabetic peripheral neuropathy, but the benefits are based on low-quality evidence. Spinal cord stimulation has demonstrated only potential benefit in case-control studies; possible limitations include adverse effects, expense, and surgical complications.17 Peripheral transcutaneous electrical nerve stimulation therapy has been studied in moderate-quality prospective trials with modest benefit compared with electromagnetic nerve stimulation with no serious adverse effects. Considering its safety and low cost, treatment with transcutaneous electrical nerve stimulation is preferable compared with other neuromodulatory techniques.21
Acupuncture has been evaluated; however, the most common outcome evaluated is nerve conduction velocity. Future acupuncture studies focused on patient-oriented outcomes are needed.51
This article updates previous articles on this topic by Snyder, et al.2; Lindsay, et al.5; and Aring, et al.52
Data Sources: Essential Evidence Plus and PubMed Clinical Queries were searched to identify systematic reviews, meta-analyses, and randomized trials. Key words included diabetic peripheral neuropathy, glycemic management, nonpharmacologic treatment, primary and secondary prevention, serotonin-norepinephrine reuptake inhibitors, tricyclic antidepressants, anticonvulsants, and topical medications. The Cochrane Database, DynaMed, and UpToDate were also searched for treatments studied in the context of diabetic peripheral neuropathy. Lexicomp was used to confirm dosing, titration, and adverse effect profiles. American Family Physician review articles and guidelines from the American Diabetes Association and American Academy of Neurology were consulted. Use of race/ethnicity was considered when writing the introduction/prevention section because disparities in diagnosis exist. No differences in treatment benefit were seen across these variables in studies we reviewed, so race/ethnicity/sexual orientation was not discussed in the treatment section. Search dates: April 30, May 3, July 2 and 20, 2023; January 18, 2024.

