Atrial fibrillation is a supraventricular arrhythmia that increases the risk of stroke and all-cause mortality. It is the most common cardiac dysrhythmia in adults in the primary care setting, and its prevalence increases with age. The U.S. Preventive Services Task Force concluded that there is insufficient evidence to assess the benefits and harms of screening asymptomatic adults older than 50 years for atrial fibrillation. Many patients with atrial fibrillation are asymptomatic, but symptoms can include palpitations, exertional dyspnea, fatigue, and chest pain. Diagnosis is based on history and physical examination findings and should be confirmed with 12-lead electrocardiography or other recording device. The initial evaluation should include transthoracic echocardiography; serum electrolyte levels; complete blood count; and thyroid, kidney, and liver function tests. Stroke risk should be assessed in patients with atrial fibrillation using the CHA2DS2-VASc score. Warfarin and direct oral anticoagulants reduce the risk of stroke by preventing atrial thrombus formation and subsequent cerebral or systemic emboli. Hemodynamically unstable patients, including those with decompensated heart failure, should be evaluated and treated emergently. Most hemodynamically stable patients should be treated initially with rate control and anticoagulation. Rhythm control, using medications or procedures, should be considered in patients with hemodynamic instability or in some patients based on risk factors and shared decision-making. Electrical cardioversion may be appropriate as first-line rhythm control. Conversion to sinus rhythm with catheter ablation may be considered in patients who are unable or unwilling to take rate or rhythm control medications long-term or if medications have been ineffective.
Atrial fibrillation (AF) is a supraventricular arrhythmia characterized by uncoordinated electrical activation of the atria leading to ineffective atrial contractions.1 It is the most common cardiac dysrhythmia in adults in the primary care setting.2 It is estimated that 3 million to 6 million adults in the United States are living with AF.2,3 Older adults are more commonly affected.1–3 Patients with AF have a higher risk of stroke, heart failure, and all-cause mortality.1,2
WHAT'S NEW ON THIS TOPIC

| In 2022, the U.S. Preventive Services Task Force concluded that there is insufficient evidence to assess the benefits and harms of screening asymptomatic adults older than 50 years for atrial fibrillation with electrocardiography, continuous monitors, or blood pressure monitoring devices. |
| Smartphones (94% sensitivity and 96% specificity) and smartwatches (94% sensitivity and 93% specificity) have similar diagnostic accuracy in detecting atrial fibrillation. |
| In some patients with atrial fibrillation and heart failure, catheter ablation may improve left ventricular ejection fraction. |
SORT: KEY RECOMMENDATIONS FOR PRACTICE
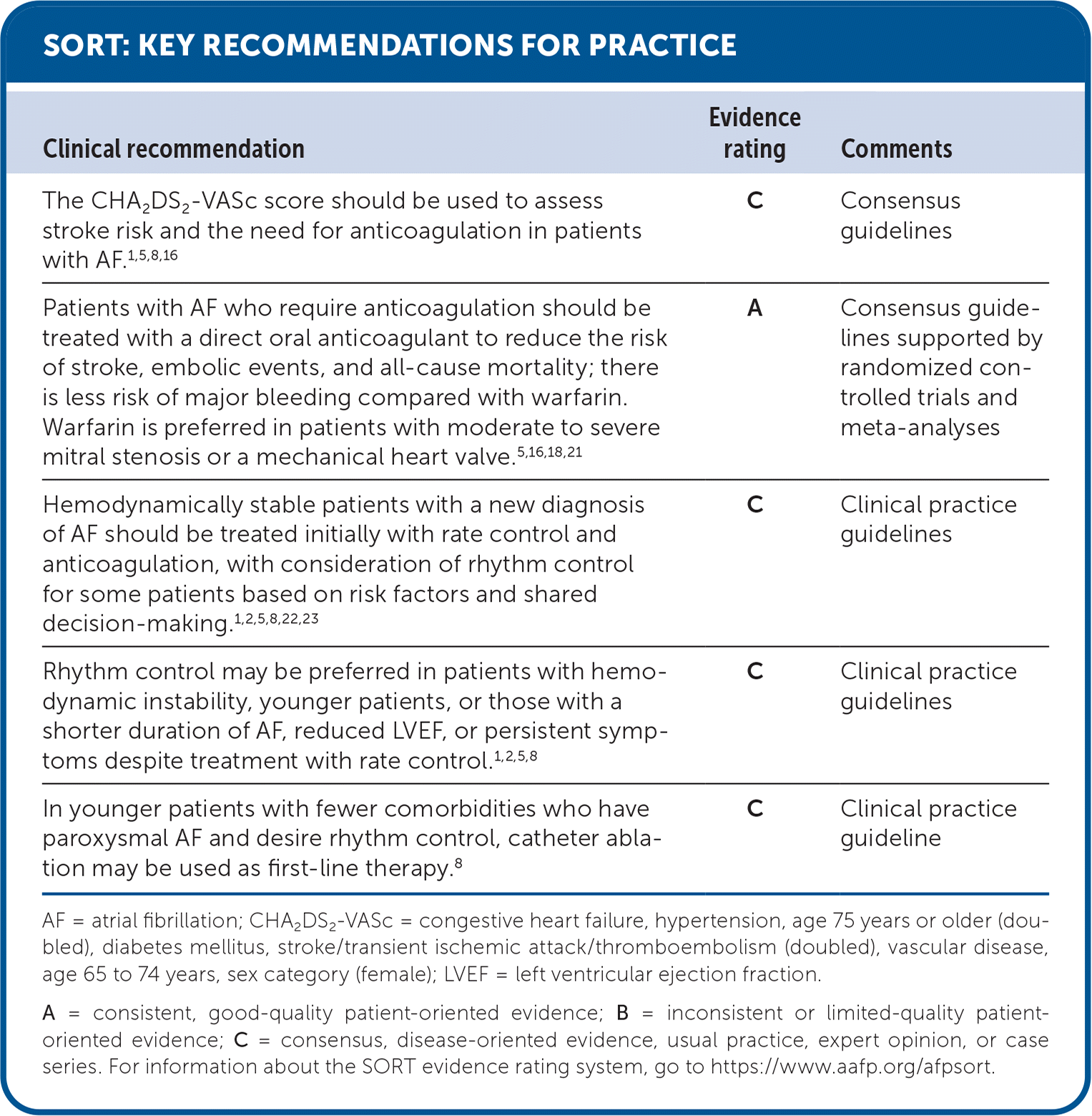
AF = atrial fibrillation; CHA2DS2-VASc = congestive heart failure, hypertension, age 75 years or older (doubled), diabetes mellitus, stroke/transient ischemic attack/thromboembolism (doubled), vascular disease, age 65 to 74 years, sex category (female); LVEF = left ventricular ejection fraction.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
What Clinical History or Symptoms Suggest AF?
Many patients with AF are asymptomatic, but the most common presenting symptom is palpitations.1,4 Other common symptoms include dyspnea on exertion, fatigue, dizziness, and chest pain.4,5 Patients with symptoms and associated risk factors should be evaluated for AF. The U.S. Preventive Services Task Force concluded that there is insufficient evidence to assess the benefits and harms of screening for AF in asymptomatic adults older than 50 years.
EVIDENCE SUMMARY
Approximately 50% to 87% of patients with AF are asymptomatic.5 The most common presenting symptom in patients with AF is palpitations, which occur in approximately one-third of symptomatic patients.1,4,5 Dyspnea on exertion, fatigue, dizziness, and chest pain are other common symptoms.5 Patients may be hemodynamically unstable and present with symptoms that include syncope, hypotension, heart failure, and myocardial ischemia due to a very rapid heartbeat caused by AF.5
The U.S. Preventive Services Task Force concluded that there is insufficient evidence to assess the benefits and harms of screening for AF with electrocardiography (ECG), continuous monitors such as a Holter monitor, or blood pressure monitoring devices in adults older than 50 years.6 A Cochrane review showed that systematic screening (i.e., screening all patients with ECG) and opportunistic screening (i.e., pulse palpation during an office visit for any reason, followed by ECG if pulse is irregular) are more effective in patients older than 65 years than the regular practice of diagnosing AF based on clinical presentation, but systematic screening is less cost-effective.7
Age is the most important risk factor for the development of AF, with the greatest risk in those older than 50 years.3,5 Patients older than 70 years are seven times more likely to develop AF than those 50 years or younger.3,4 Other common risk factors include obesity, hypertension, diabetes mellitus, smoking, myocardial infarction, heart failure, and obstructive sleep apnea.3,5
What Is Included in the Initial Evaluation of AF?
Diagnosis of AF is based on history and physical examination findings and should be confirmed with 12-lead ECG or other recording device.1,5,8 The initial evaluation should include transthoracic echocardiography; serum electrolyte levels; complete blood count; and thyroid, kidney, and liver function tests.1
EVIDENCE SUMMARY
A thorough history should be obtained from patients with suspected AF to characterize the pattern and duration of symptoms; determine potential causes; assess thromboembolic risk; review prior treatment, family history, and associated conditions; and identify reversible risk factors.1,5 The physical examination can verify the diagnosis with palpation or auscultation of an irregularly irregular heartbeat. Detection of an irregularly irregular radial pulse has a sensitivity and specificity of 92% and 82%, respectively, for diagnosing AF.9
The diagnostic standard for confirming AF is 12-lead ECG1,5,10 (Figure 111 ). Patients with paroxysmal AF who are in normal sinus rhythm at the time of presentation may require ambulatory ECG, such as a Holter monitor. Other options for ambulatory ECG monitoring include post-event, implantable, or external loop recorders.9 Newer detection methods include handheld ECG devices, wearable wrist devices (e.g., Apple Watch), and automatic blood pressure monitors.10 One meta-analysis found that the most accurate devices for detecting pulse irregularities caused by AF are non–12-lead ECG (91% sensitivity and 95% specificity) and blood pressure monitors (98% sensitivity and 92% specificity).9 Smartphones (94% sensitivity and 96% specificity) and smartwatches (94% sensitivity and 93% specificity) have similar diagnostic accuracy in detecting AF.12
FIGURE 1.
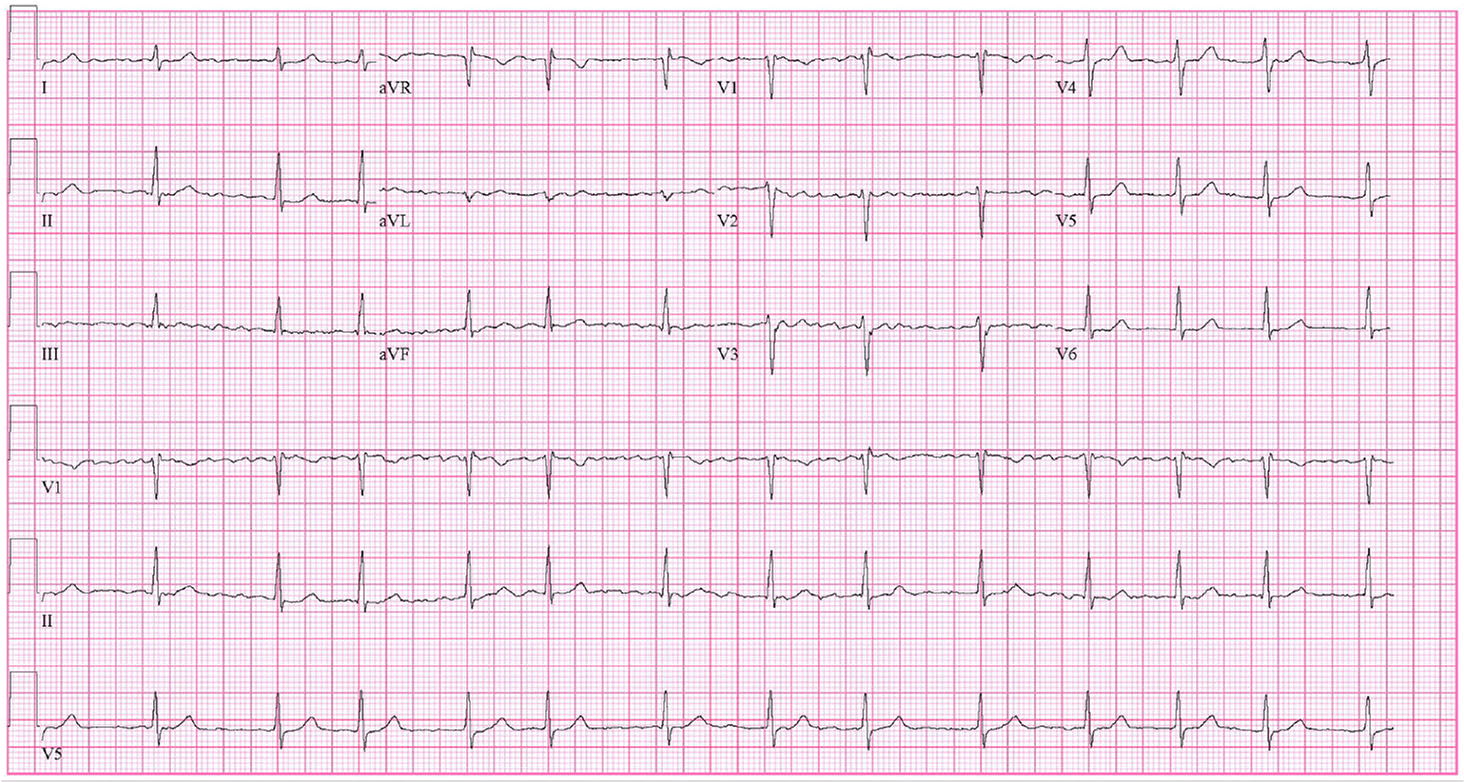
Twelve-lead electrocardiography showing atrial fibrillation. Note the absence of distinct P waves, chaotic activity of atria, and irregular R-R intervals with narrow QRS complex.
Reprinted with permission from Gutierrez C, Blanchard DG. Diagnosis and treatment of atrial fibrillation. Am Fam Physician. 2016;94(6):444.
Transthoracic echocardiography should be performed to evaluate for underlying structural heart disease and to assess cardiac function and atrial size.1,5 Transesophageal echocardiography is the most sensitive and specific modality for the evaluation of left atrial thrombus but is not routinely indicated.1 A new diagnosis of AF should prompt measurements of serum electrolyte levels; complete blood count; and thyroid, kidney, and liver function tests to assess for potential causes and guide treatment.1,5
How Is Stroke Risk Assessed and Reduced With Anticoagulation?
In patients with AF, stroke risk should be assessed with the CHA2DS2-VASc (congestive heart failure, hypertension, age 75 years or older [doubled], diabetes mellitus, stroke/transient ischemic attack/thromboembolism [doubled], vascular disease, age 65 to 74 years, sex category [female]) score1,2,5,13 (Table 11). Warfarin and direct oral anticoagulants (DOACs) reduce the risk of stroke by preventing atrial thrombus formation and subsequent cerebral or systemic emboli.1
TABLE 1. CHA2DS2-VASc Risk Stratification Score for Patients With Nonvalvular Atrial Fibrillation
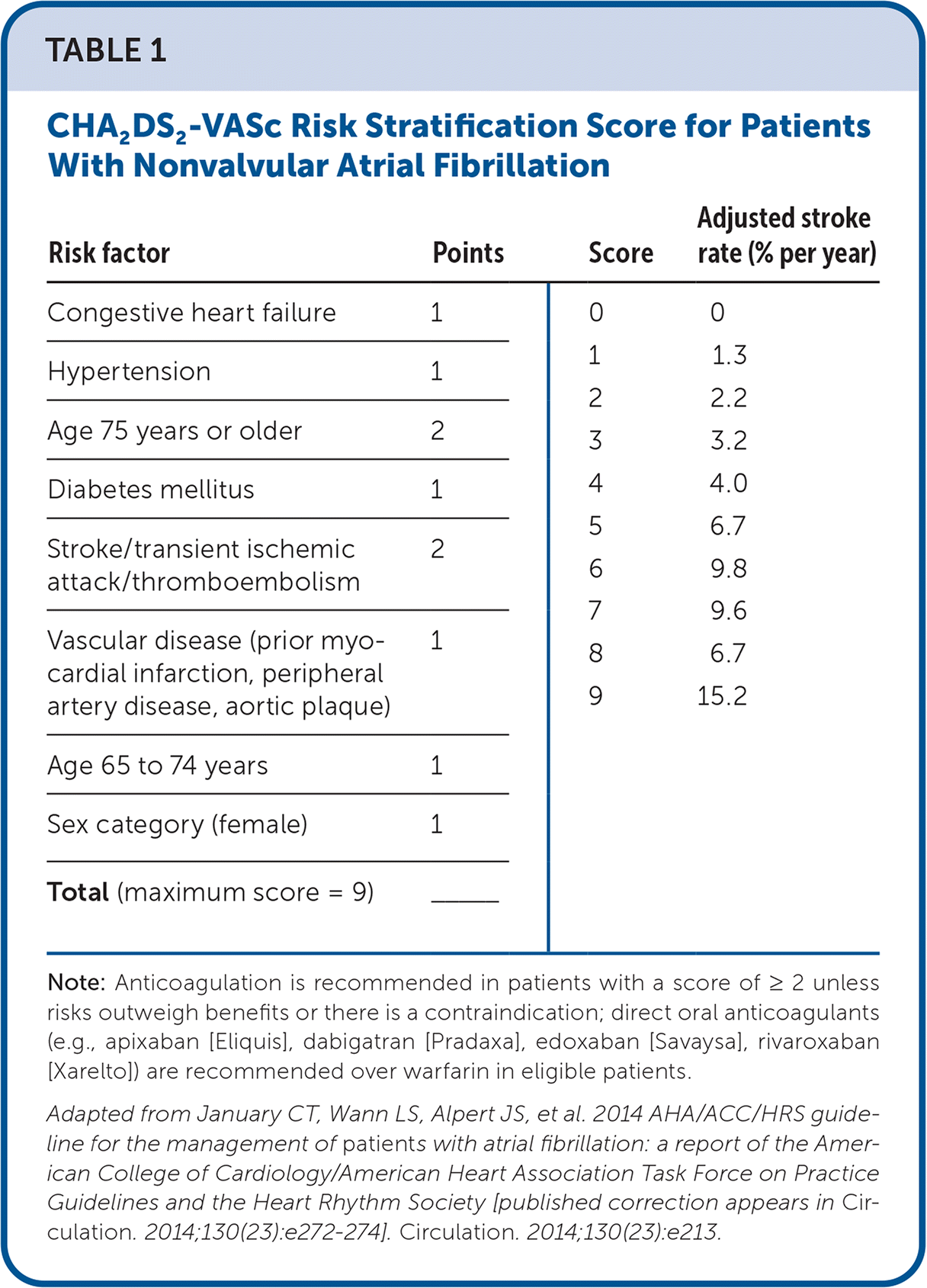
| Risk factor | Points | Score | Adjusted stroke rate (% per year) |
|---|---|---|---|
| Congestive heart failure Hypertension Age 75 years or older Diabetes mellitus Stroke/transient ischemic attack/thromboembolism Vascular disease (prior myocardial infarction, peripheral artery disease, aortic plaque) Age 65 to 74 years Sex category (female) Total (maximum score = 9) | 1 1 2 1 2 1 1 1 _____ | 0 1 2 3 4 5 6 7 8 9 | 0 1.3 2.2 3.2 4.0 6.7 9.8 9.6 6.7 15.2 |
Note: Anticoagulation is recommended in patients with a score of ≥ 2 unless risks outweigh benefits or there is a contraindication; direct oral anticoagulants (e.g., apixaban [Eliquis], dabigatran [Pradaxa], edoxaban [Savaysa], rivaroxaban [Xarelto]) are recommended over warfarin in eligible patients.
Adapted from January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patient s with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society [published correction appears in Circulation. 2014;130(23):e272–274]. Circulation. 2014;130(23):e213.
EVIDENCE SUMMARY
The CHA2DS2-VASc score should be used to determine the risk of embolic stroke and the need for anticoagulation.1,5,8,14–16 Males with a score of 2 or greater and females with a score of 3 or greater generally benefit from oral anticoagulation.1,14–16 Before initiation of anticoagulation therapy, bleeding risk should be assessed. Traditionally, the HAS-BLED (hypertension, abnormal renal and liver function, stroke, bleeding risk, labile international normalized ratio, elderly [older than 65 years], drug and alcohol use) score is used.1,5 A score of more than 3 indicates high risk of bleeding. A DOAC score has been developed to predict bleeding risk for patients with AF who are using DOACs. Although the DOAC score showed superior performance to the HAS-BLED score in two validation cohorts, it has not been incorporated into AF guidelines.17 Bleeding risk should be part of the discussion of risks vs. benefits of anticoagulation, and a high risk score alone should not prohibit use.5,16
Warfarin, a vitamin K antagonist, and DOACs, including direct thrombin inhibitors and factor Xa inhibitors, reduce stroke risk in AF. Warfarin is the only oral anticoagulant recommended for the treatment of AF in patients with moderate to severe mitral stenosis or a mechanical heart valve.5,14,18 DOACs are recommended over warfarin in patients with AF who are eligible for treatment.5,14–16,19,20 A 2018 Cochrane review concluded that DOACs significantly decreased the risk of stroke, systemic embolic events, major bleeding, and all-cause deaths compared with warfarin.21 The recommended DOACs for use in nonvalvular AF are apixaban (Eliquis), dabigatran (Pradaxa), edoxaban (Savaysa), and rivaroxaban (Xarelto).2,16 Aspirin is not recommended for reduction of stroke risk in patients with AF. Table 2 lists the pharmacologic properties of anticoagulants used for the prevention of thromboembolism in patients with AF.11 Left atrial appendage occlusion (e.g., with the Watchman device) is an alternative for patients with an increased risk of stroke who have contraindications to long-term anticoagulation.16
What Is the Initial Management of AF?
Patients who are hemodynamically unstable, including those with decompensated heart failure, should be evaluated and treated emergently with airway and blood pressure support. Admission to the intensive care unit and cardiology consultation should be considered.1,2,5 Most hemodynamically stable patients should be treated initially with rate control and anticoagulation, with consideration of rhythm control for some patients based on risk factors and shared decision-making.1,2,5,8,22,23
EVIDENCE SUMMARY
Hospitalization is indicated for patients with unstable arrhythmia, acute myocardial infarction, altered mental status, decompensated heart failure, hypotension, or intolerable symptoms.1,2,5 Nondihydropyridine calcium channel blockers and beta blockers are preferred for achieving rate control.1,5,22 Lenient rate control (less than 110 beats per minute at rest) has similar benefits as strict control (less than 80 beats per minute at rest) in patients without symptoms at higher heart rates and with normal left ventricular ejection fraction (LVEF).1,5,24 Digoxin may be considered as a second-line agent for rate control, particularly in patients with heart failure.1,5,25
Goals of treatment are to control symptoms and to prevent thromboembolism and long-term cardiovascular morbidity from tachycardia. Addressing risk factors such as obesity, sleep apnea, hypertension, diabetes, and alcohol consumption can reduce episodes and symptoms.5,26–28
Which Patients Are Good Candidates for Pharmacologic or Electrical Cardioversion?
Rhythm control may be preferred in patients with hemodynamic instability, younger patients, and those with a shorter duration of AF, reduced LVEF, or persistent symptoms despite treatment with rate control.1,2,5,8 Electrical cardioversion may be appropriate as first-line rhythm control.8
EVIDENCE SUMMARY
Select patients with new-onset AF (i.e., less than 48 hours and therefore lower risk of left atrial thrombus) or those with conditions including decompensated heart failure, acute coronary syndrome, hypotension, or acute stroke may benefit from immediate pharmacologic or electrical cardioversion. In hemodynamically stable patients, delaying cardioversion until after 4 weeks of anticoagulation is noninferior to immediate cardioversion for a return to sinus rhythm.29 However, emergent cardioversion should not be delayed in patients with hemodynamic instability.16,18,21
Older studies have found that a rhythm control strategy had similar outcomes as rate control for cardiovascular mortality, stroke, and all-cause mortality.1,2,5,23 More recent data suggest that cardioversion within 1 year of AF onset may be associated with lower mortality and no increase in adverse effects that require hospitalization.30 Additionally, antiarrhythmic medication may reduce cardiovascular morbidity and mortality in patients younger than 65 years compared with rate control.31 A variety of antiarrhythmic medications with equal effectiveness are available; therefore, adverse effects should guide choice.
Class IC antiarrhythmics, including flecainide and propafenone, are contraindicated in patients with ischemic heart disease, structural heart disease, and reduced LVEF. During initiation of these medications, QRS intervals should be monitored continuously. Class III antiarrhythmics, including sotalol and dofetilide, can prolong QT intervals and cause torsades de pointes. Patients must be monitored in the hospital for the first five doses of these medications. ECG should be repeated at least every 6 months. Beta blockers and calcium channel blockers should be used with caution when taken with class III antiarrhythmics due to an increased effect of medication at lower heart rates. Kidney function and potassium and magnesium levels should be monitored in all patients taking antiarrhythmic medications.
Amiodarone is the most prescribed antiarrhythmic in the United States, although it is considered a second-line agent due to its numerous adverse effects, including permanent liver and lung toxicity, thyroid dysfunction, and skin sensitivity. In patients taking amiodarone for longer than 3 months, ECG; thyroid, liver, and pulmonary function tests; chest radiography; and ophthalmologic and skin examinations should be performed. Thyroid and liver functions should be reassessed every 6 months. ECG, chest radiography, ophthalmologic examination, and pulmonary function tests should be repeated every 6 to 12 months. A pill in pocket approach, in which a single dose of propafenone or flecainide is taken with onset of symptoms, can be considered for some patients with infrequent episodes of paroxysmal AF.1,5,32–34
Which Patients Are Good Candidates for Catheter Ablation?
In younger patients with few comorbidities who have paroxysmal AF and desire rhythm control, catheter ablation may be used as first-line therapy.8 Conversion to sinus rhythm with catheter ablation may be considered in patients who are unable or unwilling to take long-term medications for rate or rhythm control or if medications have been ineffective.1,5,16,35
EVIDENCE SUMMARY
In some patients with AF and heart failure, catheter ablation may improve LVEF.16,35,36 Ablation is more effective at reducing recurrence of AF compared with medication alone.37,38 Up to 50% of patients may experience a temporary recurrence of AF within 3 months due to irritation from the procedure. Up to 40% of patients have continued symptoms after 3 months and may require repeat ablation. More than 50% of patients remain in sinus rhythm 3 years after ablation. The procedure is more likely to be successful in younger patients, those with paroxysmal AF, and those without structural heart disease. Patients must be able to tolerate pharmacologic anticoagulation for at least 4 weeks before and 2 months after ablation regardless of their stroke risk.35 Although ablation may eliminate the need for rate or rhythm control medications, anticoagulation should be continued indefinitely if indicated based on stroke risk.1,16,18,35
Indications for referral to cardiology include failure of rate control (in which case rhythm control should be considered) or uncontrolled symptomatic AF burden.1
This article updates previous articles on this topic by Gutierrez and Blanchard11; Gutierrez and Blanchard39; King, et al.40; King, et al.41; Dell’Orfano, et al.42; and Akhtar, et al.43
Data Sources: A PubMed search was completed in Clinical Queries using the terms atrial fibrillation, rate control, rhythm control, ablation therapy on nonvalvular atrial fibrillation, and anticoagulation therapy for nonvalvular atrial fibrillation. We also searched Essential Evidence Plus, the Cochrane Library, and guidelines on the management of atrial fibrillation from the American Heart Association/American College of Cardiology/ Heart Rhythm Society and European Society of Cardiology. The search focused on randomized controlled trials, systematic reviews, meta-analyses, and reviews. Search dates: April to July 2023, November 2023, and February 2024.

