Substance misuse and substance use disorder continue to be major causes of morbidity and mortality, and family physicians are well positioned to provide evidence-based prevention and management for these conditions. Of people 12 years and older, 13% reported using a nonprescribed controlled substance in the past month, and 24% had at least one episode of binge drinking of alcohol, defined as five or more drinks for men and four or more drinks for women on one occasion. Benzodiazepines are used by 12% of the U.S. population. Clinicians should incorporate standardized screening and brief intervention for use of alcohol and other substances into routine care of adult patients, as well as referral to specialized treatment services when indicated. Use of nonstigmatizing, person-first language has been shown to positively affect care for patients with substance use disorders. Alcohol screening and brief intervention have been shown to reduce excessive drinking by 40% in patients at 6 months postintervention. Office-based treatment of alcohol use disorder with medications approved by the U.S. Food and Drug Administration, such as acamprosate and naltrexone, remains underutilized, presenting another opportunity for family physicians to positively affect the health of their patients and communities. With elimination of the X-waiver, any clinician with Schedule III prescriptive authority can treat opioid use disorder with buprenorphine in their office-based practice. Opioid overdose education and naloxone coprescribing are other tools family physicians can employ to combat the overdose crisis.
The medical approach to substance use has significantly changed over the past decade. Decreases in opioid prescribing and increases in synthetic opioid availability have fueled a crisis of overdose deaths, exceeding 100,000 U.S. deaths annually.1 In response, research and funding priorities have shifted, and the medical profession has adopted new tools for prevention, screening, assessment, and treatment. The American Academy of Family Physicians strongly supports family physicians and residents incorporating diagnosis, treatment, and prevention of substance use disorders (SUDs) into their routine clinical practices.2,3 For purposes of this article, “substance use disorder” indicates that an individual meets the specicfic Diagnostic and Statistical Manual of Mental Disorders, 5th ed. (DSM-5), criteria for a substance use disorder 4; substance misuse indicates not meeting those criteria for the disorder and using in hazardous situations or above limits for low-risk alcohol use. This article shares key updates in the primary care field and focuses on adult care. An article focusing on adolescents has been previously published in American Family Physician.5
SORT: KEY RECOMMENDATIONS FOR PRACTICE
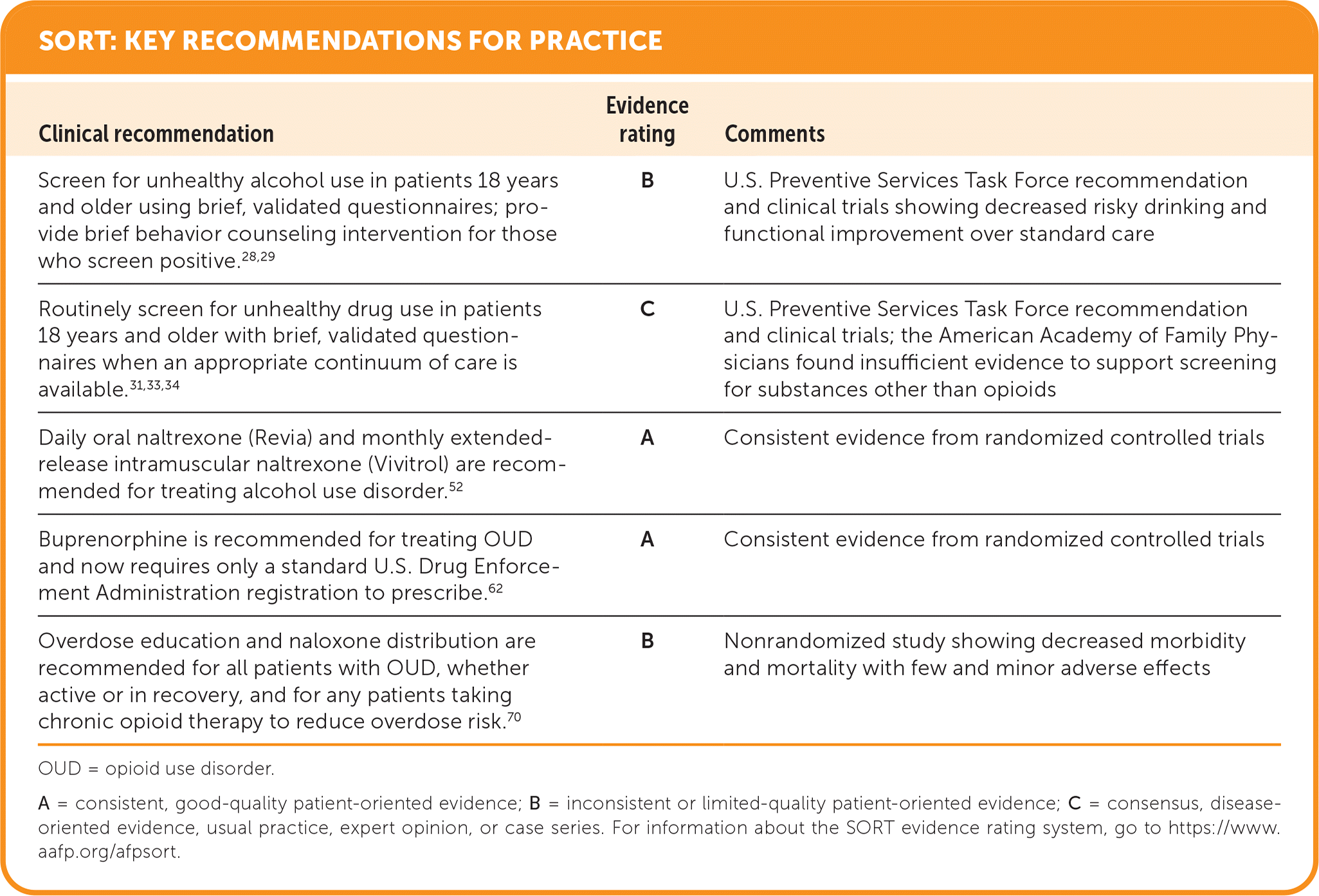
OUD = opioid use disorder.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.
Epidemiology of Substance Use
Substance misuse, morbidity, and mortality are increasing across nearly all classes of substances and populations. In 2022, of people 12 years and older, 16.5% reported using an illicit drug in the past month, and 22% had at least one episode of binge drinking of alcohol, defined as five or more drinks for men and four or more drinks for women on one occasion.6 Drug-related overdose deaths have risen 500% since 2001, topping 100,000 deaths annually since 2021.7,8
Alcohol. As of 2018, more than one-half of adults regularly consume alcohol, and 11% had an alcohol use disorder (AUD) in 2021.9,10 The National Institute on Alcohol Abuse and Alcoholism attributes approximately 95,000 deaths per year to alcohol use, including 28% of all motor vehicle–involved deaths.11
Benzodiazepines. Benzodiazepines are used by 9% of the U.S. population, with 1 in 7 reporting misuse in the past year.12 Misuse is strongly associated with diversion and combined misuse of opioids.13 When benzodiazepines are combined with opioids or alcohol, the potential for overdose increases significantly.14
Cannabis. Use of cannabis is an emerging concern. As of November 2023, recreational use of cannabis is legal in 24 states and Washington, D.C., and Guam, resulting in growing social acceptance and use. In 2021, cannabis use was reported by 18.7% of the U.S. population, and 5.8% of the U.S. population had a cannabis use disorder.15 A commonly reported misconception is that cannabis is not addictive.16 The psychoactive potency of cannabis has increased sharply over time, and newer formulations of edible cannabis and cannabis extracts, which are vaporized for inhalation through a dab rig, are available. Both formulations can result in increased adverse reactions, including cannabinoid hyperemesis syndrome.17 Although cannabis-related emergency department visits are increasing, no overdose deaths have been attributed to cannabis use alone.18
Opioids. Beginning in the early 2000s, opioid manufacturers promoted misinformation about the safety of prescription opioids, initiating the overdose crisis from within the health care industry.19 Despite policy and prescribing patterns shifting away from opioid overprescribing, overdose deaths have accelerated as use shifted first to heroin and then to more potent synthetic opioids.1 The increasing availability of synthetic opioids in unregulated markets has mirrored the steep trends showing skyrocketing overdose deaths.1
Stimulants. Deaths from stimulants (e.g., methamphetamine, cocaine) have increased fivefold in the past decade to more than 53,000 in 2021.1,20
Stigma
Fewer than 5% of people with an SUD seek treatment in a given year, and stigma is a well-established barrier.12 Use of nonstigmatizing, person-first language (e.g., person with an SUD) and avoidance of stigmatizing terminology (e.g., addict, abuser) has been shown to positively affect care.21 The National Institute on Drug Abuse provides guidance on nonstigmatizing terminology.22 Self-stigma is associated with low self-efficacy and may hinder accurate disclosure of use.23
Unconscious bias may prevent health care professionals from asking their patients about substance use. Stigmatizing attitudes from clinicians can lead to under-treatment of patients with SUDs and affect patients' sense of safety to discuss usage.24 The continued disproportionate criminalization of substance use in Black and Hispanic/Latino communities also increases stigma for patients with SUDs from these communities.25 Black and Hispanic patients are less likely to be prescribed medication for opioid use disorder (OUD) than White patients, and racial and ethnic minority adults are one-third less likely to receive treatment services for AUD.26,27 This disparity increases when accounting for socioeconomic and clinical confounders.26,27 It is critical for clinicians to examine and address their own social biases and their biases' effect on care.
Screening and Brief Intervention
The U.S. Preventive Services Task Force recommends screening, with brief behavior counseling interventions, for unhealthy alcohol use in patients 18 years and older.28–30 Alcohol screening and brief intervention have been shown to reduce excessive drinking in patients with alcohol misuse by 40% at 6 months postintervention.29
The U.S. Preventive Services Task Force also recommends screening for unhealthy drug use when an appropriate continuum of care is available.31,32 The American Academy of Family Physicians found insufficient evidence to support screening for substances other than opioids; it supports selectively screening adults for OUD when appropriate resources are available.33–35
Several validated screening tools are available for specific populations, substances, clinical settings, and time constraints (Table 1).36–42 Incorporation into electronic health record systems and routine clinic workflows allows for systematic and efficient stratification of patients' substance use along a continuum of abstinence, low-risk use, misuse, and likely use disorder.43
TABLE 1. Validated Screening Tools for Substance Use
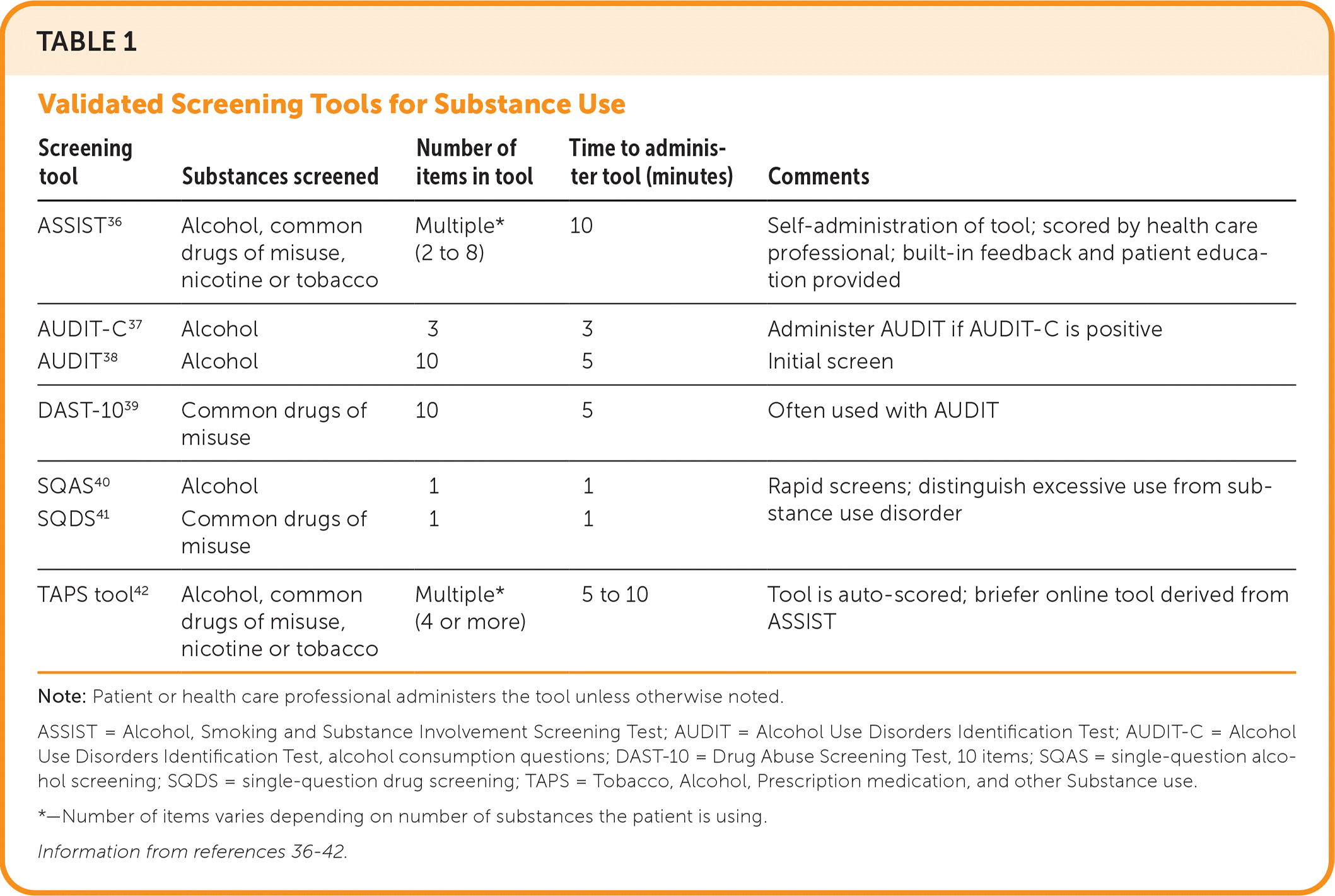
| Screening tool | Substances screened | Number of items in tool | Time to administer tool (minutes) | Comments |
|---|---|---|---|---|
| ASSIST36 | Alcohol, common drugs of misuse, nicotine or tobacco | Multiple* (2 to 8) | 10 | Self-administration of tool; scored by health care professional; built-in feedback and patient education provided |
| AUDIT-C37 | Alcohol | 3 | 3 | Administer AUDIT if AUDIT-C is positive |
| AUDIT38 | Alcohol | 10 | 5 | Initial screen |
| DAST-1039 | Common drugs of misuse | 10 | 5 | Often used with AUDIT |
| SQAS40 | Alcohol | 1 | 1 | Rapid screens; distinguish excessive use from substance use disorder |
| SQDS41 | Common drugs of misuse | 1 | 1 | |
| TAPS tool42 | Alcohol, common drugs of misuse, nicotine or tobacco | Multiple* (4 or more) | 5 to 10 | Tool is auto-scored; briefer online tool derived from ASSIST |
Note: Patient or health care professional administers the tool unless otherwise noted.
ASSIST = Alcohol, Smoking and Substance Involvement Screening Test; AUDIT = Alcohol Use Disorders Identification Test; AUDIT-C = Alcohol Use Disorders Identification Test, alcohol consumption questions; DAST-10 = Drug Abuse Screening Test, 10 items; SQAS = single-question alcohol screening; SQDS = single-question drug screening; TAPS = Tobacco, Alcohol, Prescription medication, and other Substance use.
*—Number of items varies depending on number of substances the patient is using.
Brief interventions incorporate motivational interviewing principles to enhance desire and willingness for behavior change.44 During a brief intervention, the physician, behaviorist, or other trained health professional provides nonjudgmental feedback on screening results, links the patient's use to current health concerns, evokes the patient's own reasons for change, and encourages exploration of ambivalence. For patients ready to change, brief interventions include collaborating on next steps, identifying patient strengths and available resources, and troubleshooting barriers.45
Assessment and Diagnosis
For patients who screen positive or who present seeking treatment for their substance use, physicians can further assess severity of use through a nonjudgmental conversation about substances currently being used, consumption routes, quantity and frequency of use, and effects on psychosocial functioning. SUDs are chronic, treatable diseases characterized by 11 diagnostic criteria grouped into four main categories: impaired control, social impairment, risky use, and pharmacologic criteria.46 Mirroring the changing medical approach, the DSM-5 presents a significant shift from earlier editions in its approach to substance use.4 Previously used terms abuse and dependence are now called SUDs. They range from mild to severe, depending on the number of diagnostic criteria present: Mild is two or three criteria, moderate is four or five, and severe is six or more47 (Table 24 ).
SUD Management
Depending on SUD severity and patient preference, management using a chronic care approach with longitudinal coordination of care among an interprofessional team may include pharmacotherapy, mutual aid groups, peer recovery support, specialty treatment, and behavior counseling.48,49 The U.S. Department of Veterans Affairs/Department of Defense provides a clinical practice guideline as a comprehensive, actionable resource for delineating workflows and clinical decision-making points for the management of SUDs.50
PHARMACOTHERAPY
Several medications are approved by the U.S. Food and Drug Administration for treating AUDs and OUDs in the office-based setting; however, office-based treatment of AUD and OUD with these medications remains underutilized.
Alcohol. Medical management of alcohol withdrawal may be needed before initiating AUD treatment and can be accomplished in the ambulatory setting for some using GABAergics or benzodiazepines; GABAergics are preferred because of their lower risk for misuse and overdose.51
For AUD treatment and relapse prevention, a noncontrolled opioid antagonist, such as daily oral naltrexone (Revia) or extended-release intramuscular naltrexone (Vivitrol), can be prescribed.52 Acamprosate, an oral medication taken two or three times daily, is also considered a first-line treatment.53 Several GABAergics, such as gabapentin and topiramate, can be used off-label as second-line treatment.54,55 Disulfiram, although approved by the U.S. Food and Drug Administration, is considered third line.56 Studies of psychedelics such as ketamine and psilocybin for treating AUD, as well as glucagon-like peptide-1 agonists such as semaglutide (available as brand Ozempic), are ongoing, with none currently endorsed for use in treating AUD or other SUDs outside of clinical trials.57,58 Table 3 provides additional information on medications for AUD.51–56,59 More information about prescribing medications for AUD is available in a previous American Family Physician article.60
TABLE 3. Medications for Alcohol Use Disorder
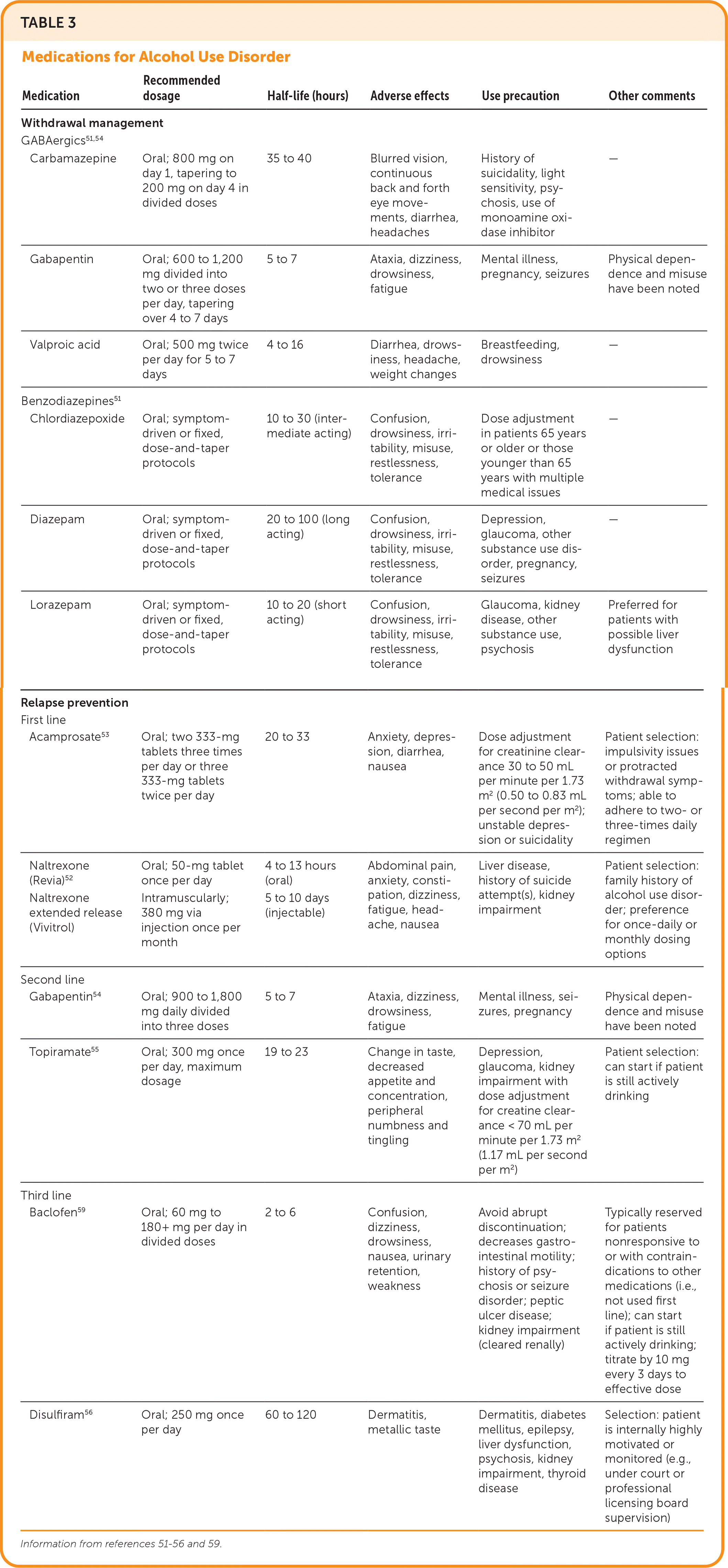
| Medication | Recommended dosage | Half-life (hours) | Adverse effects | Use precaution | Other comments |
|---|---|---|---|---|---|
| Withdrawal management | |||||
| GABAergics51,54 | |||||
| Carbamazepine | Oral; 800 mg on day 1, tapering to 200 mg on day 4 in divided doses | 35 to 40 | Blurred vision, continuous back and forth eye movements, diarrhea, headaches | History of suicidality, light sensitivity, psychosis, use of monoamine oxidase inhibitor | — |
| Gabapentin | Oral; 600 to 1,200 mg divided into two or three doses per day, tapering over 4 to 7 days | 5 to 7 | Ataxia, dizziness, drowsiness, fatigue | Mental illness, pregnancy, seizures | Physical dependence and misuse have been noted |
| Valproic acid | Oral; 500 mg twice per day for 5 to 7 days | 4 to 16 | Diarrhea, drowsiness, headache, weight changes | Breastfeeding, drowsiness | — |
| Benzodiazepines51 | |||||
| Chlordiazepoxide | Oral; symptom-driven or fixed, dose-and-taper protocols | 10 to 30 (intermediate acting) | Confusion, drowsiness, irritability, misuse, restlessness, tolerance | Dose adjustment in patients 65 years or older or those younger than 65 years with multiple medical issues | — |
| Diazepam | Oral; symptom-driven or fixed, dose-and-taper protocols | 20 to 100 (long acting) | Confusion, drowsiness, irritability, misuse, restlessness, tolerance | Depression, glaucoma, other substance use disorder, pregnancy, seizures | — |
| Lorazepam | Oral; symptom-driven or fixed, dose-and-taper protocols | 10 to 20 (short acting) | Confusion, drowsiness, irritability, misuse, restlessness, tolerance | Glaucoma, kidney disease, other substance use, psychosis | Preferred for patients with possible liver dysfunction |
| Relapse prevention | |||||
| First line | |||||
| Acamprosate53 | Oral; two 333-mg tablets three times per day or three 333-mg tablets twice per day | 20 to 33 | Anxiety, depression, diarrhea, nausea | Dose adjustment for creatinine clearance 30 to 50 mL per minute per 1.73 m2 (0.50 to 0.83 mL per second per m2); unstable depression or suicidality | Patient selection: impulsivity issues or protracted withdrawal symptoms; able to adhere to two- or three-times daily regimen |
| Naltrexone (Revia)52 Naltrexone extended release (Vivitrol) | Oral; 50-mg tablet once per day Intramuscularly; 380 mg via injection once per month | 4 to 13 hours (oral) 5 to 10 days (injectable) | Abdominal pain, anxiety, constipation, dizziness, fatigue, headache, nausea | Liver disease, history of suicide attempt(s), kidney impairment | Patient selection: family history of alcohol use disorder; preference for once-daily or monthly dosing options |
| Second line | |||||
| Gabapentin54 | Oral; 900 to 1,800 mg daily divided into three doses | 5 to 7 | Ataxia, dizziness, drowsiness, fatigue | Mental illness, seizures, pregnancy | Physical dependence and misuse have been noted |
| Topiramate55 | Oral; 300 mg once per day, maximum dosage | 19 to 23 | Change in taste, decreased appetite and concentration, peripheral numbness and tingling | Depression, glaucoma, kidney impairment with dose adjustment for creatine clearance < 70 mL per minute per 1.73 m2 (1.17 mL per second per m2) | Patient selection: can start if patient is still actively drinking |
| Third line | |||||
| Baclofen59 | Oral; 60 mg to 180+ mg per day in divided doses | 2 to 6 | Confusion, dizziness, drowsiness, nausea, urinary retention, weakness | Avoid abrupt discontinuation; decreases gastrointestinal motility; history of psychosis or seizure disorder; peptic ulcer disease; kidney impairment (cleared renally) | Typically reserved for patients nonresponsive to or with contraindications to other medications (i.e., not used first line); can start if patient is still actively drinking; titrate by 10 mg every 3 days to effective dose |
| Disulfiram56 | Oral; 250 mg once per day | 60 to 120 | Dermatitis, metallic taste | Dermatitis, diabetes mellitus, epilepsy, liver dysfunction, psychosis, kidney impairment, thyroid disease | Selection: patient is internally highly motivated or monitored (e.g., under court or professional licensing board supervision) |
Opioids. For OUD, recent elimination of the X-waiver for prescribing buprenorphine-based medication and permanent adoption of pandemic-enacted telehealth provisions allowing for audio-only and audiovisual office-based opioid treatment visits enable family physicians to more readily incorporate this medicine into their practices.61 Buprenorphine is a partial opioid agonist that decreases morbidity and mortality and increases abstinence rates in patients with OUD.62 Methadone, a full opioid agonist OUD medication with use currently restricted to licensed opioid treatment programs, can be considered first-line treatment, especially with take-home doses now federally approved from the start of treatment or when a patient with OUD continues to experience cravings, withdrawal, or relapses on maximally tolerated buprenorphine doses.61,63,64
For OUD treatment, naltrexone extended release has shown effectiveness only in the monthly, intramuscular formulation and can be started after an opioid-free period of 10 days.65 If patients present to primary care after a period of abstinence such as a recent hospitalization, they may be candidates for this approach. A test dose of naloxone is recommended before providing long-acting naltrexone to avoid precipitating withdrawal.66 Table 4 provides additional information on medications for OUD.66
TABLE 4. Medications for Opioid Use Disorder
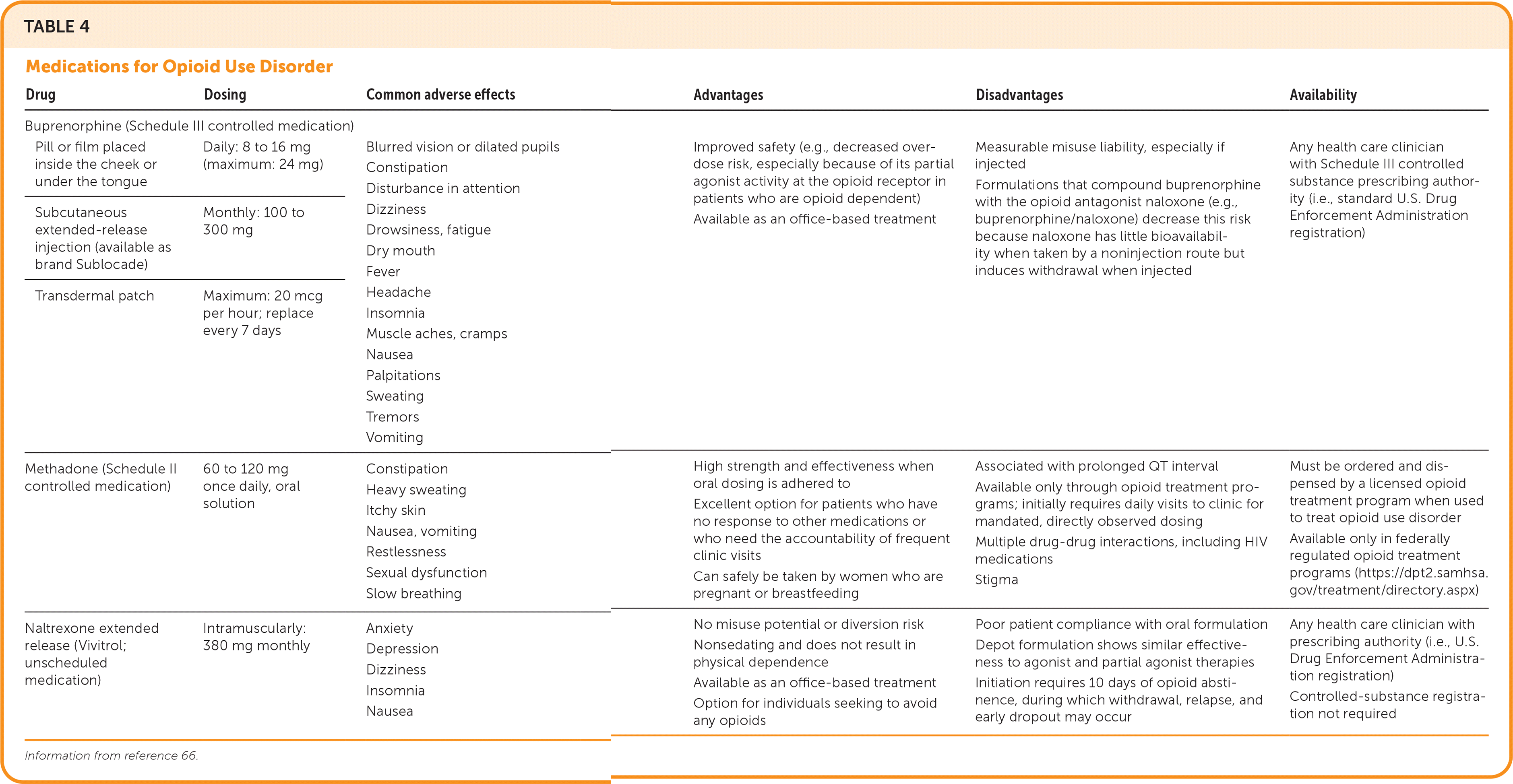
| Drug | Dosing | Common adverse effects | Advantages | Disadvantages | Availability |
|---|---|---|---|---|---|
| Buprenorphine (Schedule III controlled medication) | |||||
| Pill or film placed inside the cheek or under the tongue | Daily: 8 to 16 mg (maximum: 24 mg) | Blurred vision or dilated pupils Constipation Disturbance in attention Dizziness Drowsiness, fatigue Dry mouth Fever Headache Insomnia Muscle aches, cramps Nausea Palpitations Sweating Tremors Vomiting | Improved safety (e.g., decreased over-dose risk, especially because of its partial agonist activity at the opioid receptor in patients who are opioid dependent) Available as an office-based treatment | Measurable misuse liability, especially if injected Formulations that compound buprenorphine with the opioid antagonist naloxone (e.g., buprenorphine/naloxone) decrease this risk because naloxone has little bioavailability when taken by a noninjection route but induces withdrawal when injected | Any health care clinician with Schedule III controlled substance prescribing authority (i.e., standard U.S. Drug Enforcement Administration registration) |
| Subcutaneous extended-release injection (available as brand Sublocade) | Monthly: 100 to 300 mg | ||||
| Transdermal patch | Maximum: 20 mcg per hour; replace every 7 days | ||||
| Methadone (Schedule II controlled medication) | 60 to 120 mg once daily, oral solution | Constipation Heavy sweating Itchy skin Nausea, vomiting Restlessness Sexual dysfunction Slow breathing | High strength and effectiveness when oral dosing is adhered to Excellent option for patients who have no response to other medications or who need the accountability of frequent clinic visits Can safely be taken by women who are pregnant or breastfeeding | Associated with prolonged QT interval Available only through opioid treatment programs; initially requires daily visits to clinic for mandated, directly observed dosing Multiple drug-drug interactions, including HIV medications Stigma | Must be ordered and dispensed by a licensed opioid treatment program when used to treat opioid use disorder Available only in federally regulated opioid treatment programs (https://dpt2.samhsa.gov/treatment/directory.aspx) |
| Naltrexone extended release (Vivitrol; unscheduled medication) | Intramuscularly: 380 mg monthly | Anxiety Depression Dizziness Insomnia Nausea | No misuse potential or diversion risk Nonsedating and does not result in physical dependence Available as an office-based treatment Option for individuals seeking to avoid any opioids | Poor patient compliance with oral formulation Depot formulation shows similar effectiveness to agonist and partial agonist therapies Initiation requires 10 days of opioid abstinence, during which withdrawal, relapse, and early dropout may occur | Any health care clinician with prescribing authority (i.e., U.S. Drug Enforcement Administration registration) Controlled-substance registration not required |
Information from reference 66.
Benzodiazepines, cannabis, stimulants, and hallucinogenics. Management of benzodiazepine withdrawal is typically conducted in the ambulatory setting through gradual dose reduction.67 No medications are approved by the U.S. Food and Drug Administration for the treatment of cannabis, stimulant, or hallucinogen use disorder. Research shows promise for mirtazapine and combination therapy with bupropion and injectable naltrexone extended release as treatments for stimulant use disorder.68,69
Overdose education and naloxone distribution should be offered to all patients with OUD, whether active or in recovery, and to any patient taking chronic opioid therapy to reduce overdose risk.70
BEHAVIOR THERAPY
In primary care practices with integrated behavior health care, physicians can facilitate referral to counseling by introducing the patient to the on-site behaviorist. This warm hand-off process enhances patient engagement, retention, and satisfaction with behavior health care, increasing attendance rates from 50% to more than 90%.71
Cognitive behavior therapy and contingency management are evidence-based behavior treatments for stimulant and other use disorders that can be provided in an integrated practice setting or through referral to community SUD treatment programs. Cognitive behavior therapy involves efforts to change unhelpful thinking and behavior patterns. Contingency management, based on operant conditioning, uses nonsubstance rewards (e.g., vouchers for goods, services) to reinforce positive behavior change.72,73
REFERRAL
Patients who have severe SUD, have unstable co-occurring disorders, lack psychosocial supports, or are not responding to SUD treatment in the office-based setting should be escalated to a more intensive level of care.50 The Substance Abuse and Mental Health Services Administration provides a nationwide, searchable database of licensed treatment facilities and opioid treatment programs (https://findtreatment.gov/).
COMORBIDITIES
Mental health. Comorbidities with mental health disorders, including depression, anxiety disorders, posttraumatic stress disorder (PTSD), and personality disorders, commonly occur in patients with SUD.74–76 Substance use and mental health disorders may present similarly, and comprehensive screening and assessment can reduce mis-diagnosis. Evidence-based behavior therapies (e.g., cognitive behavior therapy, dialectical behavior therapy, assertive community treatment, exposure therapy, contingency management) can be provided alone or in combination with medications.77
Intimate partner violence. Patients who misuse substances are at greater risk of experiencing or being perpetrators of intimate partner violence, making screening important.78 During the COVID-19 pandemic, rates of physical intimate partner violence and severity of related injuries increased with more instances of substance use, depression, and anxiety; this highlights the association between mental health, intimate partner violence, and SUDs and substance misuse.79,80
Trauma. Trauma is a risk factor for behavior health disorders, including SUDs.81 Patients with trauma history may display inconsistent patterns of clinical care, including missed appointments and delayed health maintenance. Physicians should employ a trauma informed–care approach.82 Practices with integrated behavior health services can provide Seeking Safety, an evidence-based treatment for co-occurring substance use and trauma.83 Its manualized, flexible design allows delivery in individual, group, in-person, or virtual sessions, covering one or more of 25 treatment topics.83 The Primary Care PTSD Screen for DSM-5 is a useful tool for PTSD screening within health care and SUD treatment settings.84
Medical. Substance use and SUDs increase the risk of medical comorbidities (e.g., HIV, hepatitis, cardiovascular disease, chronic pain, cancer) and can negatively affect treatment access, engagement, and adherence.85 Patients with active SUDs may also have significant gaps in preventive health care. Xylazine, used in veterinarian medicine as a procedural anesthetic and sedative, is an alpha 2 agonist not approved for human use because of its sedative effects. It is increasingly present in opioids, commonly sold as “tranq,” in unregulated U.S. markets. Use of xylazine can cause deep pressure ulcers and necrotizing wounds, requiring lengthy and intensive managment.86
This article updates previous articles on this topic by Shapiro, et al.87; and Mersy.88
Data Sources: Essential Evidence Plus and PubMed were searched using key terms substance use, substance use disorder, substance misuse, alcohol use, alcohol use disorder, co-occurring disorders, and overdose. Meta-analyses, randomized controlled trials, clinical trials, and reviews were included. The Agency for Healthcare Research and Quality Effective Healthcare Reports, the Cochrane Database of Systematic Reviews, and the U.S. Preventive Services Task Force were also searched. Search date: March 9, 2024.

