Soluble fms-like tyrosine kinase-1 (sFlt-1) is a vascular endothelial growth factor and antagonist of placental growth factor (PlGF) that causes vasoconstriction and endothelial damage, which may result in preeclampsia.1 There is no standardized value for the sFlt-1/PlGF ratio in the diagnosis or exclusion of preeclampsia.2,3 The American College of Obstetricians and Gynecologists and the Society for Maternal-Fetal Medicine recommend against measurement of these angiogenic markers for diagnosis or exclusion of preeclampsia.4
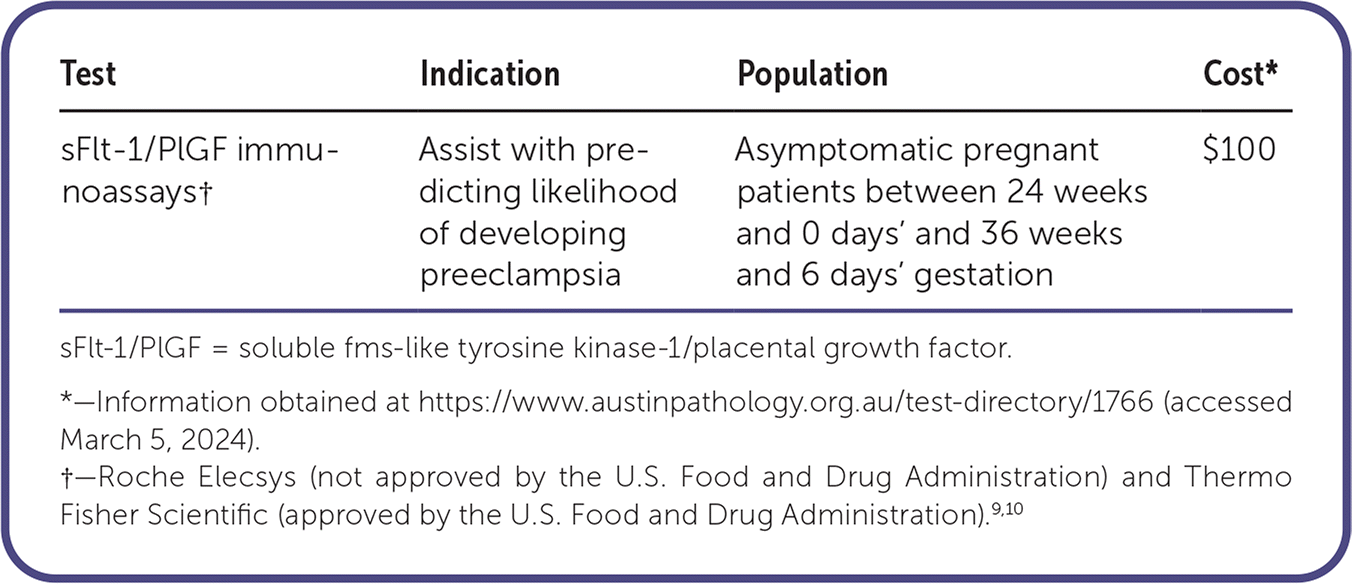
| Test | Indication | Population | Cost* |
|---|---|---|---|
| sFlt-1/PlGF immunoassays† | Assist with predicting likelihood of developing preeclampsia | Asymptomatic pregnant patients between 24 weeks and 0 days' and 36 weeks and 6 days' gestation | $100 |
sFlt-1/PlGF = soluble fms-like tyrosine kinase-1/placental growth factor.
*—Information obtained at https://www.austinpathology.org.au/test-directory/1766 (accessed March 5, 2024).
†—Roche Elecsys (not approved by the U.S. Food and Drug Administration) and Thermo Fisher Scientific (approved by the U.S. Food and Drug Administration).9,10
Accuracy
A two-phase, prospective, double-blind, multicenter, observational study of the sFlt-1/PlGF immunoassay aimed to derive (n = 500) and validate (n = 550) preeclampsia cutoffs. The study aimed to rule out preeclampsia for 1 week and rule in preeclampsia, eclampsia, and hemolysis, elevated liver enzymes, low platelet count (HELLP) syndrome within 4 weeks (based on blood pressure, laboratory values, or symptoms consistent with preeclampsia). The study included patients with a singleton pregnancy between 24 weeks and 0 days' and 36 weeks and 6 days' gestation.5 Using post hoc exploratory analyses and comparing laboratory values with the patients' clinical diagnoses, values greater than 38 correlated with preeclampsia, eclampsia, and HELLP syndrome. These values depicted a positive test (Table 1).1
TABLE 1. Development and Validation Data Using a Cutoff Point of Greater Than 38 for sFlt-1/PlGF Ratio in Predicting Preeclampsia
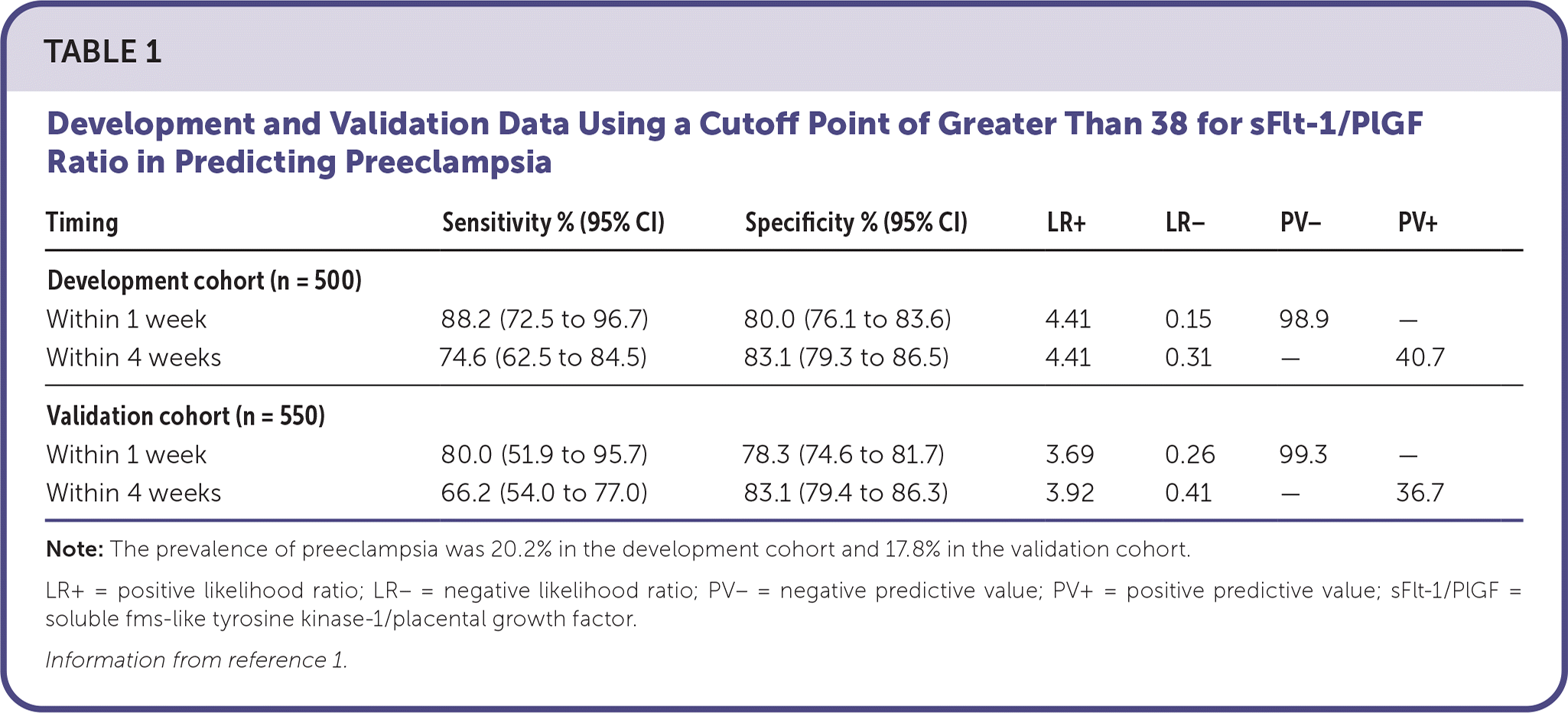
| Timing | Sensitivity % (95% CI) | Specificity % (95% CI) | LR+ | LR− | PV− | PV+ |
|---|---|---|---|---|---|---|
| Development cohort (n = 500) | ||||||
| Within 1 week | 88.2 (72.5 to 96.7) | 80.0 (76.1 to 83.6) | 4.41 | 0.15 | 98.9 | — |
| Within 4 weeks | 74.6 (62.5 to 84.5) | 83.1 (79.3 to 86.5) | 4.41 | 0.31 | — | 40.7 |
| Validation cohort (n = 550) | ||||||
| Within 1 week | 80.0 (51.9 to 95.7) | 78.3 (74.6 to 81.7) | 3.69 | 0.26 | 99.3 | — |
| Within 4 weeks | 66.2 (54.0 to 77.0) | 83.1 (79.4 to 86.3) | 3.92 | 0.41 | — | 36.7 |
Note: The prevalence of preeclampsia was 20.2% in the development cohort and 17.8% in the validation cohort.
LR+ = positive likelihood ratio; LR− = negative likelihood ratio; PV– = negative predictive value; PV+ = positive predictive value; sFlt-1/PlGF = soluble fms-like tyrosine kinase-1/placental growth factor.
Information from reference 1.
A systematic review and meta-analysis of 20 studies (10 prospective cohort and 10 case-control) including pregnant patients from 6 to 41 weeks' gestation with preeclampsia (n = 828) and without preeclampsia (n = 6,138) were completed. The study investigated the accuracy of the sFlt-1/PlGF ratio in predicting preeclampsia.3 The pooled diagnostic sensitivity and specificity of sFlt-1/PlGF testing were 0.78 (95% CI, 0.67 to 0.86) and 0.84 (95% CI, 0.77 to 0.89), respectively, with a 4.9 positive likelihood ratio and 0.26 negative likelihood ratio.3 Study size and differing cutoff values (ranging from 1.4 to 196) contributed to high heterogeneity. In the four studies that had cutoff levels near 35, sensitivity ranged from 0.76 to 1.00 and specificity from 0.74 to 0.95.3
Benefits
The sFlt-1/PlGF ratio can increase the likelihood of predicting preeclampsia. In a prospective randomized controlled study (n = 370), pregnant patients who met diagnostic criteria for preeclampsia received a positive diagnosis 100% of the time when the ratio was included, but only 83% of the time when the ratio was not used.6
Harms
The sFlt-1/PlGF ratio does not change hospital admission rates due to preeclampsia.6 Additionally, sFlt-1/PlGF ratios may be elevated in pregnancies with intrauterine growth restriction or intrauterine fetal demise.7
Cost
According to the Centers for Medicare and Medicaid Services, the cost of the sFlt-1/PlGF test is not covered, with commercial insurance likely to follow the same guidance. A retrospective cost analysis that used sFlt-1/PlGF ratios in the evaluation and management of preeclampsia in pregnant patients between 20 and 34 weeks' gestation found an out-of-pocket cost of around $100 per test.8 The two manufacturers are Roche Elecsys (not approved by the U.S. Food and Drug Administration) and Thermo Fisher Scientific (approved by the U.S. Food and Drug Administration).9,10
Bottom Line
The sFlt-1/PlGF ratio cutoff must be standardized and further validated with additional clinical trials before implementation in the prediction and diagnosis of preeclampsia.
The opinions and assertions contained herein are the private views of the author and are not to be construed as official or as reflecting the views of the U.S. Army Medical Department or the U.S. Army at large.

