Treatment-resistant depression is defined as absence of remission despite trials of two or more antidepressant medications and can occur in up to 31% of patients with major depressive disorder. Partial response to treatment is defined as less than 50% reduction in depression-rating scores. Before diagnosing treatment-resistant depression or partial response to treatment, adherence to adequate doses and duration of medications should be confirmed. Management strategies include adding psychotherapy, switching antidepressant medication class, or augmenting with additional medications. Current guidelines recommend augmentation with a second-generation antidepressant, an atypical antipsychotic, tricyclic antidepressants, lithium, or a triiodothyronine medication as pharmacologic options. Ketamine and esketamine can also be used as augmentation for treatment-resistant depression and may help reduce suicidal ideation. Electroconvulsive therapy and repetitive transcranial magnetic stimulation may be effective. Pharmacogenetic testing has limited evidence and is not recommended. Nonpharmacologic therapies include psychotherapy, exercise, and focused dietary changes.
Epidemiology
Depression is among the most common psychiatric disorders in the United States. Major depressive disorder has a 12-month prevalence of 10% and a lifetime prevalence of 21%, with an average onset at 30 years of age.1 Only about one-third of patients respond to an initial trial of a first-line antidepressant medication, and a significant proportion of patients will not respond despite multiple interventions.2 Although it lacks a universal definition, treatment-resistant depression is most commonly defined as absence of remission using validated scoring tools despite trials of two or more antidepressant medications at adequate dose, duration, and adherence.3,4 This definition, however, does not account for partial response to treatment, which is defined as less than 50% reduction in depression-rating scores. When applied to treatment, these often overlapping criteria can make comparing and synthesizing studies difficult.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
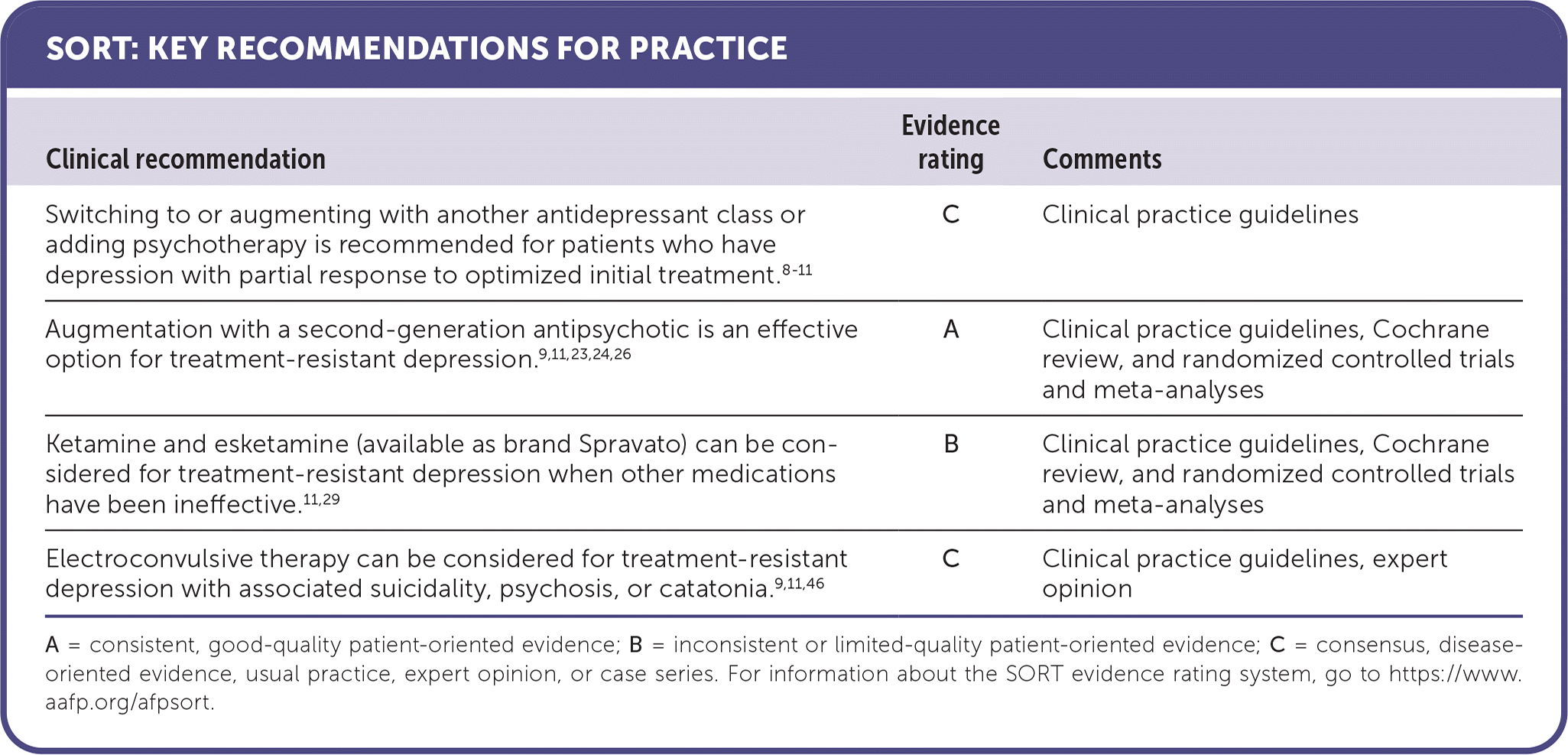
| Clinical recommendation | Evidence rating | Comments |
|---|---|---|
| Switching to or augmenting with another antidepressant class or adding psychotherapy is recommended for patients who have depression with partial response to optimized initial treatment.8–11 | C | Clinical practice guidelines |
| Augmentation with a second-generation antipsychotic is an effective option for treatment-resistant depression.9,11,23,24,26 | A | Clinical practice guidelines, Cochrane review, and randomized controlled trials and meta-analyses |
| Ketamine and esketamine (available as brand Spravato) can be considered for treatment-resistant depression when other medications have been ineffective.11,29 | B | Clinical practice guidelines, Cochrane review, and randomized controlled trials and meta-analyses |
| Electroconvulsive therapy can be considered for treatment-resistant depression with associated suicidality, psychosis, or catatonia.9,11,46 | C | Clinical practice guidelines, expert opinion |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Before diagnosing treatment-resistant depression or partial response to treatment, patients should be assessed to verify the accuracy of the depression diagnosis and evaluated for comorbid or exacerbating medical or psychiatric conditions. Adherence to medications should also be confirmed. Diagnosis of bipolar disorder should specifically be considered; patients should be screened for symptoms of mania or hypomania because up to 25% of patients presenting with depression or anxiety in a primary care setting are diagnosed with bipolar disorder.5 Particular attention should be paid to medication adherence because nonadherence rates for antidepressant medications has been estimated to be 46%.6 The prevalence of treatment-resistant depression among patients with major depressive disorder is 31%.7 The general approach to treatment-resistant depression and partial response to treatment includes adding psychotherapy, switching antidepressant medication class, or augmenting with additional medications.8–11
Risk Factors and Comorbid Conditions
Risk factors for treatment resistance include total burden of medical disease, substance use disorder, comorbid anxiety disorders, personality disorder, and early age of onset of depression.12–14 Childhood trauma has also been associated with treatment resistance.15 Many organic medical conditions—from cardiovascular disease to nutritional deficiencies—can also cause and mimic depression.16,17 Substance use disorder and other comorbid psychiatric disorders should be assessed and treated because they can mimic or exacerbate depression and affect response to treatment and attainment of remission.
Pharmacologic Treatment Strategies
PHARMACOGENETIC GUIDANCE
Pharmacogenetic analyses use genetic testing to match patients to treatments. A 2023 systematic review and meta-analysis on the effectiveness of pharmacogenetics for major depressive disorder included 11 randomized controlled trials (n = 5,347).18 The analysis compared usual care vs. pharmacogenetic-guided care for medication choice. In this review, use of pharmacogenetic testing increased response and remission rates at 8 and 12 weeks, although no differences were seen at 4 and 24 weeks. The review also noted a decrease in serious adverse events for patients who received pharmacogenetic testing; however, the cost-effectiveness of testing remains unclear.18 For now, assessment of factors such as cost, adverse effects, and severity or urgency of symptoms should guide the treatment approach.
SWITCHING AND AUGMENTATING MEDICATIONS
Multiple studies have assessed a variety of strategies for pharmacologic treatment for patients who partially respond to initial treatment with a first-line antidepressant (e.g., selective serotonin reuptake inhibitor, selective norepinephrine reuptake inhibitor, bupropion, mirtazapine, trazodone, vilazodone [Viibryd], vortioxetine [available as brand Trintellix]) and for those with treatment-resistant depression.11,19 Strategies include switching or augmenting current therapy. Switching medications includes changes within the same class of medications or across classes. Augmentation refers to adding an antidepressant or nonantidepressant medication to the patient's current regimen.3,8
Multiple guidelines address care of patients who have inadequate or partial response to initial treatment with a first-line antidepressant, but only two specifically address treatment-resistant depression.8–11,19 Switching medications is a reasonable strategy, especially for patients experiencing adverse effects or intolerance to the initial antidepressant or lack of response after appropriate titration over 4 to 12 weeks.3,8–11,19 If switching medications does not show improvement in patient responses to depression-rating scoring, a reasonable next step is consideration of augmentation with an additional medication. Treatment options include second-generation antidepressants, second-generation antipsychotics, buspirone, tricyclic antidepressants, lithium, triiodothyronine (synthetic T3 augmentation), ketamine, and esketamine (available as brand Spravato).8–11,19 Table 1 lists medications used for augmentation, their doses, and common adverse effects.8,11 Figure 1 provides management options for treatment-resistant depression.10,11
TABLE 1. Medications for Augmenting Depression Treatment
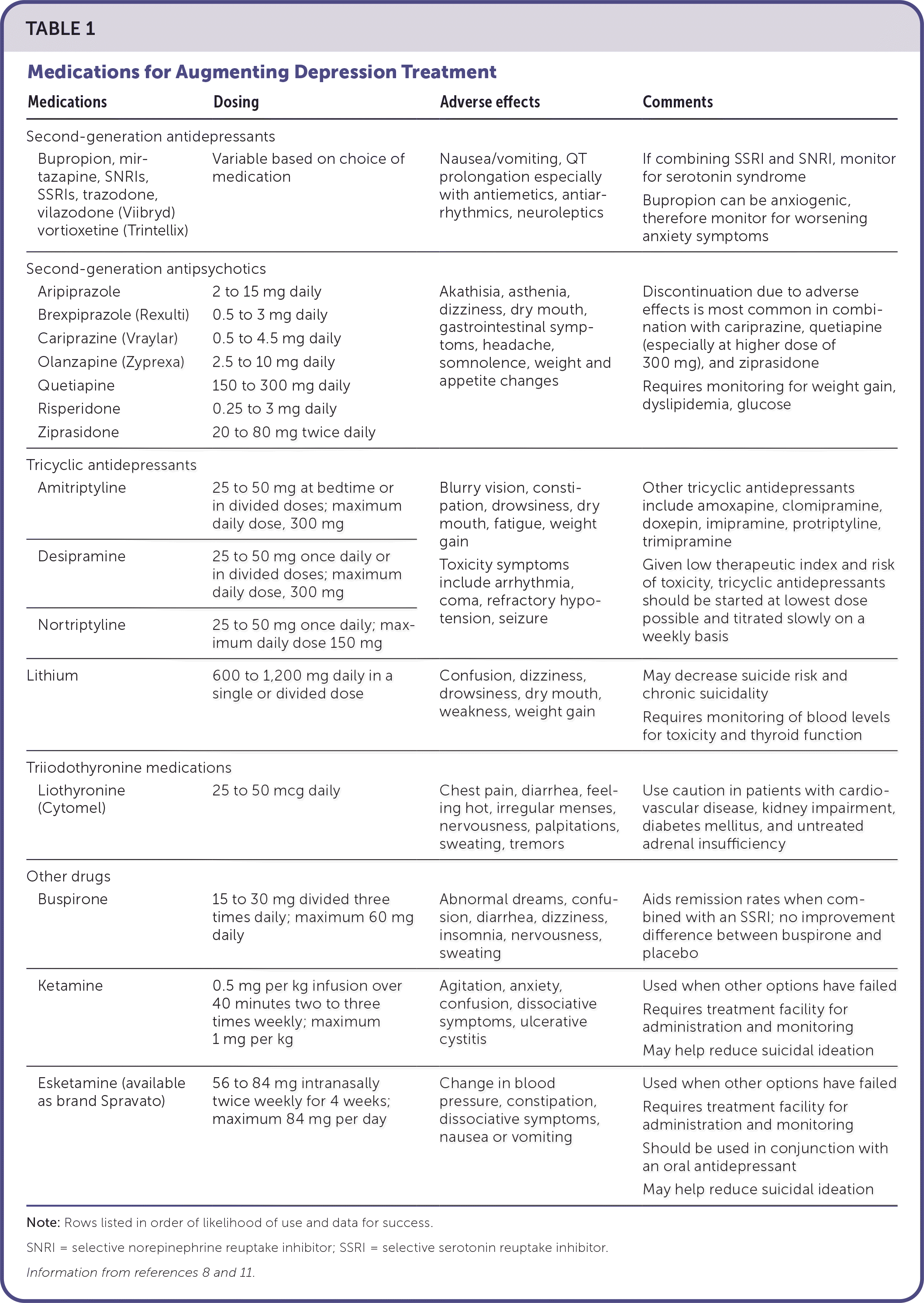
| Medications | Dosing | Adverse effects | Comments |
|---|---|---|---|
| Second-generation antidepressants | |||
| Bupropion, mirtazapine, SNRIs, SSRIs, trazodone, vilazodone (Viibryd) vortioxetine (Trintellix) | Variable based on choice of medication | Nausea/vomiting, QT prolongation especially with antiemetics, antiarrhythmics, neuroleptics | If combining SSRI and SNRI, monitor for serotonin syndrome Bupropion can be anxiogenic, therefore monitor for worsening anxiety symptoms |
| Second-generation antipsychotics | |||
| Aripiprazole Brexpiprazole (Rexulti) Cariprazine (Vraylar) Olanzapine (Zyprexa) Quetiapine Risperidone Ziprasidone | 2 to 15 mg daily 0.5 to 3 mg daily 0.5 to 4.5 mg daily 2.5 to 10 mg daily 150 to 300 mg daily 0.25 to 3 mg daily 20 to 80 mg twice daily | Akathisia, asthenia, dizziness, dry mouth, gastrointestinal symptoms, headache, somnolence, weight and appetite changes | Discontinuation due to adverse effects is most common in combination with cariprazine, quetiapine (especially at higher dose of 300 mg), and ziprasidone Requires monitoring for weight gain, dyslipidemia, glucose |
| Tricyclic antidepressants | |||
| Amitriptyline | 25 to 50 mg at bedtime or in divided doses; maximum daily dose, 300 mg | Blurry vision, constipation, drowsiness, dry mouth, fatigue, weight gain Toxicity symptoms include arrhythmia, coma, refractory hypotension, seizure | Other tricyclic antidepressants include amoxapine, clomipramine, doxepin, imipramine, protriptyline, trimipramine Given low therapeutic index and risk of toxicity, tricyclic antidepressants should be started at lowest dose possible and titrated slowly on a weekly basis |
| Desipramine | 25 to 50 mg once daily or in divided doses; maximum daily dose, 300 mg | ||
| Nortriptyline | 25 to 50 mg once daily; maximum daily dose 150 mg | ||
| Lithium | 600 to 1,200 mg daily in a single or divided dose | Confusion, dizziness, drowsiness, dry mouth, weakness, weight gain | May decrease suicide risk and chronic suicidality Requires monitoring of blood levels for toxicity and thyroid function |
| Triiodothyronine medications | |||
| Liothyronine (Cytomel) | 25 to 50 mcg daily | Chest pain, diarrhea, feeling hot, irregular menses, nervousness, palpitations, sweating, tremors | Use caution in patients with cardiovascular disease, kidney impairment, diabetes mellitus, and untreated adrenal insufficiency |
| Other drugs | |||
| Buspirone | 15 to 30 mg divided three times daily; maximum 60 mg daily | Abnormal dreams, confusion, diarrhea, dizziness, insomnia, nervousness, sweating | Aids remission rates when combined with an SSRI; no improvement difference between buspirone and placebo |
| Ketamine | 0.5 mg per kg infusion over 40 minutes two to three times weekly; maximum 1 mg per kg | Agitation, anxiety, confusion, dissociative symptoms, ulcerative cystitis | Used when other options have failed Requires treatment facility for administration and monitoring May help reduce suicidal ideation |
| Esketamine (available as brand Spravato) | 56 to 84 mg intranasally twice weekly for 4 weeks; maximum 84 mg per day | Change in blood pressure, constipation, dissociative symptoms, nausea or vomiting | Used when other options have failed Requires treatment facility for administration and monitoring Should be used in conjunction with an oral antidepressant May help reduce suicidal ideation |
Note: Rows listed in order of likelihood of use and data for success.
SNRI = selective norepinephrine reuptake inhibitor; SSRI = selective serotonin reuptake inhibitor.
FIGURE 1.
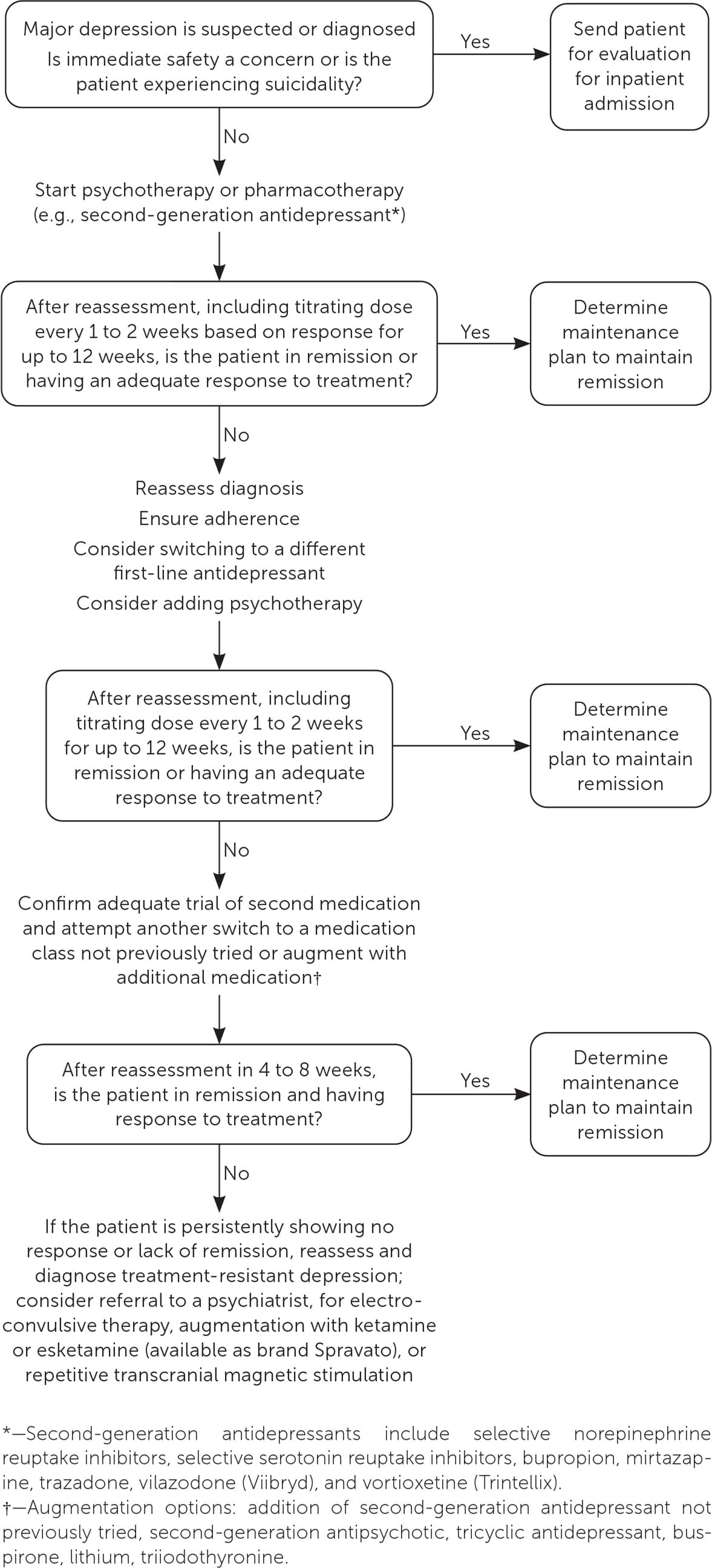
Treatment strategy for depression with partial response to treatment and treatment-resistant depression.
Second-generation Antidepressants. Although second-generation antidepressants are often used when treating depression, studies have suggested that neither dose escalation nor within-class switching is effective.20,21 Augmentation by adding a second-generation antidepressant with a different mechanism of action may have some benefit. A 2023 study suggests that bupropion augmentation may be as effective as augmentation with aripiprazole in older adults (60 years or older) with treatment-resistant depression.22
Buspirone. Evidence regarding busprirone augmentation is mixed. The Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial noted that the addition of buspirone aided remission rates when combined with a selective serotonin reuptake inhibitor.2 An additional study showed no difference in improvement between buspirone and placebo.23
Second-generation Antipsychotics. Multiple systematic reviews demonstrate the benefit of augmentation with second-generation antipsychotics, (i.e., atypical antipsychotics). Second-generation antipsychotics include aripiprazole, brexpiprazole (available as brand Rexulti), cariprazine (available as brand Vraylar), olanzapine (Zyprexa), quetiapine, risperidone, and ziprasidone. Reviews from 2015 and 2021 noted that augmentation with aripiprazole improved response rates (number needed to treat [NNT] = 8) and increased remission rates for depression; however, a separate review noted weak evidence for its use.22,24–26 In older adults (i.e., 60 years or older), aripiprazole augmentation improved well-being scores and remission rates in treatment-resistant depression.22 A 2019 review noted reduced depression-rating scores at 8 to 12 weeks with cariprazine (NNT = 10), quetiapine (NNT = 9), and ziprasidone (NNT = 7) compared with placebo. Olanzapine had low-quality evidence for its use, and only quetiapine demonstrated increased remission rates (NNT = 9).23 Augmentation with brexpiprazole, a third-generation antipsychotic, also improved response and remission rates.26,27
Tricyclic Antidepressants. Before development of second-generation antidepressants, tricyclic antidepressants were considered a first-line option for treatment-resistant depression. A 2018 network meta-analysis noted modest evidence that amitriptyline reduces depression symptoms by more than 50% at 8 weeks.28 Because of risk of toxicity, tricyclic antidepressants are no longer recommended as first-line agents, but their use can be beneficial for patients who partially respond to other medication classes or have treatment-resistant depression.
Lithium. Adding lithium, a mood stabilizer, has also been studied as an augmentation strategy for partial response to treatment or treatment-resistant depression; however, evidence is mixed. A comprehensive review of open-label and randomized controlled trials of lithium augmentation demonstrated mixed results, with some studies noting improved response rates and remission and others no improvement.26 Most of these studies, however, used lithium as add-on therapy to tricyclic antidepressants.26 The results of systematic reviews have also been mixed for response and remission rates, and discontinuation rates are high compared with placebo.23,24 Lithium may decrease suicide risk, but the benefit should be weighed against known toxicity, need for monitoring, and other drug interactions.26
Triiodothyronine. Synthetic T3 augmentation (liothyronine [Cytomel]) was effective in improving remission rates in the STAR*D trial, which evaluated various treatment steps in depression management.2 This effect was irrespective of any underlying thyroid abnormality. Synthetic T3 is typically better tolerated than lithium; however, concerns for hyper-thyroid adverse effects often limit its use in clinical practice.2
Ketamine. A 2021 Cochrane review evaluated the use of the N-methyl-d-aspartate receptor antagonists ketamine and esketamine in treatment-resistant depression.29 In the review, 31 studies evaluated ketamine compared with placebo and other pharmacologically active drugs.29 Overall, ketamine increased response and remission of depression and included a reduction in depression-rating scores at 24 hours; however, the evidence quality was low, and effects were limited to 7 days, dissipating after 2 weeks.29 Ketamine has been shown to decrease suicidal ideation in patients with depression, thus providing a stabilizing option for patients needing emergent intervention.30,31
Esketamine. Administered intranasally, esketamine was more effective than placebo in terms of response rate at 24 hours and at 1, 2, and 4 weeks.30 Esketamine decreased depression-rating scores at 24 and 72 hours and at 1, 2, and 4 weeks; however, no difference was noted at 3 months.30 A 2023 randomized controlled trial of patients with treatment-resistant depression who were taking a selective serotonin reuptake inhibitor or a selective norepinephrine reuptake inhibitor compared esketamine augmentation with quetiapine augmentation.32 The trial found that esketamine augmentation improved remission rates (27.1% vs. 17.6%) after 8 weeks without relapse at 32 weeks.32
Other Medications. Many other medications have been studied and used for partial response to treatment and treatment-resistant depression. These include lamotrigine, lisdexamfetamine (Vyvanse), methylphenidate, modafinil, and pindolol; the evidence supporting their use is mixed. Psilocybin, a hallucinogenic, has been a focus of ongoing research for treatment-resistant depression. A phase 2 trial published in 2022 noted reduced depression-rating scores with a single 25-mg dose for patients with treatment-resistant depression, although adverse effects were common.33 Other dosing regimens (e.g., 1 mg daily for 3 weeks, single 10-mg dose) did not show the same improvement in depression-rating scores.33 Another study of a single 25-mg dose of psilocybin found improved symptoms scores for up to 6 weeks but not improved remission compared with the control group.34 Therefore, more research of longer duration and with comparisons are needed.33,34 Cannabis and 3,4-methylenedioxymethamphetamine (MDMA) have also been studied for use in treatment-resistant depression; however, because of limited evidence and safety concerns, they are not recommended.35,36
Nonpharmacologic Treatment Strategies
PSYCHOTHERAPY
In treatment-resistant depression and with partial response to initial treatment, psychotherapy is a recommended option.8–11 When added to usual care, it has demonstrated a short-term benefit in self-report data up to 6 months, but long-term evidence is lacking.37 No specific type of psychotherapy has been proven to be more effective than another. Switching the type of psychotherapy may be beneficial and is recommended because individual patient preference and need may be more adequately treated with different techniques.9,11,19
EXERCISE
Many studies have demonstrated improvement in major depressive disorder and depressive symptoms with the addition of exercise. Aerobic exercise has shown significant improvement in the severity of depression. Resistance training showed similar results, although the combination of aerobic and resistance training demonstrated a smaller effect than either form individually; this suggests that preference may play a role in achieving a better outcome.38 The intensity of exercise also adjusted the effect, with moderate activity showing a 47% improvement from baseline in symptoms.39 Having a structured exercise program, in comparison with unsupervised exercise, that meets the needs and ability of the patient shows an approximate 20% reduction in severity of depressive symptoms, which may be because of the individual tailoring of the exercise with patient's ability.40
NUTRITION HABITS AND SUPPLEMENTS
Dietary changes have been proposed to improve depression symptoms and remission. The SMILES trial (n = 67) enrolled patients with known major depression, with the intervention group receiving dietary support and advice on how to optimize their diet.41 After 12 weeks, the intervention group had improved depression-rating scores, and 32% had achieved remission compared with only 8% in the control group; however, the study has been criticized for its lack of blinding and concerns for bias.41,42 When used for several years, folate or B12 vitamins have shown some benefit by decreasing the risk of relapse; but they have shown no benefit in short-term use.43 St. John's wort demonstrated no benefit in a study of major depression.44
ELECTROCONVULSIVE THERAPY
Electroconvulsive therapy (ECT) has been shown to provide rapid, short-term improvement of severe symptoms but usually requires 6 to 12 total sessions that are generally given twice a week for 3 to 6 weeks.9 ECT is approved for those 13 years and older with treatment-resistant depression or catatonia or in those who need rapid response because of severe underlying medical or psychiatric conditions that pharmacologic therapy is unable or unlikely to provide.11,45 ECT decreases hospitalization rates and risk of suicide while increasing function and quality of life.46 In treatment-resistant depression, ECT improves response rates up to 80% and remission rates up to 60%.46 Cardiology consultation should be considered for patients with a known history of cardiac disease because adverse effects can include cardiac arrhythmias, although this is rare. Common adverse effects include jaw soreness, headache, fatigue, and cognitive impairment.46 Cognitive adverse effects vary with ECT techniques, but most are mild and resolve within 2 to 4 weeks.16,46
OTHER THERAPIES
Massage therapy has a lack of evidence supporting its beneficial effect on depression symptoms.47 Bright light therapy can reduce depression symptoms even for those whose depression is not seasonal. Studies are hindered, however, by lack of blinding and often small sample size.11 Implanted vagal nerve stimulation, in which a device is surgically implanted into the body and a wire is used to stimulate the afferent vagal nerve fibers, was found to have no major safety concerns, but the evidence of effectiveness is lacking. Several adverse effects can occur (e.g., voice change, cough, dysphonia), which may decrease over time.9,11
Repetitive transcranial magnetic stimulation uses magnetic fields within the brain to stimulate nerve cells involved in mood control. Repetitive transcranial magnetic stimulation thus can induce subconvulsive electrical cortical activity. In some studies, repetitive transcranial magnetic stimulation improved baseline depression-rating score by 50% (NNT = 3 to 9) and remission rates (NNT = 5 to 7) in patients with treatment-resistant depression.35 Although repetitive transcranial magnetic stimulation showed no major safety concerns, its effectiveness is only for short-term symptom relief because most studies were only 1 to 3 weeks and were of low quality.35 A U.S. Department of Veterans Affairs study noted no difference between active and sham treatment groups.48 Despite weak evidence, repetitive transcranial magnetic stimulation is approved by the U.S. Food and Drug Administration as a treatment option for treatment-resistant depression.11
Data Sources: A PubMed search was completed in Clinical Queries using the key terms depression, resistant depression, and refractory depression. The search included meta-analyses, randomized controlled trials, clinical trials, and reviews. The Agency for Healthcare Research and Quality Effective Healthcare Reports, the Cochrane database, DynaMed, and Essential Evidence Plus were also searched. We critically reviewed studies that used patient categories such as race and/or gender but did not define how these categories were assigned, stating their limitations in the text. Search dates: December 1, 2023, and April 1, 2024.

